Abstract
Objective: To investigate if intraovarian autologous platelet-rich plasma (PRP) infusion improve ovarian reserve markers and fertility outcomes in women with diminished ovarian reserve (DOR).
Methods: A single-arm, prospective intervention study was conducted between July 2023 and April 2024. Women aged 21–40 years with DOR and previous poor response to maximal ovarian stimulation during IVF were recruited. Anti-mullerian hormone (AMH) and antral follicle count (AFC) were measured on menstrual cycle days 2–3, followed by PRP injection on days 5–15. AMH and AFC were reassessed 2–3 months later, and ovarian stimulation commenced 3–6 months post PRP. Primary outcomes were changes in ovarian reserve markers and fertility outcomes. Secondary outcomes were clinical pregnancy and live birth rates.
Results: Thirteen women underwent PRP injection (median age: 38.0 years; median body mass index: 22.2 kg/m2). Median AMH and AFC showed a numerical increase post-PRP (0.17 to 0.26 ng/ml and 5.0 to 6.0 respectively), though these changes did not reach statistical significance (p=0.695 and p=0.509). Median number oocytes retrieved increased (1.8 to 3.0 (p=0.099)). Median number of mature oocytes retrieved increased significantly (2.0 to 3.0; p=0.019) and embryos created (1.0 to 2.0; p=0.025). Clinical pregnancy rate was 41.7% and live birth rate was 33.3%. No complications were reported during PRP infusion.
Conclusion: Intraovarian PRP is a safe treatment option which may improve reproductive outcomes in DOR women. Larger, well-controlled studies to establish clinical applicability are warranted.
Keywords
Platelet-rich plasma (PRP), Autologous PRP, Diminished ovarian reserves, Poor ovarian response, In vitro fertilization (IVF), Assisted reproductive technology (ART)
Introduction
Diminished ovarian reserve (DOR), defined as the quantitative and potentially qualitative reduction of oocytes in reproductive age women [1], occurs in approximately 10% of the subfertile population. Premature ovarian insufficiency (POI), affecting 1% of women, is defined by the complete loss of ovarian activity (Follicle-stimulating hormone (FSH) >25IU/L and amenorrhea of >6 months) in women less than the age of 40 years [2]. It can exist as a spectrum and represents the most extreme clinical manifestation of accelerated ovarian follicular depletion. Multiple factors affect ovarian function [3]—advanced maternal age, genetics, iatrogenic (ovarian surgery) or environmental factors, with a decline in ovarian function reflected by reduction of ovarian reserve testing (Anti-Mullerian Hormone (AMH) and Antral Follicle Count (AFC) in serum and ultrasound scan respectively) [4].
There is no effective treatment to date to prevent or reverse a decrease in ovarian reserves. Current treatments are limited— maximum gonadotrophins usage in in vitro fertilization (IVF) cycles, supplements such as dehydroepiandrosterone (DHEA) and coenzyme Q10 [5], and experimental treatment such as in vitro activation [6]. Ultimately, the only proven reproductive treatment is oocyte donation [6] or adoption—options that may be fraught with ethical considerations.
Intraovarian autologous platelet-rich plasma (PRP) injection was recently introduced as a potential treatment with promising results [7–14]. PRP has a high concentration of platelets containing multiple vasoactive peptides and cytokines such as vascular endothelial growth factor, platelet-derived growth factor and Sphingosine-1-phosphate, many of which have important roles in ovarian function, follicular genesis and oocyte maturation [15]. Improvements in ovarian function (AMH, AFC, FSH), IVF outcomes and pregnancy rates have been reported. We report a pilot study to compare ovarian reserve markers and response to ovarian stimulation in IVF cycles before and after PRP injection in women with DOR/POI. Primary outcomes were changes in ovarian reserve markers (AMH and AFC) and fertility outcomes (number of oocytes collected and embryos created). Secondary outcomes were clinical pregnancy and live birth rates.
Materials and Methods
Study design and population
A single-arm, prospective intervention study was piloted at KK Women’s and Children’s Hospital, Singapore, between July 2023 and April 2024. Women aged between 21 to 40 years with significantly diminished ovarian reserves (AMH between 0.2-0.5ng/ml or AFC <5), and a history of poor ovarian response at prior IVF stimulation cycle despite maximum gonadotrophin dose (<4 oocytes retrieved with gonadotrophin doses of at least 300 IU per day) were recruited. Exclusion criteria included amenorrhea of >1 year, presence of medical conditions (diabetes mellitus, hypertension, anemia. collagen vascular / thyroid / thrombophilic disorders), known history of ovarian cysts or hydrosalpinges, heterologous sperm use and the inability to be monitored in our center for at least a year.
