Abstract
Introduction: Neurodevelopmental disorders (NDDs) such as autism spectrum disorder (ASD) and attention-deficit hyperactivity disorder (ADHD) are increasingly prevalent in children globally, including Cameroon. Emerging evidence suggests that early-life exposure to environmental neurotoxins, particularly mycotoxins, may disrupt brain development and contribute to these disorders.
Objective: This review aimed to assess the potential impact of dietary exposure to neurotoxic mycotoxins, particularly aflatoxin B1, fumonisin B1, and ochratoxin A, on brain development and the risk of NDDs in children in Cameroon.
Methods: A comprehensive review of peer-reviewed studies, national reports, and biomonitoring data from 1993 to 2024 was conducted. Literature on food contamination, mycotoxin toxicology, exposure levels, and neurodevelopmental outcomes in both humans and animal models was synthesized.
Results and Discussion: Children in Cameroon are exposed to mycotoxins through contaminated staple foods such as maize and groundnuts, with exposures often exceeding international safety limits. These toxins can cross the blood-brain barrier and placenta, disrupt key neurodevelopmental processes (e.g., neuroplasticity, neurogenesis) through various mechanisms such as oxidative stress, neuroinflammation, mitochondrial dysfunction, and result in behavioral and cognitive impairments. Animal studies show strong links between chronic mycotoxin exposure and NDD-like symptoms. In Cameroon, these risks are intensified by poor food storage, weak regulations, and low dietary diversity.
Conclusion: Dietary mycotoxin exposure in children is a serious but underrecognized contributor to adverse neurodevelopmental outcomes in Cameroon. There is an urgent need for national surveillance, child-specific risk assessments, stronger food safety enforcement, and public health education to mitigate exposure and protect vulnerable populations.
Keywords
Neurotoxic mycotoxins, brain development, neurodevelopmental disorders, dietary exposure, children, Cameroon
Introduction
Neurodevelopmental disorders (NDDs) are a group of brain disorders affecting proper brain growth and function, characterized by developmental deficits in cognition, language, behavior, and/or motor skills that cause impairment of personal, social, academic, and/or occupational functioning [1]. The neurodevelopmental disorders in children are the most prevalent disabilities encountered in pediatric health services, which include autism spectrum disorder (ASD), attention deficit hyperactivity disorder (ADHD), intellectual disability (mental retardation), cerebral palsy (CP), learning disabilities, neurodevelopmental motor disorders, conduct disorders, impairment in vision and hearing [2,3,4], with ASD and ADHD the most common of the NDDs. These groups of disorders generally affect children in their early life when the brain is still developing and can persist in adult life. The common symptoms of NDDs, which are generally observed during a child’s preschool years (24–36 months), are problems with language and speech, difficulties with learning, social skills deficits, abnormal behaviors, and impaired motor skills. The diagnosis of NDDs in children is done through various tests, including neurologic examination, developmental test, brain imaging, laboratory test, and physical examination [4].
The development of the brain begins a few days after conception and continues until adulthood, with a critical period from birth to early life (0–5 years old). During this period, all the processes necessary for brain growth and function, such as neurogenesis, growth of axons and dendrites, synaptogenesis, gliogenesis, and others, are taking place [4]. Disruption of brain development by environmental factors such as stress, socioeconomic status, and diets contaminated with neurotoxins can affect the complete process, resulting in NNDs. However, the etiology of NDDs has been extensively based on genetic factors. Environmental factors have been identified as the major factors that can increase the risk of NDDs, in some cases by interacting with genetic factors. Children's brains are more vulnerable to environmental toxicants due to their limited ability to metabolize and detoxify toxicants [5]. Among the environmental toxicants with potential effects on brain development and neurodevelopmental outcomes in children, mycotoxins are inevitable due to their frequent occurrence on food commodities consumed by children. The increase in the prevalence of NDDs in children, such as ASD and ADHD, in developing countries has gained much attention on the possible role of mycotoxins in brain development and developmental disorders.
