Abstract
Background: Dermatophytes are non-motile filamentous fungi prevalent in tropical regions and are primarily diagnosed based on clinical presentation. However, KOH mount and fungal culture detection improve diagnostic accuracy and guide further treatment.
Aim: To correlate the diagnostic performance of KOH with mycological culture in clinically suspected patients of tinea corporis.
Method: A cross-sectional study was conducted in the Department of Microbiology, Basic Medical Sciences Institute, Karachi, from June 2018 to June 2019. A total of 301 samples were collected using a non-probability convenience sampling technique after informed consent was obtained from all participants. Collected samples were examined with a KOH mount and fungal culture. Data were analyzed using the Statistical Package for Social Sciences (SPSS), version 21. Sensitivity, specificity, PPV, NPV, and diagnostic accuracy were calculated using culture as the gold standard.
Results: A total of 301 samples were analyzed to compare the performance of the KOH mount with fungal culture. Among these, 136 samples were culture-positive (84 dermatophytes, 52 non-dermatophytes), while 165 were culture-negative. KOH mount detected fungal elements in 208 samples and was negative in 93 samples. The KOH mount demonstrated a sensitivity of 76.5% and a specificity of 36.9%. The positive predictive value (PPV) was 50.0%, while the negative predictive value (NPV) was 65.6%. Overall, the accuracy of the KOH mount in detecting fungal elements was 55.5%.
Conclusion: KOH mount is a rapid and cost-effective screening tool, while culture remains essential for confirmation, species identification, and treatment guidance when results are uncertain.
Keywords
Tinea corporis, Dermatophytes, KOH mount, Fungal culture, Diagnostic accuracy
Introduction
Mycosis, or Fungal infection, is a disorder caused by fungi and is typically categorized into superficial, subcutaneous, or systemic mycoses, depending on the area of the body affected [1]. Most common among them are Superficial fungal infections (SFIs), which affect approximately 20 to 25 per cent of the world population at any given time [2]. Dermatophytes are a family of closely related fungi that are non-motile, filamentous organisms and can invade the keratinised tissue of humans, causing infections known as dermatophytosis, also referred to as ringworm [3–5]. The most common causative fungi belong to the genera Trichophyton, Epidermophyton, and Microsporum [6]. As they thrive in hot and humid conditions, dermatophyte infections are increasingly prevalent in many tropical and developing countries [7]. Risk factors for the rising incidence of dermatophytosis include overcrowding, shared living spaces, socioeconomic challenges, urbanization, large-scale international travel, immigration from tropical regions, and contact with animals, particularly pets. Moreover, the use of immunosuppressive drugs and advancing age are the predisposing factors for the rise of dermatophytosis-induced morbidity in humans [7,8].
One of the common superficial dermatophyte infections of the skin is tinea corporis, which typically affects the body's glabrous skin, excluding the hands, feet, scalp, bearded areas, face, groin, and nails [6,9]. This condition usually manifests as annular, scaly plaques with central clearing and an active, erythematous, sometimes raised border. The lifetime risk of developing tinea corporis is approximately 10–20% [9]. A cross-sectional study conducted in Visakhapatnam found that dermatophytosis significantly impairs individuals' psychosocial and financial well-being, especially those in the most productive age group, 30-45 years. Regardless of gender, number of lesions, or type of illness, the Dermatology Life Quality Index (DLQI) revealed that most patients experienced a "very large" or "extremely large" impact on their quality of life (QoL) [10]. Another study indicates that a multidisciplinary approach involving psychiatrists and psychologists, along with adequate counseling, leads to better therapeutic outcomes [11] Although the Diagnosis of superficial mycosis is often established clinically, laboratory confirmation is required for more difficult and atypical lesions to identify the causative fungus. The two primary laboratory diagnostic procedures in dermatological mycology are direct microscopy with potassium hydroxide (KOH) mount and culture. Potassium hydroxide (KOH) wet-mount preparation used for direct microscopy is generally considered a conventional rapid test [12]. KOH is significant because it is a simple, quick, inexpensive, and efficient screening method with a sensitivity of 55.9% to 80% and a specificity of 95% [13,14]. On the contrary, Fungal culture is the gold standard for diagnosing dermatophytosis, especially when the diagnosis is in doubt, inconclusive, or the infection is widespread, severe, or resistant to treatment [9].