Study procedures
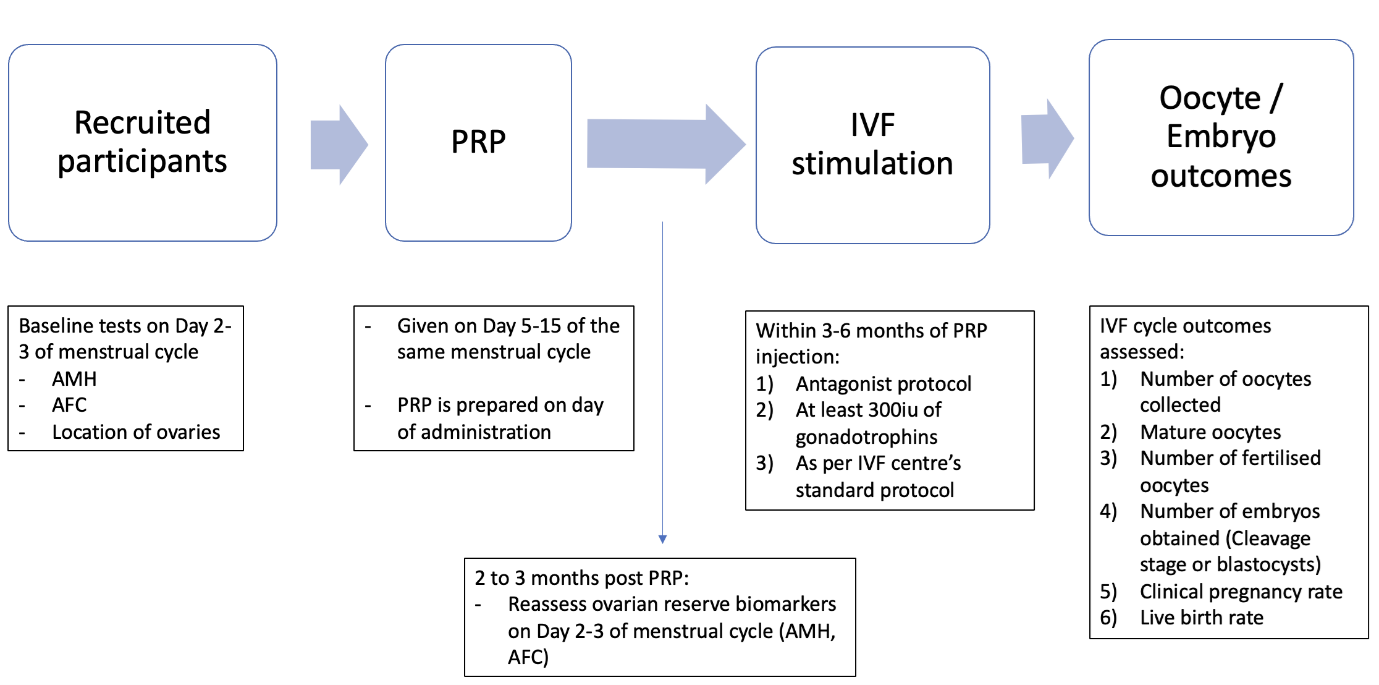
AMH and AFC were measured on menstrual cycle Day 2–3. Location and accessibility of ovaries were recorded. Subsequently, autologous PRP was injected transvaginally into the ovaries on day 5–15 of the same menstrual cycle. Patients returned 2 to 3 months post PRP injection for a repeat day 2–3 AMH and AFC measurements, and commenced ovarian stimulation 3 to 6 months from PRP injection (Figure 1).
Figure 1. Protocol on platelet-rich plasma (PRP) infusion. AMH: Anti-Mullerian Hormone; AFC: Antral Follicle Count; IVF: In vitro Fertilization.
PRP preparation and PRP injection
A one-step closed system using RegenKit®-THT kit (Regen Laboratory, Le Mont-sur-Lausanne, Switzerland) was used for PRP preparation according to manufacturer’s instructions. Peripheral blood (16 mls) from recruited women was collected via routine venepuncture an hour before the procedure into two RegenKit-THT-3 PRP tubes (8 ml per tube). These tubes were inverted 4–5 times to mix the anticoagulant with the blood and centrifuged at 1,500 RCF (or xg) for 9 minutes to obtain 4–5 ml of plasma per tube. The top 2–3 ml of plasma was removed before the tube was inverted in a see-saw motion 20–30 times to resuspend the platelets and leukocytes within the plasma. The PRP was drawn into a 5 cc luer lock syringe and connected to a 17G transvaginal needle. Platelet count in whole blood and PRP sample were evaluated for each patient.
PRP injection was performed as an outpatient procedure, under sedation in the operating theatre, within 30 minutes of preparation. PRP was injected intramedullary and subcapsularly on multiple sites in both ovaries using a 17-gauge single-lumen needle transvaginally under ultrasound guidance. Patients were monitored and discharged post procedure as per standard clinical practice.
Ovarian stimulation, oocyte retrieval (OR) and embryo transfer
All patients underwent controlled ovarian stimulation using a GnRH-antagonist protocol with gonadotrophin doses of at least 300 IU per day, 3 to 6 months post PRP injection. GnRH antagonist (Ganirelix 250mcg/day, Organon NV, The Netherlands) was initiated when there was a leading follicle of ≥12 mm. Final ovarian maturation was triggered using only recombinant HCG injection (OVIDREL®; Merck), or dual trigger (recombinant HCG combined with GnRH agonist (Triptorelin 0.2 mg; Ferring Pharmaceuticals)), and was administered when at least one follicle was ≥17 mm. Transvaginal OR was done 34–36 h post trigger injection. Intracytoplasmic sperm injection (ICSI) was performed in all cases—oocytes were denuded (hyaluronidase treatment) and injection was performed in all mature oocytes at MII stage. All embryos were monitored and a fresh embryo transfer (ET) of either cleavage stage embryo or blastocyst were performed. Embryologists were blinded to the patient’s PRP treatment status during oocyte and embryo assessment. In women undergoing frozen ET, endometrial preparation was done using oral estradiol valerate (Progynova; Bayer) tablets. A fixed dose (6 mg/day) was administered from cycle day 2. Embryo transfer was scheduled when the endometrial thickness was ≥7 mm. Luteal-phase support consisting of vaginal gel progesterone (Crinone 8%; Merck) was administered twice a day starting two days after OR (Day 2) where a fresh embryo-transfer was performed, and 2–5 days before a planned frozen ET, depending on the embryo stage.