Mycotoxins are secondary metabolites from fungi, which pose threats to animal and human health, especially to vulnerable populations such as children. Mycotoxins generally infiltrate the crops and food commodities during storage, transportation, pre-and post-harvest conditions, often occurring in developing countries due to weather, poor management, and inadequate methods of cultivation. Among the mycotoxins, aflatoxin, fumonisin, and Ochratoxin. Zearalenone and deoxynivalenol are the common mycotoxins found in cereals and cereal-based products. According to statistics, 25% of the world’s agricultural products are contaminated with mycotoxins, which implies that most of the foods usually consumed by humans are contaminated, thus raising food safety concerns [6]. Recent studies have shown that mycotoxin exposure can result in acute or chronic neurotoxicity and long-term neurodevelopmental disorders [6–8].
Children in Cameroon face heightened vulnerability to neurotoxic mycotoxins due to a combination of high consumption of contaminated staple foods such as maize and groundnuts [9–11], poor post-harvest handling and storage practices [12], limited regulatory enforcement of food safety standards [13], and low dietary diversity among rural and low-income populations [14]. Moreover, the frequent use of these mycotoxin-prone crops in complementary feeding for infants and young children significantly increases the risk of early-life exposure during critical windows of immunity (and potentially brain) development [15–17]. The situation is further exacerbated by limited public awareness and inadequate national monitoring systems, making it difficult to identify and mitigate exposure risks in vulnerable child populations [18–20].
The primary objective of this study is to critically examine the potential impact of dietary exposure to neurotoxic mycotoxins on brain development and the risk of neurodevelopmental disorders in children in Cameroon. This involved synthesizing available toxicological, epidemiological, and food contamination evidence to highlight early-life vulnerabilities, assess exposure sources, and support the need for child-specific dietary risk assessment and public health interventions.
Methodology
This review employed a systematic narrative approach to synthesize current evidence on the impact of dietary exposure to neurotoxic mycotoxins on brain development and neurodevelopmental disorders (NDDs) in children, with a specific focus on Cameroon. Relevant literature was identified through comprehensive searches of peer-reviewed scientific databases, including PubMed, Scopus, ScienceDirect, and Google Scholar. The review did not incorporate gray literature or national reports. Keywords and Boolean combinations used included: mycotoxins, aflatoxin, fumonisin, ochratoxin A, neurotoxicity, brain development, neurodevelopmental disorders, children, Cameroon, dietary exposure, food contamination, and risk assessment. The search covered studies published between 1990 and 2025. There were no stringent quality standards for article inclusion in the review.
Inclusion criteria consisted of (1) studies reporting on the occurrence of mycotoxins in food or biological samples in Cameroon, (2) experimental studies detailing neurotoxic mechanisms of mycotoxins, (3) biomonitoring or dietary exposure assessments relevant to children or maternal populations, and (4) articles addressing links between mycotoxin exposure and neurodevelopmental or cognitive outcomes. Exclusion criteria included non-English articles, studies not involving mycotoxins or children, and reports lacking sufficient methodological transparency.
The screening process was done manually. Selected literature was reviewed and categorized into key thematic areas: (1) types and occurrence of neurotoxic mycotoxins in Cameroon, (2) molecular and cellular mechanisms of neurotoxicity, (3) exposure pathways and early-life vulnerability, (4) experimental models of neurodevelopmental effects, and (5) dietary risk assessments and public health implications. Quality assessment of studies was based on sample size, relevance, analytical methods, and risk of bias.
