Commentary
Homocysteine (Hcy) metabolism is at the intersection of two metabolic pathways: remethylation and trans-sulfuration, which abnormality can result in hyperhomocysteinemia (HHcy). In the past few years our team has been working on the molecular mechanism of HHcy development. We have discovered that cystathionine-β?synthase (CBS), a key enzyme in the trans-sulfuration pathway of Hcy metabolism, can be nitrated at Tyr163, Tyr223, Tyr381 and Tyr518 residues, nitration blunts its enzymatic activity and results in HHcy [1]. Next, we tried to explore further in the post-translational modification (PTM) of trans-sulfuration pathway [2], focused on the PTM of Sp1-CSE-H2S pathway and revealed four major findings: (1) CSE, another key enzyme in the trans-sulfuration pathway, can be nitrated under physiological condition, Hcy-accumulation augmented its nitration, inhibited its bio-activity, and thus caused hydrogen sulfide (H2S) deficiency. (2) H2S deficiency alleviated the S-sulfhydration of Sp1 (Specificity protein 1, a transcription factor of CSE), thus down-regulated CSE level and decreased the H2S level further, which in turn weakened its own S-sulfhydration. (3) The S-sulfhydration sites of CSE were Cys84, Cys109, Cys172, Cys229, Cys252, Cys307 and Cys310 under physiological condition, among which the S-sulfhydration of Cys84, Cys109, Cys229, Cys252 and Cys307 was necessary for its normal bio-activity. (4) H2S deficiency could trap Hcy-metabolism into a vicious cycle, which could be broken by either blocking nitration or restoring S-sulfhydration. It seems to be the end of our story, however, another protein enters our sight. In a newly published report of our team [3], we discovered that methylenetet rahydrofolate reductase (MTHFR), a key enzyme in remethlytion pathway, could be S-sulfhydrated by H2S under physiological conditions. Cysteine residues 32, 130, 131, 193, and 306 were sites at which MTHFR was S-sulfhydrated, and that these residues were essential for its bioactivity. Our results suggested that regulation of MTHFR bioactivity was not only due to polymorphisms or phosphorylation, but also an outcome of change in S-sulfhydration level.
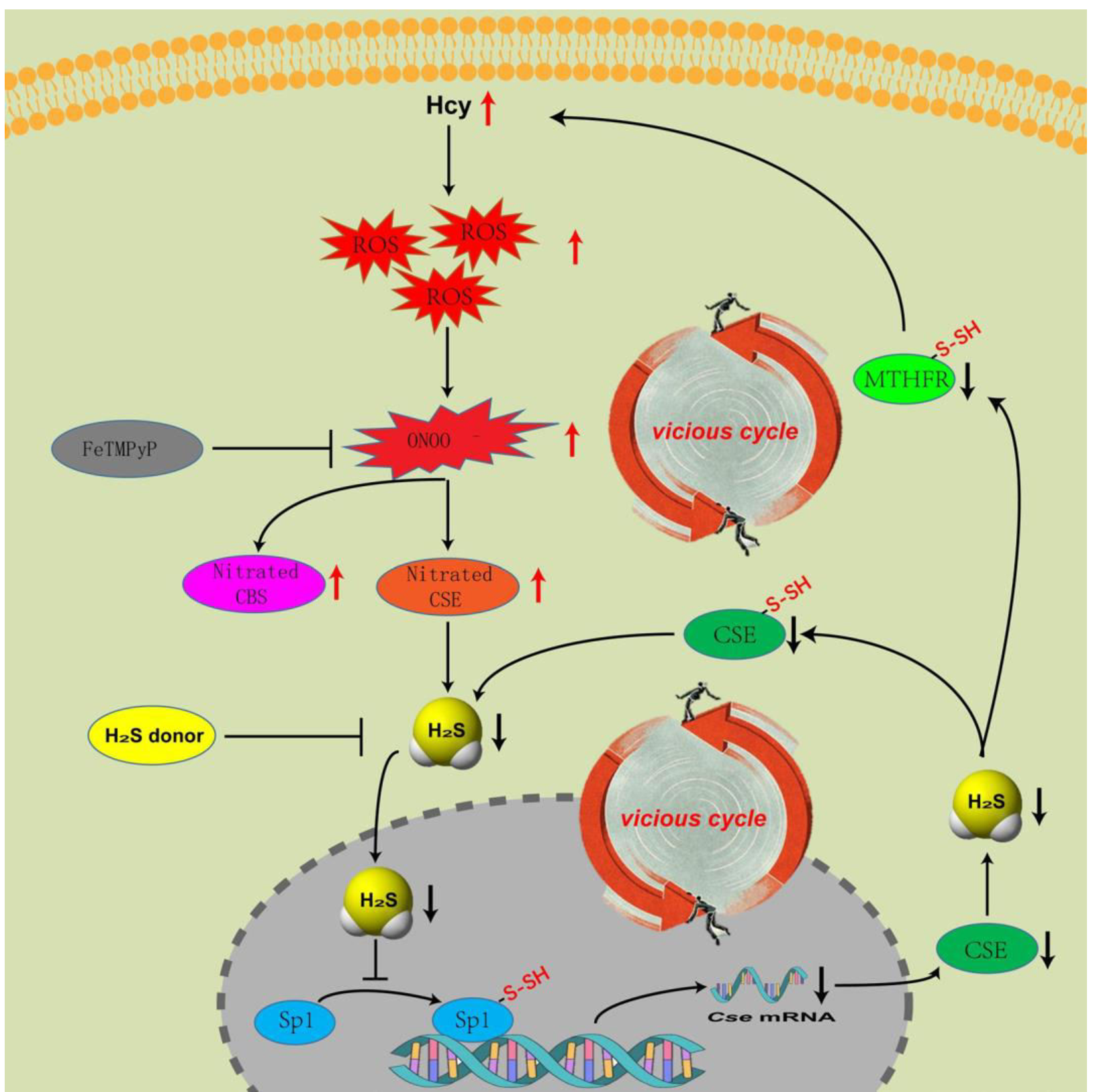