Outcome measures
Primary outcomes were changes in ovarian reserve markers (AMH and AFC) measured on menstrual cycle days 2–3, number of oocytes collected during ovarian stimulation and embryos created before and after PRP injection. Serum AMH levels were measured using the chemiluminescent microparticle immunoassay (Roche Diagnostics GmbH, Mannheim, Germany) on a Roche Immunoanalyser (Roche Cobas e 411, Basel, Switzerland). AFC was measured between menstrual cycle days 2–3 transvaginally using the Samsung HS50 machine; ER4-9 probe, where antral follicles with a mean diameter of 2–9 mm were included.
Secondary outcomes were clinical pregnancy and live birth rates. Clinical pregnancy rate is defined as pregnancies with the presence of fetal heartbeat or gestational sac 4–6 weeks after ET procedure divided by number of embryo transfer procedures; and live birth rate is defined as pregnancies resulting in fetal delivery after 24 weeks of gestation divided by number of embryo transfer procedures.
Statistical analysis
Continuous variables were presented as medians (25th–75th percentiles) and categorical variables as counts (percentages). We assessed the normality of difference score distributions visually and using the Shapiro–Wilk test. Wilcoxon signed-rank test was used to analyze pre–post changes in ovarian reserve markers (AMH and AFC), the number of oocytes retrieved and embryos created. Stuart-Maxwell test with exact significance probability was used to assess marginal homogeneity of embryo transfer outcomes before and after PRP treatment. As an additional analysis, multivariable linear regression was performed to examine the association between the amount of platelet injected and relative changes in AMH and AFC, adjusting for potential covariates including age, BMI, and the duration between PRP injection and follow-up measurements. To explore time-dependent mechanisms of PRP action, we stratified the regression analysis by the median duration between PRP injection and follow-up measurements. No formal interaction testing was performed due to the small sample size. We reported results as β coefficients with corresponding 95% confidence intervals (CI). Statistical analyses were performed using Stata 16 (Stata Corporation).
Ethical approval
This pilot study was approved by the Centralised Singapore Institutional Review Board (Reference number: 2022-2222). The study was performed in accordance with the ethical standards as laid down in the 1975 Declaration of Helsinki and its later amendments or comparable ethical standards. Participation was voluntarily, and all recruited women received and signed a written approved consent form.
Results
Thirteen women with DOR underwent intraovarian PRP injection between July 2023 and April 2024. The median age was 38.0 (interquartile range 36.5–39.0) years old and the median BMI was 22.2 (21.5–26.3) kg/m2. These women had undergone between 1–5 prior IVF stimulation cycles—three women had cycle cancellation before oocyte collection due to poor follicular growth, while five had no oocytes collected in previous cycle(s). Four women never reached the embryo transfer stage due to either cycle cancellation and/or absence of oocytes. All women underwent only cleavage stage transfers previously. Two women had achieved pregnancies from previous cycles—one had a live birth and the other ended in miscarriage. Table 1 shows the baseline demographics and details of the IVF cycle post PRP.
|
Variables |
Values |
|
Baseline Characteristics (n=13) |
Median (25th–75th percentile) |
|
Age, years |
38.0 (36.5–39.0) |
|
Body mass index, kg/m2 |
22.2 (21.5–26.3) |
|
Number of IVF cycles done previously, n |
3 (1–3.5) |
|
Parity Nulliparous Multiparous |
n (%) 8 (61.5) 5 (38.5) |
|
IVF cycle details (n=12)a |
Median (25th–75th percentile) |
|
Stimulation duration, days |
10 (8–11) |
|
Total gonadotrophin dose, IU |
3600 (2700–4950) |
|
Gonadotrophin dose, IU/day |
450 (300–450) |
|
Total volume of PRP injected, ml |
4.0 (4.0–4.0) |
|
Platelet concentration in peripheral blood, x103 per µL |
269.50 (231.00–334.25) |
|
Platelet concentration in PRP, x103 per µL |
410.50 (303.50–457.50) |
|
Total amount of platelet injected, x109 |
1.76 (1.27–1.84) |
|
Type of trigger Ovidrel Dual trigger (Ovidrel + Triptorelin) |
n (%) 7 (58.3) 5 (41.7) |
|
IVF: In vitro Fertilization; PRP: Platelet-Rich Plasma aOne woman had a cycle cancellation due to no follicular growth. |
|
Median volume of PRP injected was 4.0 (4.0–4.0) ml. The median platelet concentration in whole blood was 269.50 (231.00–334.25) x103 per μL and in the PRP was 410.50 (303.50–457.50) x103 per μL, with an average of 1.76 (1.27–1.84) x109 platelets injected into the ovaries. Majority (84.6%; 11/13 women) had a platelet concentration of more than 1 x 109 injected. All women (n=13) underwent IVF stimulation, with a median duration of 123 (98–145) days post PRP injection. The median gonadotrophin dose was 450 (300–450) IU per day, with a median stimulation duration of 10 (8–11) days.