Despite the frequent use of KOH mount in dermatological diagnosis, limited data from Pakistan exist on the comparative diagnostic accuracy between KOH and fungal culture. Our study aims to detect region-specific data, as well as the accuracy, sensitivity, and specificity of KOH mounts compared to fungal culture. In our setting, the concurrent use of microscopy and culture is not consistently practiced due to limited laboratory resources, lack of standardized protocols, and variations in diagnostic capacity. Consequently, there are insufficient data to demonstrate the combined diagnostic yield and the practical feasibility of implementing both techniques routinely in our country. Our study was therefore designed to provide local evidence to support and strengthen national diagnostic practices in alignment with international recommendations.
Material and Methods
Study design and setting
This cross-sectional study was carried out in the Department of Microbiology, Basic Medical Sciences Institute (BMSI), and Jinnah Postgraduate Medical Centre (JPMC) Karachi, in collaboration with the Department of Dermatology (JPMC) for the collection of study samples from June 2018 to June 2019. Through letter number F.2-81/2019-GENL/33048/JPMC, the committee of Jinnah Postgraduate Medical Centre (JPMC), Karachi, obtained ethical approval from the Institutional Review Board (IRB). We acquired informed consent from each individual. The information gathered was kept extremely confidential.
Sample size calculation
Samples were collected using a non-probability convenience sampling technique after obtaining informed consent from all participants. A total of 301 samples were collected in this study, which were clinically suspected of being patients with tinea corporis. Sample size was calculated by using Open-Source Epidemiologic Statistics for Public Health, version 3.01 (online) with an expected prevalence of 26.7%, confidence level of 95 % and a precision of ± 5%. The reference study used for this sample size is “Prevalence of tinea corporis and tinea cruris in an outpatient department of dermatology unit of a tertiary care hospital” [15].
Inclusion and exclusion criteria
Inclusion: All patients suspected of tinea corporis were included in the study, regardless of their age or gender.
Exclusion: Patients who refused to give their consent or who had been receiving active treatment with oral or topical antifungal medications for the previous two weeks.
Data collection procedure
A complete medical history was obtained for each patient. Thorough laboratory techniques and meticulous sample collection were used to diagnose and analyze tinea corporis. After cleaning the lesion sites with ethanol, specimens were gathered by scraping the edges of the lesions.
Laboratory identification
Koh mount microscopy: By degrading keratin, direct microscopic analysis with potassium hydroxide (KOH) exposed fungal structures. The test is performed by adding a drop of 10–20% KOH to the scrapings on a microscopic slide. The specimen is gently heated for five to ten seconds to hasten the degradation of the squamous cells. The epithelial tissue is dissolved by KOH, leaving behind easily visualized septate hyphae with or without arthroconidiospores. Dimethyl sulfoxide added to KOH may permit more rapid examination without heating [9].
Fungal culture
The specimens were cultivated on Dermatophyte Test Medium (DTM) and Sabouraud Dextrose Agar (SDA). Regardless of the KOH result, every sample was cultured. Each medium was inoculated and incubated in two vials at 25°C and 37°C. Vials were observed on a daily basis for the first ten days to see if there was any growth. After that, vials were checked periodically; if no growth was detected after six weeks, these vials were abandoned and labelled as growth negative. Sabouraud dextrose agar (SDA, 4% peptone, 1% glucose, agar, water), which serves as the agent utilized for the majority of morphologic descriptions, was incubated for six weeks. The addition of gentamicin, chloramphenicol, and cycloheximide to modified SDA makes it more selective for dermatophytes, as chloramphenicol promotes the growth of fungi by inhibiting bacterial contamination [16]. Dermatophyte test medium is a substitute for isolation media that contains phenol red, a pH indicator. Dermatophytes utilize the protein, which generates an alkaline environment and an excess of ammonium ions, causing the medium to turn from yellow to bright red [16]. The morphology, pigmentation, and microscopic characteristics of fungal development were examined using cellophane tape mounts and Lactophenol Cotton Blue (LPCB). Accurate species identification and long-term viability were guaranteed by urease tests and stock culture preservation using water culture and oil overlay procedures.