Based on the recent findings [1-3], we can primarily draw a mechanism diagram about the relationship between nitration and S-sulhydration in the metabolism of Hcy (Graphical abstract). In the trans-sulfuration pathway, Hcy accumulation augments the nitration of CBS [1] and CSE [2], blunts their enzymatic activity, lowers H2S production, which subsequently decreases the S-sulfhydration of Sp1. Insufficient S-sulfhydration of Sp1 down-regulates the transcription of Cse. The level of H2S decreases further with lapsed time, which in turn induces the S-sulfhydration deficiency of CSE. In the end, declines in both quality (activity) and quantity (expression) of CSE occur. What’s worse, in the remethylation pathway [3], MTHFR’s activity was inhibited due to H2S deficiency caused by the blunt of trans-sulfuration pathway. Finally, the progress of HHcy falls into a vicious cycle. Our team provided a novel insight into the progress of HHcy in terms of the mechanism, which highlighted the vicious cycle caused by the abnormality of nitration and S-sulfhydration.
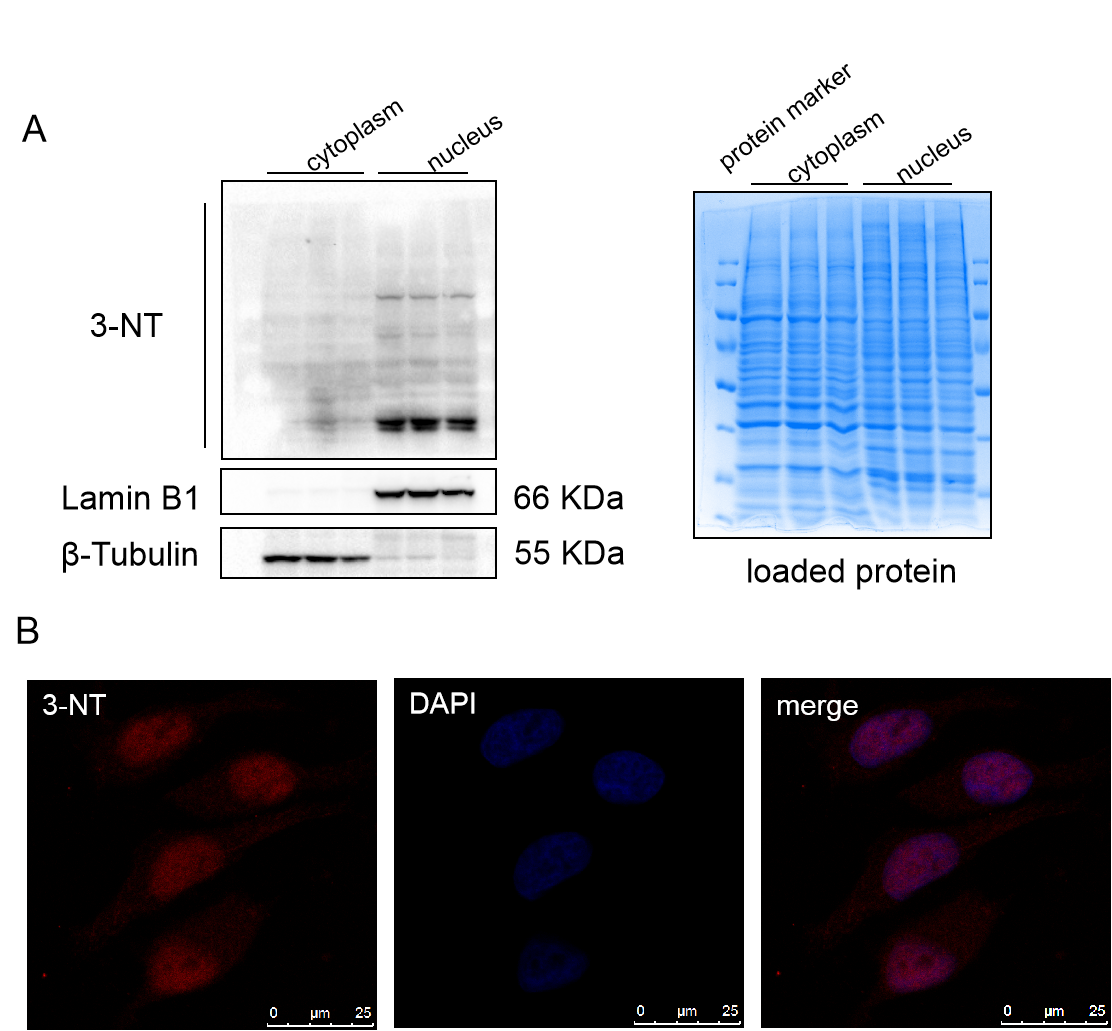
Our study confirmed that nitration inhibited the biological activity of CBS and CSE, and then, what about the nitration of Sp1 or other proteins? We tried to detect the nitration of Sp1, and surprisingly found that the level of nitration of Sp1 remained virtually unchanged after treatment with Hcy or other reagents (SIN-1, FeTMPyP, or hydrogen sulfide donors), either in vivo or in vitro. Further, we detected the location of 3-NT in QSG-7701 cells by immunofluorescence and nucleoplasmic separation, and found that similar to Sp1, 3-NT was mostly distributed in the nucleus (Figure 1). These interesting findings suggest that the level of nitration maybe necessary to maintain the physiological function of Sp1. In general, nitration can deactivate the biological activity of the target protein, thus acting as an inhibitor or blocker. However, in some cases, nitration of proteins also plays a positive role. Balafanova et al. [4] found that the nitrification modification of PKCε can promote its binding to RACK2 and enhance the transcriptional activity of the latter. The study of Yakovlev et al. [5] showed that nitration modification of p53 can promote its oligomerization and activation. Similarly, many transcription factors are also localized in the nucleus, so is nitration modification necessary for the biological activity of these transcription factors? Will be worthy of study.