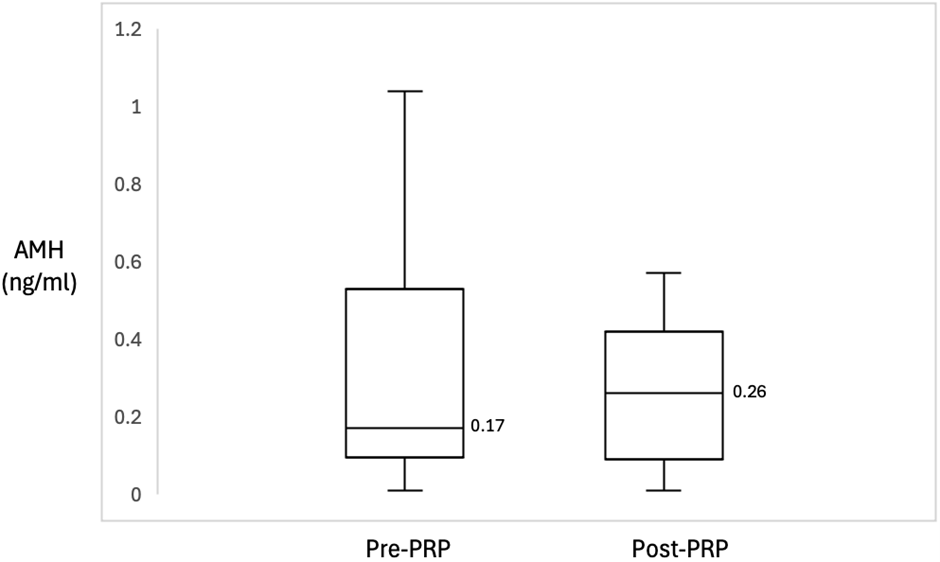
Median baseline AMH increased from 0.17 (0.10–0.53) ng/ml to 0.26 (0.09–0.42) ng/ml post PRP (p=0.695). AMH levels increased in five women (38.5%), decreased in seven women (53.8%) and remained unchanged in one woman (7.7%) (Figure 2).
Figure 2. Box plot of AMH before and after PRP treatment showed an increase of the median from 0.17 ng/ml to 0.26 ng/ml, p-value: 0.695. The boxes represent the interquartile range, while the whiskers represent the most extreme data points. Middle solid line represents data median. AMH: Anti-Mullerian Hormone; PRP: Platelet-Rich Plasma.
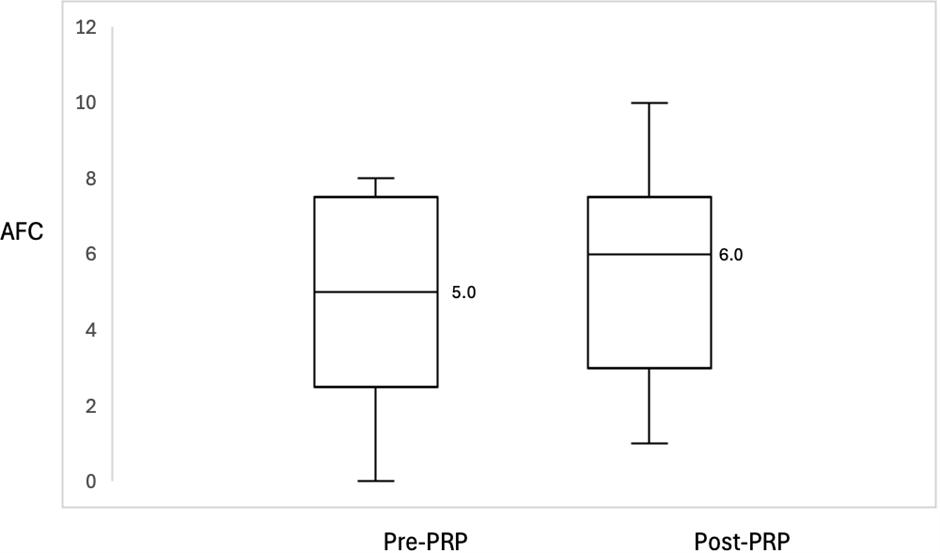
Median AFC also increased from 5.0 (2.5–7.5) to 6.0 (3.0–7.5) post PRP injection (p=0.509). AFC increased in six women (46.2%), decreased in three women (23.0%) and remain unchanged in four women (30.8%) (Figure 3).
Figure 3. Box plot of AFC before and after PRP treatment showed an increase of the median from 5.0 to 6.0, p-value: 0.509. The boxes represent the interquartile range, while the whiskers represent the most extreme data points. Middle solid line represents data median. AFC: Antral Follicle Count; PRP: Platelet-Rich Plasma.