Statistical analysis
A structural proforma was used to collect all data. This database was created and analyzed with the Statistical Package for Social Sciences (SPSS, version 21). Frequencies and percentages were calculated using descriptive statistics, and the chi-square test was employed. The association between variables was analyzed using the chi-square test. P-values less than 0.05 were considered significant.
Result
This study involved 301 patients with tinea corporis. Among the 301 participants, 130 (43.2%) were female and 171 (56.8%) were male. Fungal growth was observed in 136 (45.1%) samples, while 165 (54.8%) isolates were deemed growth negative. Of the fungal positive results, 84 (28%) consisted of dermatophytes, whereas 52 (17.2%) included non-dermatophytes. A comparison of KOH microscopy and culture for Tinea corporis diagnosis revealed that 104 (63.1%) of KOH-positive cases were culture-negative, whereas 104 (76.5%) were culture-positive. Among KOH-negative cases, 32 (23.5%) were culture-positive, and 61 (36.9%) were culture-negative. KOH and culture outcomes demonstrated a statistically significant correlation (p=0.01). This distribution indicates that while KOH identified most culture-positive cases, it also produced a considerable number of false positives compared with culture results (Table 1).
|
KOH Result |
Culture Positive |
Culture Negative |
Total |
|
Positive |
104 (76.5%) |
104 (63.1%) |
208 (68.4%) |
|
Negative |
32 (23.5%) |
61 (36.9%) |
93 (31.6%) |
|
Total |
136 (44.7%) |
165 (55.3%) |
301 (100%) |
The KOH test has a sensitivity of 76.5% correctly identifying culture-positive cases (good sensitivity). However, it correctly rules out only 37% of culture-negative cases (low specificity). The PPV (50%) suggests that only half of KOH-positive cases are confirmed by culture. The NPV (65.6%) indicates that about two-thirds of KOH-negative cases are truly negative. This shows that KOH is a good screening test (it catches most true positives) but not a reliable confirmatory test (many false positives) (Table 2).
|
Parameter |
Value (%) |
95% Confidence Interval |
|
Sensitivity |
76.5 |
69.0–82.6 |
|
Specificity |
37.0 |
29.7–45.0 |
|
Positive Predictive Value (PPV) |
50.0 |
43.3–56.7 |
|
Negative Predictive Value (NPV) |
65.6 |
55.3–74.6 |
The observed agreement between KOH and culture is 54.8%, only slightly above what would be expected by chance (48.3%). The Cohen’s kappa value (0.13) indicates “slight agreement” according to Landis and Koch’s scale. Even though the agreement is statistically significant, KOH and culture results do not correlate strongly, meaning they often disagree. The overall interpretation of these tables is that the KOH test has moderate sensitivity but low specificity. There is only slight agreement between KOH and culture methods. While KOH can be used for rapid screening, culture remains the gold standard for confirming fungal infections (Table 3).