Results and Discussion
Neurotoxic mycotoxins and mechanisms of brain toxicity
Many studies have reported that the neurotoxicity of mycotoxins such as aflatoxins, fumonisins, and Ochratoxin A, are amongst the most potent mycotoxins commonly found in staple foods dedicated to children. Aflatoxins, particularly aflatoxin B1, are the most harmful to humans and are generally found in maize, peanuts, rice, and nuts, while ochratoxin A is found in cereals and fruits, and fumonisin B1 is found in cereals and rice [21]. A few studies from Cameroon by Ediage et al. [14], and Abia et al. [11,18,22,23] have revealed strong co-occurrence of aflatoxins and fumonisins in food and biological samples, occasionally at levels exceeding legislative limits and resulting in unacceptable exposure levels among various consumer groups. These mycotoxins have a strong affinity with the brain and can reach the systemic circulation through consumption of contaminated foods [24]. Mycotoxins induce neurotoxicity through various cellular and molecular mechanisms, including disruption of blood-brain barrier, oxidative stress, mitochondrial dysfunction, neuroinflammation, interference with the neurotransmitter system, leading to disruption of neuronal growth and synaptic function [7,25]. Ingestion of various neurotoxic mycotoxins may induce or serve as a risk factor for various toxicities in the brain by different mechanisms (Figure 1).
Figure 1. Different mechanisms of neurotoxic mycotoxins induce toxicity in a developing brain.
Oxidative stress occurs when there is an imbalance between antioxidants and pro-oxidants, often resulting in the overproduction of reactive oxygen species (ROS). The brain is the most susceptible organ to oxidative stress because of its higher consumption of energy and oxygen, and its higher production of fatty acid peroxide [26]. Mycotoxins have been reported to alter the antioxidant systems, including glutathione transferase (GST), glutathione peroxidase (GPx), catalase (CAT), superoxide dismutase (SOD), and glutathione reductase (GR); stimulate the production of reactive oxygen species (ROS) and reactive nitrogen species (RNS), leading to mitochondrial dysfunction with decrease of mitochondrial membrane potential (MMP), an increase of the ration pro-apoptotic protein Bax/anti-apoptotic protein Bcl-2, ATP (Adenosine triphosphate) depletion [24]. The presence of ROS/RNS can activate the signaling pathways, including p53, mitogen-activated protein kinases (MAPK), Janus kinase-signal transducer and activator of transcription (JAK-STAT), and AKT pathways, leading to apoptosis [24]. Apoptosis is often a result of oxidative stress with activation of caspase-3 [27]. Some studies demonstrated that AFB1, FB1, and OTA reduced brain antioxidant levels, increased the malondialdehyde (MDA) levels, induced the production of ROS and RNS, and up regulated some signaling pathways implicated in oxidative stress [6,8,26,28].
Mitochondria play an important role in the generation of metabolic energy in form of ATP necessary for the cell survival and proper functioning, disruption of mitochondrial function in the brain result in the reduction of ATP levels, impairment of all energy-dependent cellular functions, disturbance of the generation of biosynthetic precursors leading to neurological and neurodevelopmental disorders [29]. Exposure to mycotoxins (AFB1, FB1, and OTA) may induce or serve as a risk factor for the dysfunction of mitochondrial by targeting some enzymes and complexes within the mitochondria and regulating mitochondrial respiratory chain proteins. Mitochondrial dysfunction can also be a result of oxidative stress occurring in neurons, microglia, and astrocytes [6,26,28,30].
Neuroinflammation plays an important role in the outcome of neurodevelopmental disorders due to the activation of the immune system [31]. The presence of mycotoxins in the brain can induce or serve as risk factor for the activation of the immune cells (microglial, astrocytes and endothelial cells) through the modulation of toll like receptors (TLRs), and subsequently the activation of different signaling pathways with release of inflammatory cytokines (Tumor necrosis factor-α (TNF-α), Interleukin-6 (Il-6), Interleukin-1β (Il-1β)), nitric oxide synthase (iNOS) and chemokines. This inflammatory process exacerbated neuronal damage and chronic neuroinflammation, with immune cells generally recognizing mycotoxins as a danger signal [24,32]. Some studies have shown that AFB1 induces neuroinflammatory responses in the brain by activating various signaling pathways, such as Nucleotide-binding oligomerization domain (NOD)-like-receptor (NLR) family pyrin domain containing 3 (NLRP3), TLR2/4, and nuclear factor kappa-light chain-enhancer of activated B cells (NF-κB), with production of proinflammatory cytokines and chemokines [8,33]. Additionally, OTA also induces the release of proinflammatory cytokines TNF-α and IL-6 [34].