The amount of PRP injected was not associated with relative changes in AMH (β 118.74; 95% CI -194.80, 432.29) and AFC (31.94; -92.47, 156.35) in the overall model. However, in the stratified analysis by median duration between PRP injection and follow-up measurements, a greater amount of PRP was associated with a higher relative increase in AFC (238.28%; 13.22, 463.34) among women with a longer duration between intervention and outcome assessment (Table 2).
|
|
Relative changes in AMH (%) |
Relative changes in AFC (%) |
||||
|
|
Model 1 (n=12) |
Model 2 (n=6) |
Model 3 (n=6) |
Model 1 (n=12) |
Model 2 (n=6) |
Model 3 (n=6) |
|
Duration between PRP injection and follow-up measurements (days) |
61–112 |
61–72 |
73–112 |
61–112 |
61–72 |
73–112 |
|
PRP injected (x109)
|
118.74 (-194.80, 432.29) |
-54.44 (-1177.35, 1068.484) |
183.892 (-1471.22, 1839.00) |
31.94 (-92.47, 156.35) |
-29.07 (-473.39, 415.25) |
238.28 (13.22, 463.34) |
|
Results are presented as β coefficients with 95% confidence intervals, derived from multivariable linear regression models. Model 1 represents the overall model, adjusted for age, body mass index, and the duration between PRP injection and follow-up measurements. Models 2 and 3 represent stratified models based on the median duration between treatment and follow-up, each adjusted for age and body mass index. PRP: Platelet-Rich Plasma; AMH: Anti-Mullerian Hormone; AFC: Antral Follicle Count |
||||||
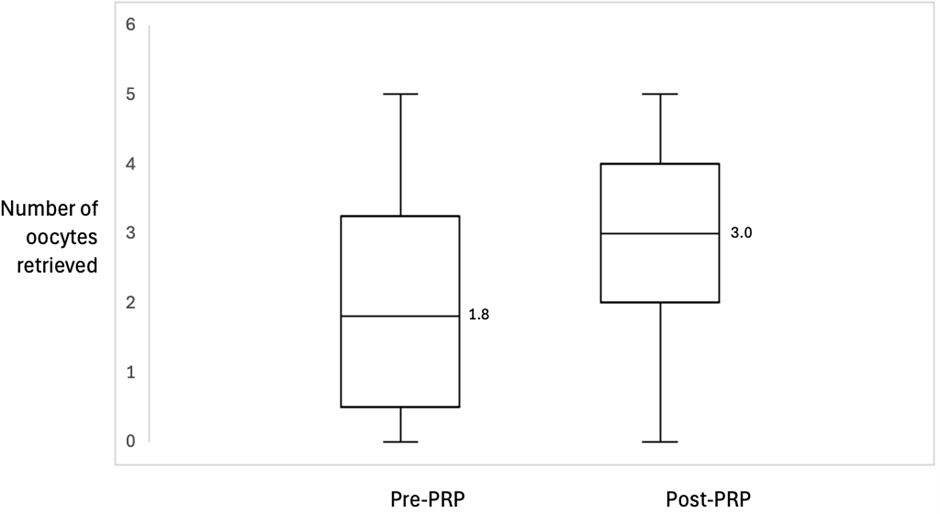
Twelve of 13 patients reached OR—one patient had cycle cancellation due to lack of follicular development. All patients had at least one oocyte retrieved, with nine women (69.2%) showing an improvement compared to prior cycles, culminating in an increase in the number of oocytes retrieved post-PRP treatment (1.8 (0.5–3.3) to 3.0 (2.0–4.0) (p=0.099) (Figure 4).
Figure 4. Box plot of number of oocytes retrieved before and after PRP treatment showed an increase in the median from 1.8 to 3.0, p-value: 0.099. The boxes represent the interquartile range, while the whiskers represent the most extreme data points. Middle solid line represents data median. PRP: Platelet-Rich Plasma.
The median number of mature oocytes collected increased significantly post-PRP (2.0 (1.4–2.8) to 3.0 (2.0–3.0) (p=0.019)). Median number of embryos created increased significantly as well (1.0 (0.3–1.8) to 2.0 (1.0–3.0) (p=0.025)) (Table 3).
|
|
Pre-PRP injection |
Post-PRP injection |
P values |
|
n=13 |
|
|
|
|
AMH, ng/ml |
0.17 (0.10–0.53) |
0.26 (0.09–0.42) |
0.695 |
|
AFC, n |
5.0 (2.5–7.5) |
6.0 (3.0–7.5) |
0.509 |
|
n=12a |
|
|
|
|
Retrieved oocytes, n |
1.8 (0.5–3.3) |
3.0 (2.0–4.0) |
0.099 |
|
Mature oocytes, n |
2.0 (1.4–2.8) |
3.0 (2.0–3.0) |
0.019* |
|
Embryos created, n |
1.0 (0.3–1.8) |
2.0 (1.0–3.0) |
0.025* |
|
Embryo transfer, n (%) No embryo transfer Cleavage staged Blastocyst |
2 (16.7) 10 (83.3) 0 |
0 11 (91.7) 1 (8.3) |
0.500 |
|
Notes: Values are presented in median (25th–75th percentile), unless otherwise stated. P values were derived from the Wilcoxon signed-rank test for continuous variables and the Stuart-Maxwell test for categorical variables. AMH: Anti-Mullerian Hormone; AFC: Antral Follicle Count; PRP: Platelet-Rich Plasma aOne women had a cycle cancellation due to no follicular growth. *Number of mature oocytes and embryos created were statistically significant. |
|||
Twelve women underwent ET—ten women (83.3%) had a fresh ET while two (16.7%) underwent frozen ET. One woman who underwent a fresh ET subsequently went on to have a frozen ET, resulting in a live birth. Cleavage stage ET were done for 11 women (91.6%), while 1 woman had a blastocyst transfer (8.4%). All mature oocytes were fertilized via ICSI. There was no significant change in stage of embryo transfer before and after PRP (p=0.500). Seven women (58.3%) had double embryo transfers, while five women (41.7%) had single embryo transfer. Clinical pregnancy rate was 41.7%, and the live birth rate was 33.3%. These were all singleton pregnancies delivered at full term. There was a first trimester miscarriage.
