Commentary
Exercise is increasingly being recognized as an important part of cancer treatment, as it has been shown to alleviate treatment- and disease-related side effects and improve quality of life of patients with cancer [1]. Furthermore, exercise has been linked to a reduced risk of cancer recurrence, particularly in cases of breast and colorectal cancer [2]. In recent years, the question has come to the forefront: “How?” does exercise influences cancer survival, and with it the interest in the underlying physiological mechanisms.
The tumor microenvironment (TME) and its individual composition can be involved in both metastasis formation and the response to treatment and survival of cancer cells, making it an extremely interesting target for therapeutic approaches [3]. Four pathways are currently known to influence the TME: The immune system, vascularization, cancer cell metabolism, and muscle-cancer cross talk [4,5]. All of these pathways can be influenced by exercise through myokines. In the following, we will focus on the last pathway — muscle-cancer cross talk.
Recently, a pilot study by Gunasekara et al. [6] demonstrated that an acute endurance training session appears to influence the growth of breast cancer cells. Serum collected before and after one hour of endurance exercise on a bicycle ergometer with an intensity of 50% VO2peak for 20 min and 60% VO2peak for the remaining 40 min was analyzed to assess its effects on breast cancer cell growth in vitro. Hormone-independent cells, specifically MDA-MB-231 cells (triple-negative breast cancer cells), were incubated with this serum, and cell vitality and proliferation were evaluated.
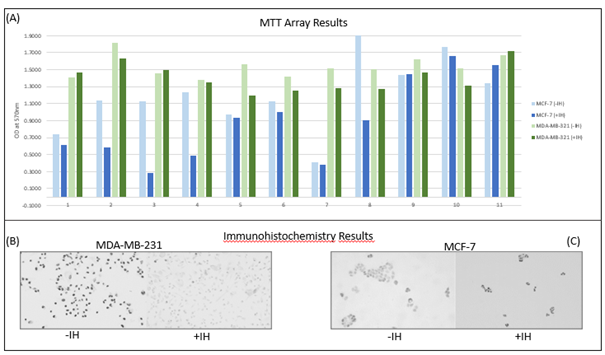
Additionally, the chemokine CXCL9 was identified as a promising key element in the pathway of muscle-cancer cross talk using cytokine arrays. The analyses showed that CXCL9 levels decreased following the acute endurance exercise compared to the resting control condition. To investigate whether the reduced cell activity was related to CXCL9 concentration, the CXCL9 signaling pathway was blocked using a CXCR3 antagonist, and cytotoxicity and proliferation assays were repeated with serum before the intervention and with serum plus the inhibitor. The results indicated that inhibition of CXCL9 led to growth-inhibitory effects (Figures 1A and 1B). These findings suggest that serum conditioned by an endurance intervention induces changes in cancer cell growth, possibly mediated through the CXCR3 axis [6].
The chemokine CXCL9 has the property of recruiting and activating immune cells, particularly CD8+ T cells, which play a crucial role in targeted attack and elimination of cancer cells [7]. To our knowledge, direct interactions with cancer cells have not yet been described.
This result highlights the importance of further research into the role of CXCL9 and other possible myokines in cancer biology. Therefore, in a further step, analyses were first conducted using a hormone-dependent breast cancer cell line (MCF-7) to examine whether other cancer cells respond differently to exercise-conditioned serum. The MCF-7 cells were also incubated with serum samples from all subjects under two experimental conditions: without addition (-IH) and with addition of the CXCR3 inhibitor (+IH). The MTT analysis showed a significant decrease in cell viability after CXCR3 inhibition (d= 0,75; p= .032) (Figure 1A), while immunohistochemical analysis revealed increased Ki-67 expression under the inhibitor condition (+IH) compared to the control condition (-IH) (Figure 1C).
These results suggest that although blockade of the CXCR3 signaling pathway influences metabolic processes, it does not cause clear antiproliferative effects in hormone receptor-positive breast cancer cells. Overall, the study underscores the complex role of the CXCR3 signaling pathway in the TME and provides key points for future research. The effects also appear to be cell type-specific, which should also be taken into account in future studies. Further, in the next step, analyses will be conducted to investigate whether acute strength training has similar effects on reducing cancer cell growth. This would support the hypothesis that exercise could influence the TME in a way that is beneficial for the patient. In future, more complex in vitro cancer cell models, which also include extra-cellular matrices and vessels, could be used to identify more complex influences of exercise on tumor growth.
This newly gained insight emphasizes the significance of exercise and its impact on the TME. Exercise could support the immune system's ability to target and eliminate tumor cells, especially in immunoreactive cancers such as triple-negative breast cancer by influencing the TME.
References
2. Cormie P, Zopf EM, Zhang X, Schmitz KH. The Impact of Exercise on Cancer Mortality, Recurrence, and Treatment-Related Adverse Effects. Epidemiol Rev. 2017 Jan 1;39(1):71–92.
3. Arneth B. Tumor Microenvironment. Medicina. 2019;56(1):15.
4. Spiliopoulou P, Gavriatopoulou M, Kastritis E, Dimopoulos MA, Terzis G. Exercise-Induced Changes in Tumor Growth via Tumor Immunity. Sports (Basel). 2021 Mar 30;9(4):46.
5. Pedersen L, Christensen JF, Hojman P. Effects of exercise on tumor physiology and metabolism. Cancer J. 2015 Mar-Apr;21(2):111–6.
6. Gunasekara N, Clauss D, Voss A, Schurz K, Fleck K, Neu-Gil P, et al. The Influence of an Acute Endurance Intervention on Breast Cancer Cell Growth-A Pilot Study. Int J Mol Sci. 2025 Apr 23;26(9):3976.
7. Pan M, Wei X, Xiang X, Liu Y, Zhou Q, Yang W. Targeting CXCL9/10/11-CXCR3 axis: an important component of tumor-promoting and antitumor immunity. Clin Transl Oncol. 2023 Aug;25(8):2306–20.