|
Statistic |
Value |
|
Observed agreement (Po) |
54.8% |
|
Expected agreement (Pe) |
48.3% |
|
Cohen’s kappa (κ) |
0.13 |
|
95% Confidence Interval (CI) |
0.02–0.24 |
|
Strength of agreement |
Slight agreement |
|
p<0.05 |
Statistically significant |
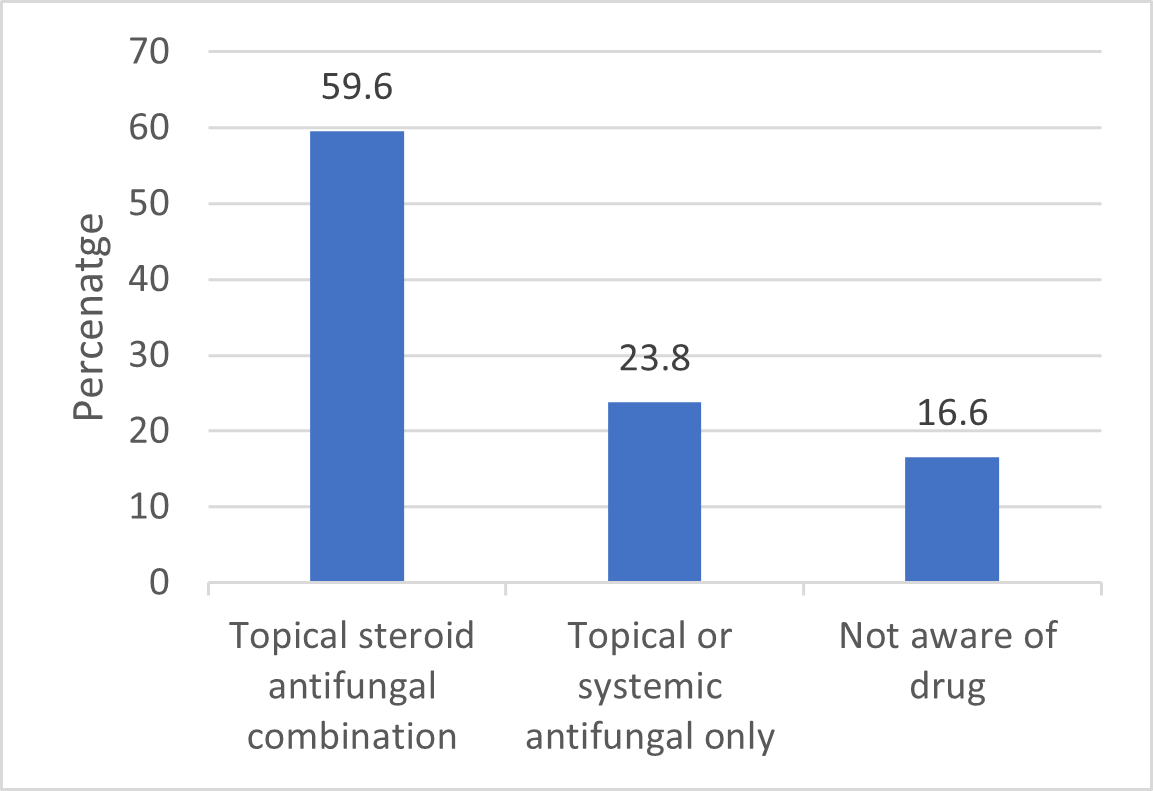
Our findings disclose that 168 (55.8%) of our study participants had previously used topical or oral medications before the two-week mark. The majority of patients, 100 (59.6%), were using a topical and steroid antifungal combination at the time; 40 (23.8%) were only receiving topical or systemic antifungals, and 28 (16.6%) of patients were unaware of the type of medication they were taking (Figure 1).
Figure 1. Distribution of drug history among the study population.