No adverse reactions were associated with intraovarian PRP injection (bleeding, injection, organ injury, allergic reaction).
Discussion
Our study demonstrated improvements in ovarian reserve markers (AMH and AFC) and IVF outcomes (number of oocytes retrieved, embryos created) after PRP injection.
Autologous PRP in recent years have been utilized in women with DOR, with improvements reported in ovarian reserve markers and IVF outcomes including oocyte and embryo yield post PRP [7–14]. Melo et al. [7], the first prospective controlled non-randomized comparative study published, reported significant improvement in AMH (0.62 ng/ml to 1.01 ng/ml, p<0.001) and AFC (4 to 7, p<0.001). Eighty-three women were recruited (Control (n=37) and PRP (n=46)). Clinical pregnancy rates were significantly higher in the PRP group (23.9% versus 5.4%, P=0.03), although differences in live birth rates were not seen. Proportion of good quality embryos generated post PRP treatment improved, suggesting PRP improving both oocyte number and quality. Platelet concentrations in the PRP injected were, however, not reported. Éliás et al. [8] reported a statistically significant improvement in the main fertility parameters and IVF outcomes—spontaneous pregnancy rate following PRP was 7% (n=1370, 95% CI: 0.04–0.12) and livebirth was 11% (n=1482, 95% CI: 0.07–0.15) in the largest meta-analysis of 2,256 women with DOR in 38 studies.
Several factors could affect success of PRP injection—preparation methods, volume and platelet concentration in the PRP and timing of IVF stimulation post PRP, with no standardization in published data. PRP concentrations vary widely from 2.5 to 8 times the concentration found in whole blood [16]. Sfakianoudis et al. [9] reported an ideal PRP platelet concentration of approximately 1,000,000 platelets per μL from 60 ml of peripheral blood, while Hosseinisadat et al. [17] reported a PRP platelet concentration of 1,000,000 platelets per μL from 20 ml of blood with no mention of total volume injected. Peripheral blood volumes required ranged from 8 ml [10] to 60–70 ml [11,12,18], with a median of 20 ml [13], which formed our basis of 16 ml. Our study measured amount and platelet concentration of PRP injected. We yielded a comparable platelet concentration per volume of blood drawn (0.38 million/μL from 16 ml of blood) as compared to what Sfakianoudis et al. reported (1 million/μL from 60 ml of blood) [9]. Volumes of injected PRP and preparation kits vary widely, from 1–8 ml injected per ovary [8]. Sills et al. [10] observed a positive correlation between platelet concentration in PRP injected and AMH measured post PRP in the minority of patients (51/182 women)—this was not observed in our study.
For our study, we chose a one-step closed system for efficiency and reproducibility reasons. Our method of preparation utilizes a separator gel instead of buffy coat system, similar to what has been published [9]. The separator gel has a specific gravity lower than red and white blood cells but more than the platelets [19], allowing a distinct separation of the plasma at the top, platelets and leukocytes resting on the gel and all other components at the bottom of the tube. While this may result in a significantly lower platelet concentration than the buffy coat method, it depletes more than 99.5% of red blood cells, preferentially depletes proinflammatory granulocytes, while recovering approximately 95% of platelets. In our study, there was 1.5 times increase in PRP platelet concentration as per manufacturer’s advice, with approximately 1.76 (1.27– 1.84) x 109 platelet injected in each patient. The cycle cancellation in our study was a 28 year old lady with secondary subfertility (2 pregnancy terminations). Her first IVF cycle was cancelled due to no follicular growth during ovarian stimulation, and second cycle yielded no oocytes at oocyte collection. She had less than 1 x 109 platelets injected. Post PRP, her AMH remained unchanged at 0.01 ng/ml, and AFC increased from 0 to 1, the lowest in our study.
Ovarian reserve testing after PRP have been reported to vary between 1–3 months —Sills et al. [10] reported the highest increase in AMH seen 4 weeks post PRP (28%; 51/182 women). Numerous studies have also reported varying time interval between PRP injection and IVF stimulation commencement, from the menstrual cycle immediately post PRP [14,20], and up to 3 months after [9]. Herlihy et al. [20] reported no improvement in pregnancy rates and oocytes collected with a time interval of 28 days from PRP to OR. Yu et al. [21] reported significant improvements where women underwent IVF stimulation 1–2 months post PRP, but not in those 3 months after, citing natural degradation of growth factors in PRP resulting in diminished regenerative effects. Barrenetxea et al. [14] reported on POR women undergoing three consecutive ovarian stimulation cycles, where either PRP or saline (sham procedure) was injected during the first OR. Number of mature oocytes increased in both groups with successive OR, where mechanical effect of OR may contribute to follicular growth through Hippo signaling disruption. Recruitment of non-growing follicles takes approximately 120 days to reach primary / secondary follicular development [22], and IVF stimulation was commenced 3–6 months post PRP in our study to evaluate the effects of PRP on the non-growing follicular pool.