Neurotransmitter systems, including cholinergic, dopaminergic, glutamatergic, serotonergic GABAergic, and neurotransmitters such as acetylcholine, dopamine, GABA, serotonin, and norepinephrine, play a particular role in the central nervous system (CNS), as they are essential for behavioral functions in the body. Alteration of a neurotransmitter system, including neurotransmitters as well as enzymes, results in cognitive and behavioral changes [35]. Mycotoxins are capable of modifying the metabolism of some neurotransmitters (acetylcholine, serotonin, and dopamine) and modulating the cholinergic, serotonergic, and dopaminergic transmission, leading to an unbalance of neurotransmitters in the brain, affecting communication between neurons [36]. AFB1 exposure resulted in the upregulation of acetylcholinesterase, butyrylcholinesterase, as well as adenosine deaminase, leading to alterations in memory and cognitive function [37].
Blood-brain barrier (BBB) is a semipermeable membrane barrier situated between the bloodstream and brain, with the role to regulate the influx and efflux of exogenous compounds and xenobiotics into the brain, or biological substances needed for brain metabolic activity and neuronal function [38]. Some substances, due to their polarity and size, can easily cross the blood-brain barrier and induce neurotoxicity. Due to their ability to easily cross blood-brain barrier, mycotoxins have been classified as a potential neurotoxin and harmful to the neurological system. They compromised directly or indirectly the BBB, leading to the other neurotoxicity mechanisms such as oxidative stress, mitochondrial dysfunction and neuroinflammation [6]. AFB1, FB1, and OTA are fat-soluble small molecules that can easily cross the BBB, enter the nerve cells via transporters, and accumulate in different brain regions [8,39]. In the brain, they can bind to the cell membrane, causing lipid peroxidation, or bind to the receptors with activation of the cascade of events and signaling pathways. Additionally, AFB1 can reduce the levels of L-proline, a neuroprotective metabolite crucial for maintaining the integrity of the BBB [40].
Mycotoxin story in Cameroon: occurrences and exposures
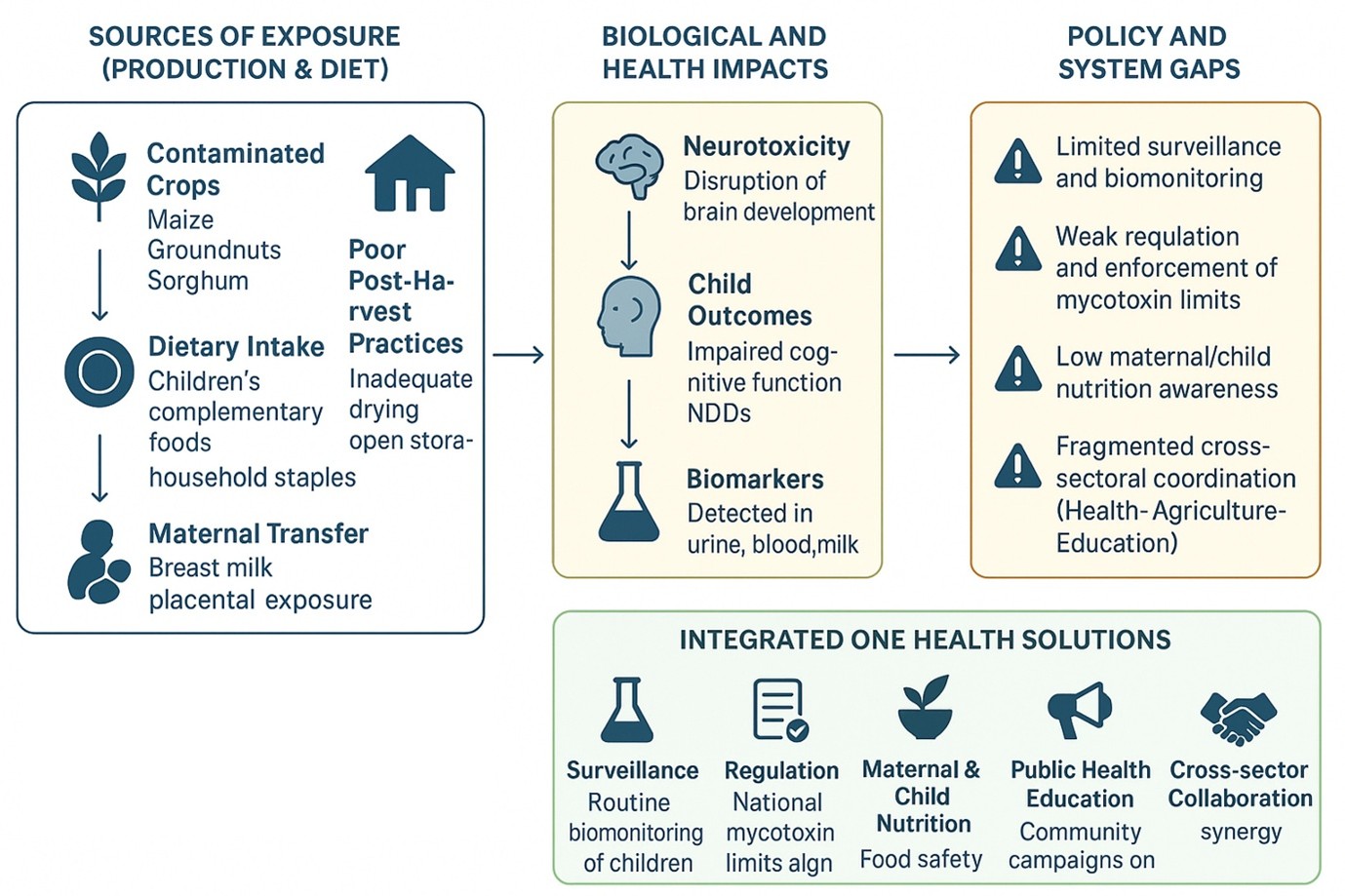