Discussion
Tinea corporis is one of the most common forms of dermatophytosis, accounting for approximately 22% of all cases. In Dermatology OPDs, the most prevalent superficial fungal infection is tinea corporis, which has the highest morbidity, recurrence, and antifungal resistance. An article revealed that in Pakistan, its prevalence ranges from 11.6% to 34.8%. However, it is increasing due to certain host and pathogen factors [17]. Although the disease rarely causes death, it is typically a refractory infection that impacts quality of life through social stigma and daily distress, making it a significant public health issue affecting all age groups [18]. The present study showed male predominance, 56.8% which was also in correspondence with that of Shakir et al. [19] (66.6%). Several studies showed that tinea corporis is more common in young male, the most productive age group, because they are more likely to have professions that involve a major portion of their job as outdoor work, thus resulting in excessive sweat production and increased exposure for transmission/reception of infection as well [20]. It is highly contagious and can spread through direct contact with an infected person, animals, and contaminated fomites [4]. Changes brought by excessive sweating in skin physiology make it more vulnerable to fungal infection [21]. In females, there are higher levels of androgenic hormones (progesterone and estradiol) that give protection to females by inhibiting fungal growth on the skin as compared to men, so this is one of the reasons for the decreased incidence of infection in females [22]. There is growing evidence of significant epidemiological shifts in tinea corporis worldwide in recent years, with some species having global prevalence and others regional distribution. Tinea corporis has become a major public health concern, especially in recent years, with a noticeable upward trend, particularly in recurrent and chronic infections [19]. Our analysis showed that 80.6% of samples were KOH positive, while 68.9% were culture positive (dermatophytes and non-dermatophytes). Additionally, our findings agree with those of Fatima et al. [23] and Kaur et al. [24]. Our data contradicts the 70.97% KOH-positive cases reported by Kaur et al. [25] and Chhabra et al. [26]. KOH versus fungal culture sensitivity has conflicting results. However, many studies have shown high culture positivity, indicating that culture is the superior method for identifying fungi from clinical samples. Our findings suggest that the KOH mount is a simple, quick, and essential tool for initial diagnosis, helping clinicians prompt treatment and potentially prevent the spread of dermatophytosis. In contrast, mycological culture is considered the gold standard for diagnosing fungal infections; however, it requires at least two to three weeks to yield results, making it less useful in urgent situations [23]. Fungal culture provides the definitive identification of fungal species. However, its routine application is deferred as it lacks the sensitivity (23% to 84.6%), prolonged turnaround time (TAT) (7–14 days or even up to 4 weeks), and lack of availability [9,12,14]. The widespread misuse of irrational combinations of topical steroid formulations in dermatophyte infections can lead to untoward adverse effects like tinea incognita (less scaling/less well-defined margins), atrophy, acneiform eruptions, etc., as well as chronicity [27]. Less scaling/well-defined margins make it harder to identify the optimal site for scraping. Multiple recent reviews and case series highlight how steroid-modified lesions are frequently misdiagnosed clinically and may show lower culture or delayed positive results; several clinicians advocate stopping steroids and repeating mycological tests after clinical unmasking, identifying the optimal site for scraping [28]. Use of a combination of antifungal and steroids may mask symptoms, suppress but not eradicate organisms, and produce false negatives or recurrent disease. Reviews warn that these combinations both complicate diagnosis and promote chronic/recurrent infections [29].
The Indian expert consensus suggests that the KOH mount is the point-of-care test for the rapid detection of dermatophytosis, and steroid use is strictly inhibited, as this would worsen the condition and make the diagnosis difficult [14]. Early diagnostic confirmation via KOH can help clinicians rule out the infection and avoid unnecessary drug exposure and resistance. This growing challenge also highlights the importance of early, accessible, and reliable diagnostic techniques, especially in resource-limited settings like Pakistan, to detect infection as soon as possible and reduce the burden of disease, misdiagnosis, and prevent misuse of topical creams.
Limitations
Our study had several limitations. Firstly, our study was conducted in a single tertiary care hospital, and the sample size is small, so it does not reflect broader regional trends. Secondly, we were unable to pursue this project to the molecular level.
Conclusion
This study concludes that fungal culture is also essential to rule out dermatophytosis, since KOH mount is a quick, inexpensive, and simple method for diagnosing tinea corporis, but alone is not sufficient. In routine diagnostic protocols, both methods (microscopy and macroscopic examination) are still used for detection. However, when hyphae or arthroconidia are observed in the lesions via microscopy, antifungal treatment can be initiated, but culture should still be performed to confirm the diagnosis. This approach can help reduce the disease burden and transmission through effective management.