Commonly used parameters to assess a woman’s fertility potential (AMH and AFC) have limitations in reliably predicting pregnancy outcomes following IVF [23]. In our study, although 6/13 women (46.2%) demonstrated an increase in AMH after PRP injection, majority showed improvements in number of oocytes collected (9/13 women; 69.2%) and embryos created (10/13 women; 76.9%). Of the women with clinical pregnancies, 60% (3/5 women) and 20% (1/5 women) had a drop in AMH and AFC respectively post PRP. PRP has a high concentration of platelets containing multiple vasoactive peptides and cytokines such as vascular endothelial growth factor, platelet-derived growth factor and Sphingosine-1-phosphate, many of which have important roles in ovarian function, follicular genesis and oocyte maturation [15]. Improvement in oocyte quality could explain the increase in embryos created and clinical pregnancies.
Improvements in ovarian reserve markers and fertility outcomes post PRP have been reported in DOR women, similar to our study. No correlation was found between amount of PRP injected and relative changes in AMH and AFC. Stratified analysis demonstrated an association between a greater amount of PRP with a higher relative increase in AFC among women with a longer interval between PRP injection and ovarian reserve marker assessment. This suggests that the interplay among PRP concentration, time interval between PRP of outcome assessment / IVF stimulation, is crucial in optimizing the efficacy of PRP.
Strengths of our study are that all women recruited are classified as POR in Groups 3 and 4 by the POSEIDON (Patient-Oriented Strategies Encompassing IndividualizeD Oocyte Number) classification system [24]. Platelet concentration before and after PRP were also measured for an objective evaluation. Platelets concentrations delivered were consistently above 1 x 109 in majority of our patients (84.6%; 11/13 women), although less than what was reported previously [9], may suggest that other factors could influence the effect of PRP.
Limitations of our study would be the small sample size and the lack of control group which restricts the interpretability of inferential analyses. There were also variations in stimulation protocol in the IVF cycles post PRP, although all women received at least 300 IU of gonadotrophins daily. Inter-cycle variability in measurements may also confound the study. Larger randomized controlled trials are required to further establish and confirm the potential benefits of intraovarian PRP in women with DOR.
Conclusion
In conclusion, this study suggests that intraovarian PRP injection may improve ovarian response and fertility outcomes in women with DOR with a history of poor ovarian response at prior IVF stimulation cycle despite maximum gonadotrophin dose. These women often have exhausted options, and PRP is a safe treatment option to offer, offering them an opportunity to achieve a genetically related offspring. Inter operator variations in preparation, timing of and volume of PRP injected however necessitate technique standardization and larger, well-controlled studies to establish its efficacy and clinical applicability before intraovarian PRP may be considered as an adjuvant for women with DOR.
Conflict of Interest
All authors have no affiliations or financial involvement with any commercial organizations with a direct financial interest in the subject or materials discussed in the manuscript. SLL, CWK and JKYC are supported by the National Medical Research Council, Ministry of Health, Singapore (SLL: NMRC/MOH-001537-01; CWK: NMRC/MOH-001773-00; JKYC: NMRC/CSA-SI-008-2016, MOH-001266-01, MOH-001221-01 and MOH-000932-01). JKYC received funding assistance from the Lien Foundation’s Healthy Early Life Moments in Singapore.
Funding Statement
This study received funding from the SingHealth Duke-NUS Academic Medicine (AM) Research Grant (AM/SU066/2022).
Acknowledgements
The authors acknowledge Ms Trish Koon and Dr Julin Wong for their support in patient recruitment during the study and would like to express our gratitude to the patients who participated in the trial.
Authors Contributions
Conceptualization: MJML, YF and JKYC; Methodology: MJML, YF, KYW, CWK, TXE, TYT, VVF, SL, SN, SFL and JKYC; Formal analysis and investigation: MJML, YPF, SLL and JKYC; Writing - original draft preparation: MJML; Writing - review and editing: MJML, YF, KYW, CWK, TXE, TYT, VVF, SL, SN, SFL and JKYC; Supervision: JKYC. All authors read and approved the manuscript.
Data Availability
The datasets generated and analyzed during this study are available from the corresponding author upon reasonable request.
Consent for Publication
The authors consent to the publication of this paper in the Journal of Clinical and Experimental Reproductive Medicine.
References
2. Kovanci E, Schutt AK. Premature ovarian failure: clinical presentation and treatment. Obstet Gynecol Clin North Am. 2015 Mar;42(1):153–61.
3. Rudnicka E, Kruszewska J, Klicka K, Kowalczyk J, Grymowicz M, Skórska J, et al. Premature ovarian insufficiency - aetiopathology, epidemiology, and diagnostic evaluation. Prz Menopauzalny. 2018 Sep;17(3):105–8.
4. Lee RWK, Khin LW, Hendricks MS, Tan HH, Nadarajah S, Tee NWS, et al. Ovarian biomarkers predict controlled ovarian stimulation for in vitro fertilisation treatment in Singapore. Singapore Med J. 2020 Sep;61(9):463–8.
5. Gat I, Blanco Mejia S, Balakier H, Librach CL, Claessens A, Ryan EA. The use of coenzyme Q10 and DHEA during IUI and IVF cycles in patients with decreased ovarian reserve. Gynecol Endocrinol. 2016 Jul;32(7):534–7.
6. Ferreri J, Fàbregues F, Calafell JM, Solernou R, Borrás A, Saco A, et al. Drug-free in-vitro activation of follicles and fresh tissue autotransplantation as a therapeutic option in patients with primary ovarian insufficiency. Reprod Biomed Online. 2020 Feb;40(2):254–60.