Figure 1. 3-NT mainly distributes in nucleus. (A) We separated the cytoplasm and nucleus of QSG-7701 cell, detected the sub-cellular distribution of 3-NT by western blot, the right is coomassie bright blue staining of the protein, n=3. (B) Immunofluorescence was used to detect the sub-cellular distribution of 3-NT, bar: 25 μm.
The vicious cycle here we found is in the metabolism of Hcy, and similar vicious cycles are prevalent in the occurrence and development of cancer. Emmet et al. found a vicious cycle of proliferation, synaptic integration and brain hyperactivity in the progression of glioblastoma (GBM) [6]. In a recent study on Esophageal squamous cell carcinoma (ESCC), Li et al. demonstrated that GSK3? interacts with and phosphorylates SOX2 at residue S251, which blocks SOX2 from ubiquitination and proteasome-dependent degradation instigated by ubiquitin E3 ligase CUL4ADET1-COP1. Interestingly they found that SOX2 enhanced GSK3? expression transcriptionally, suggesting the existence of a vicious cycle that drives a coordinated GSK3? and SOX2 overexpression in ESCC cells [7]. In a recent study on brain-metastasizing melanoma cells and microglia, Sivan et al. discovered that Microglia cells exposed to melanoma-derived IL-6 exhibited upregulated levels of STAT3 phosphorylation and SOCS3 expression, which, in turn, formed a vicious cycle and promoted melanoma cell viability and metastatic potential [8]. Chang et al. discovered that primary PCa expressing ADAM9 protein enables the transformation of OBs into PCa-associated osteoblasts (PCa-OBs), inducing WISP-1 secretion from PCa-OBs in the bone microenvironment. The upregulation of WISP-1 in bone provided feedback to primary PCa, formed a vicious cycle and promoted PCa cell aggressiveness via epithelial-mesenchymal transition (EMT) activity [9]. Cancer cells often express high levels of CBS and (or) CSE to produce more endogenous hydrogen sulfide [10,11]. Therefore, whether there is a vicious cycle caused by S-sulfhydrylation or nitrification modification in the Sp1-CSE-H2S pathway of cancer cells, thus promoting the occurrence and development of cancer, will be a topic worthy of further investigation.