Author Contributions
V.K., S.K., M.E., and K.K. conceptualized and designed the study. K.K., H.A., H.R.A., and M.E. acquired, analyzed, and interpreted the data. M.E., K.K., and H.R.A. prepared the initial draft of the manuscript. H.A., H.R.A., V.K., S.K., K.K., and M.E. critically reviewed the manuscript for important intellectual content. S.K. and V.K. supervised the study. All authors read and approved the final version of the manuscript for publication.
Ethics statement
Informed consent for treatment and open access publication was obtained or waived by all participants in this study. The Institutional Review Board of Jinnah Postgraduate Medical Centre issued approval through letter number F.2-81/2019-GENL/33048/JPMC.
Conflicts of interest
The author declares no conflict of interest.
References
2. Aboul-Ella H, Hamed R, Abo-Elyazeed H. Recent trends in rapid diagnostic techniques for dermatophytosis. Int J Vet Sci Med. 2020 Dec 17;8(1):115–23
3. Nida Imtiaz, Kausar Saeed, Adil Khan, Zakirullah, Hameed Ur Rehman, Umar Zeb, et al. To evaluate the fungal skin infestation in human population of central hospital, Rawalpindi. J Entomol Zool Stud 2016;4(6):706–708.
4. AL-Khikani FH. Dermatophytosis a worldwide contiguous fungal infection: Growing challenge and few solutions. Biomed Biotechnol Res J (BBRJ). 2020 Apr 1;4(2):117–22.
5. Rahman MA, Chowdhury OA, Debnath MR, Ahmed SM, Das S, Choudhury R, et al. Comparison among Different Culture Media for the Detection of Dermatophytes. Mymensingh Med J. 2018 Jul;27(3):626–30.
6. Yee G, Syed HA, Al Aboud AM. Tinea corporis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. Updated 2025 Feb 14.
7. Kruithoff C, Gamal A, McCormick TS, Ghannoum MA. Dermatophyte Infections Worldwide: Increase in Incidence and Associated Antifungal Resistance. Life (Basel). 2023 Dec 19;14(1):1.
8. Begum J, Mir NA, Lingaraju MC, Buyamayum B, Dev K. Recent advances in the diagnosis of dermatophytosis. J Basic Microbiol. 2020 Apr;60(4):293–303.
9. Leung AK, Lam JM, Leong KF, Hon KL. Tinea corporis: an updated review. Drugs Context. 2020 Jul 20;9:2020-5–6.
10. Karanam P, Veldi VDK, Metta AK, Angara SSP, Rani N, Sandhya P, et al. Psychosocial and Financial Impact of Dermatophytosis on Patients Attending a Tertiary Care Centre at Visakhapatnam: A Cross-Sectional Study. Cureus. 2025 Apr 18;17(4):e82483.
11. Das A, Sil A, Fatima F, Podder I, Jafferany M. Impact of chronic and recurrent dermatophytosis on quality of life and psychologic morbidity-a cross-sectional study. J Cosmet Dermatol. 2022 Aug;21(8):3586–92.
12. Afshar P, Larijani LV, Rouhanizadeh H. A comparison of conventional rapid methods in diagnosis of superficial and cutaneous mycoses based on KOH, Chicago sky blue 6B and calcofluor white stains. Iran J Microbiol. 2018 Dec;10(6):433–40.
13. South Carolina Blue Cross Blue Shield. Onychomycosis testing [Internet]. South Carolina Blue Cross Blue Shield; cited 2025 Nov 13. vailable from: https://www.southcarolinablues.com/web/public/brands/medicalpolicy/external-policies/onychomycosis-testing/.
14. Rajagopalan M, Inamadar A, Mittal A, Miskeen AK, Srinivas CR, Sardana K, et al. Expert Consensus on The Management of Dermatophytosis in India (ECTODERM India). BMC Dermatol. 2018 Jul 24;18(1):6.