7. Melo P, Navarro C, Jones C, Coward K, Coleman L. The use of autologous platelet-rich plasma (PRP) versus no intervention in women with low ovarian reserve undergoing fertility treatment: a non-randomized interventional study. J Assist Reprod Genet. 2020 Apr;37(4):855–63.
8. Éliás M, Kónya M, Kekk Z, Turan C, das Virgens IPA, Tóth R, et al. Platelet-rich plasma (PRP) treatment of the ovaries significantly improves fertility parameters and reproductive outcomes in diminished ovarian reserve patients: a systematic review and meta-analysis. J Ovarian Res. 2024 May 17;17(1):104.
9. Sfakianoudis K, Simopoulou M, Grigoriadis S, Pantou A, Tsioulou P, Maziotis E, et al. Reactivating Ovarian Function through Autologous Platelet-Rich Plasma Intraovarian Infusion: Pilot Data on Premature Ovarian Insufficiency, Perimenopausal, Menopausal, and Poor Responder Women. J Clin Med. 2020 Jun 10;9(6):1809.
10. Sills ES, Petersen JL, Rickers NS, Wood SH, Li X. Regenerative effect of intraovarian injection of activated autologous platelet rich plasma: serum anti-mullerian hormone levels measured among poor-prognosis in vitro fertilization patients. International Journal of Regenerative Medicine. 2020 Mar 18;2020(1):1–5.
11. Stojkovska S, Dimitrov G, Stamenkovska N, Hadzi-Lega M, Petanovski Z. Live Birth Rates in Poor Responders' Group after Previous Treatment with Autologous Platelet-Rich Plasma and Low Dose Ovarian Stimulation Compared with Poor Responders Used Only Low Dose Ovarian Stimulation Before in Vitro Fertilization. Open Access Maced J Med Sci. 2019 Sep 14;7(19):3184–8.
12. Sfakianoudis K, Simopoulou M, Nitsos N, Rapani A, Pantou A, Vaxevanoglou T, et al. A Case Series on Platelet-Rich Plasma Revolutionary Management of Poor Responder Patients. Gynecol Obstet Invest. 2019;84(1):99–106.
13. Cakiroglu Y, Yuceturk A, Karaosmanoglu O, Kopuk SY, Korun ZEU, Herlihy N, et al. Ovarian reserve parameters and IVF outcomes in 510 women with poor ovarian response (POR) treated with intraovarian injection of autologous platelet rich plasma (PRP). Aging (Albany NY). 2022 Mar 22;14(6):2513–23.
14. Barrenetxea G, Celis R, Barrenetxea J, Martínez E, De Las Heras M, Gómez O, et al. Intraovarian platelet-rich plasma injection and IVF outcomes in patients with poor ovarian response: a double-blind randomized controlled trial. Hum Reprod. 2024 Apr 3;39(4):760–9.
15. Atkinson L, Martin F, Sturmey RG. Intraovarian injection of platelet-rich plasma in assisted reproduction: too much too soon? Hum Reprod. 2021 Jun 18;36(7):1737–50.
16. de Mos M, van der Windt AE, Jahr H, van Schie HT, Weinans H, Verhaar JA, et al. Can platelet-rich plasma enhance tendon repair? A cell culture study. Am J Sports Med. 2008 Jun;36(6):1171–8.
17. Hosseinisadat R, Farsi Nejad A, Mohammadi F. Intra-ovarian infusion of autologous platelet-rich plasma in women with poor ovarian reserve: A before and after study. Eur J Obstet Gynecol Reprod Biol. 2023 Jan;280:60–3.
18. Pantos K, Simopoulou M, Pantou A, Rapani A, Tsioulou P, Nitsos N, et al. A Case Series on Natural Conceptions Resulting in Ongoing Pregnancies in Menopausal and Prematurely Menopausal Women Following Platelet-Rich Plasma Treatment. Cell Transplant. 2019 Sep-Oct;28(9-10):1333–40.
19. Dashore S, Chouhan K, Nanda S, Sharma A. Platelet-Rich Fibrin, Preparation and Use in Dermatology. Indian Dermatol Online J. 2021 Nov 25;12(Suppl 1):S55–S65.
20. Herlihy NS, Cakiroglu Y, Whitehead C, Reig A, Tiras B, Scott RT Jr, et al. Effect of intraovarian platelet-rich plasma injection on IVF outcomes in women with poor ovarian response: the PROVA randomized controlled trial. Hum Reprod. 2024 May 9:deae093.
21. Yu TN, Chen MJ, Lee TH, Chen YC, Cheng EH, Huang CC, et al. Intraovarian platelet-rich plasma injection significantly improves blastocyst yield and quality in IVF patients. Sci Rep. 2025 Jan 8;15(1):1301.
22. McGee EA, Hsueh AJ. Initial and cyclic recruitment of ovarian follicles. Endocr Rev. 2000 Apr;21(2):200–14.
23. Barrenetxea G, Martínez E, De las Heras M, Arambarri JI, Axpe M, Barrenetxea J. Lack of predictive value of ovarian reserve tests for pregnancy likelihood. The huge difference between quantity and quality. AJBSR. 2019;3:133–41.
24. Alviggi C, Andersen CY, Buehler K, Conforti A, De Placido G, Esteves SC, et al. A new more detailed stratification of low responders to ovarian stimulation: from a poor ovarian response to a low prognosis concept. Fertil Steril. 2016 Jun;105(6):1452–3.