The vicious cycle here we found is in the metabolism of Hcy, and similar vicious cycles are prevalent in the occurrence and development of cancer. Emmet et al. found a vicious cycle of proliferation, synaptic integration and brain hyperactivity in the progression of glioblastoma (GBM) [6]. In a recent study on Esophageal squamous cell carcinoma (ESCC), Li et al. demonstrated that GSK3β interacts with and phosphorylates SOX2 at residue S251, which blocks SOX2 from ubiquitination and proteasome-dependent degradation instigated by ubiquitin E3 ligase CUL4ADET1-COP1. Interestingly they found that SOX2 enhanced GSK3β expression transcriptionally, suggesting the existence of a vicious cycle that drives a coordinated GSK3β and SOX2 overexpression in ESCC cells [7]. In a recent study on brain-metastasizing melanoma cells and microglia, Sivan et al. discovered that Microglia cells exposed to melanoma-derived IL-6 exhibited upregulated levels of STAT3 phosphorylation and SOCS3 expression, which, in turn, formed a vicious cycle and promoted melanoma cell viability and metastatic potential [8]. Chang et al. discovered that primary PCa expressing ADAM9 protein enables the transformation of OBs into PCa-associated osteoblasts (PCa-OBs), inducing WISP-1 secretion from PCa-OBs in the bone microenvironment. The upregulation of WISP-1 in bone provided feedback to primary PCa, formed a vicious cycle and promoted PCa cell aggressiveness via epithelial-mesenchymal transition (EMT) activity [9]. Cancer cells often express high levels of CBS and (or) CSE to produce more endogenous hydrogen sulfide [10,11]. Therefore, whether there is a vicious cycle caused by S-sulfhydrylation or nitrification modification in the Sp1-CSE-H2S pathway of cancer cells, thus promoting the occurrence and development of cancer, will be a topic worthy of further investigation.
References
2. Luo C, Ji D, Li Y, Cao Y, Zhang S, Yan W, et al. Abnormal nitration and S-sulfhydration modification of Sp1-CSE-H2S pathway trap the progress of hyperhomocysteinemia into a vicious cycle. Free Radical Biology and Medicine. 2021 Feb 20;164:20-33.
3. Ji D, Luo C, Liu J, Cao Y, Wu J, Yan W, et al. Insufficient S-sulfhydration of methylenetetrahydrofolate reductase contributes to the progress of hyperhomocysteinemia. Antioxidants & Redox Signaling. 2022 Jan 1;36(1-3):1-4.
4. Jarosz AP, Wei W, Gauld JW, Auld J, Özcan F, Aslan M, et al. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is inactivated by S-sulfuration in vitro. Free Radical Biology and Medicine. 2015 Dec 1;89:512-21.
5. El-Khairy L, Ueland PM, Nygård O, Refsum H, Vollset SE. Lifestyle and cardiovascular disease risk factors as determinants of total cysteine in plasma: the Hordaland Homocysteine Study. The American Journal of Clinical Nutrition. 1999 Dec 1;70(6):1016-24.
6. Huang-Hobbs E, Cheng YT, Ko Y, Luna-Figueroa E, Lozzi B, Taylor KR, et al. Remote neuronal activity drives glioma progression through SEMA4F. Nature. 2023 Jun 28:1-7.
7. Kang L, Liu Y, He J, Wang Y, Xue M, Wu X, et al. GSK3β-driven SOX2 overexpression is a targetable vulnerability in esophageal squamous cell carcinoma. Oncogene. 2023 Jun42(30):2297-314.
8. Izraely S, Ben-Menachem S, Malka S, Sagi-Assif O, Bustos MA, Adir O, et al. The Vicious Cycle of Melanoma-Microglia Crosstalk: Inter-Melanoma Variations in the Brain-Metastasis-Promoting IL-6/JAK/STAT3 Signaling Pathway. Cells. 2023 May 30;12(11):1513.
9. Chang AC, Lin LW, Chen YC, Chen PC, Liu SC, Tai HC, et al. The ADAM9/WISP-1 axis cooperates with osteoblasts to stimulate primary prostate tumor growth and metastasis. International Journal of Biological Sciences. 2023;19(3):760-71.
10. Ascenção K, Lheimeur B, Szabo C. Regulation of CyR61 expression and release by 3-mercaptopyruvate sulfurtransferase in colon cancer cells. Redox Biology. 2022 Oct 1;56:102466.
11. Cao X, Ding L, Xie ZZ, Yang Y, Whiteman M, Moore PK, et al. A review of hydrogen sulfide synthesis, metabolism, and measurement: is modulation of hydrogen sulfide a novel therapeutic for cancer?. Antioxidants & Redox Signaling. 2019 Jul 1;31(1):1-38.