15. Brigida S, Muthiah N. Pediatric Sedation: Prevalence of Tinea Corporis and Tinea Cruris in Outpatient Department of Dermatology Unit of a Tertiary Care Hospital. J of Pharmacol & Clin Res. 2017; 3(1): 555602.
16. Sahoo AK, Mahajan R. Management of tinea corporis, tinea cruris, and tinea pedis: A comprehensive review. Indian Dermatol Online J. 2016 Mar-Apr;7(2):77–86.
17. Suleri ZS. Recurrent tinea corporis and cruris: Antifungal resistance or poor compliance?. J Pak Assoc Dermatol. 2020 Aug 7;30(1):1–2.
18. Bitew A. Dermatophytosis: Prevalence of Dermatophytes and Non-Dermatophyte Fungi from Patients Attending Arsho Advanced Medical Laboratory, Addis Ababa, Ethiopia. Dermatol Res Pract. 2018 Oct 3;2018:8164757.
19. Shakir S, Saleem S, Rizvi W, Waheed A, Iqbal J. Isolation, identification and antifungal susceptibility of dermatophytes isolated from clinically suspected cases of tinea infections in Pakistan. Microbiol Res J Int. 2019;29(5):1–11.
20. Janagond AB, Rajendran T, Acharya S, Vithiya G, Ramesh A, Charles J. Spectrum of dermatophytes causing tinea corporis and possible risk factors in rural patients of Madurai region, South India. National Journal of Laboratory Medicine. 2016;5(4):MO29–MO32.
21. Sudha M, Ramani CP, Heber Anandan. Prevalence of dermatophytosis in patients in a tertiary care centre. International Journal of Contemporary Medical Research. 2016;3(8):2399–401.
22. Oktaviana N, Kawilarang AP, Damayanti P. Patient profile of tinea corporis in dr. soetomo general hospital Surabaya from 2014 to 2015. JBE. 2018;6(3):200–8.
23. Kashif S, Uddin F, Nasir F, Zafar S, Jabeen S, Kumar S. PREVALENCE OF DERMATOPHYTES IN SUPERFICIAL SKIN INFECTIONS IN A TERTIARY CARE HOSPITAL. J Pak Assoc Dermatol. 2021;31(3):484–8.
24. Ganjoo S, Sawhney M, Agarwal P. Clinico-epidemiologic profile of dermatophytosis in KOH positive patients in a tertiary care hospital in Gurugram, Haryana. Indian Journal of Health Sciences and Care. 2020;7:51–57.
25. Noronha TM, Tophakhane RS, Nadiger S. Clinico-microbiological study of dermatophytosis in a tertiary-care hospital in North Karnataka. Indian Dermatol Online J. 2016 Jul-Aug;7(4):264–71.
26. Chhabra N, Khare S, Das P, Wankhade AB. Clinicomycological Profile of Chronic Dermatophytosisin a Tertiary Care Centerfrom Raipur, Chhattisgarh. Indian Dermatol Online J. 2020 Sep 28;12(1):165–8.
27. A NP, Kaviarasan PK, K K, B P, C A. Clinicoepidemiological profile of steroid modified dermatophytosis following abuse of irrational combination of topical corticosteroid containing preparations (ICSP) [Internet]. IP Indian J Clin Exp Dermatol. 2020;6(4):318–24.
28. Zacharopoulou A, Tsiogka A, Tsimpidakis A, Lamia A, Koumaki D, Gregoriou S. Tinea Incognito: Challenges in Diagnosis and Management. J Clin Med. 2024 May 31;13(11):3267.
29. Chanyachailert P, Leeyaphan C, Bunyaratavej S. Cutaneous Fungal Infections Caused by Dermatophytes and Non-Dermatophytes: An Updated Comprehensive Review of Epidemiology, Clinical Presentations, and Diagnostic Testing. J Fungi (Basel). 2023 Jun 14;9(6):669.