Mycotoxin, especially aflatoxins, contamination in staple foods such as maize and groundnuts poses a major public health concern in Cameroon [18]. These crops, widely consumed across all age groups, are highly susceptible to fungal infections by Aspergillus and Fusarium species, which produce aflatoxins and fumonisins, respectively. Several studies have documented widespread contamination in both raw and processed products in Cameroon, and this has been well reviewed with comprehensive tables of occurrences between 1993 to 2024 [41,42]. Processed foods derived from these staples, such as maize flour, pap (a fermented porridge), and ‘mboh’/’nnam owondo’ (steamed groundnut pudding), often retain high mycotoxin levels due to insufficient processing controls [43]. Inadequate drying, poor storage conditions, and humid climates further promote fungal growth and toxin production. These challenges are compounded by limited regulation, weak enforcement, and a lack of awareness among producers and consumers [18]. Mycotoxin contamination in staple foods represents a chronic exposure risk, especially for children and rural households who consume these foods daily. Addressing this issue requires a combination of good agricultural practices, improved post-harvest handling, and national-level surveillance programs to monitor and reduce contamination levels.
Mycotoxin dietary exposure in Cameroon is a serious and under-recognized public health issue, particularly among vulnerable populations such as children, pregnant women, and subsistence farmers. The widespread contamination of commonly consumed foods, particularly maize and groundnuts, means that chronic ingestion of aflatoxins and fumonisins is likely in both rural and urban settings, requiring biomonitoring studies. Urinary mycotoxin exposure studies from Cameroon have been reviewed by Monono et al. [41] except for the recent studies by Abia et al [44] that detected aflatoxin M1 in 42% of adult urine samples in a range of not detected-0.21 μg/L. Additionally, the estimated daily intakes for at least one individual exceeded the tolerable daily intakes for fumonisin B1, deoxynivalenol, and zearalenone, excluding aflatoxins for which no TDI is set. Associated health effects have been indicated by Nguegwouo et al. [42], including carcinogenic, immunosuppressive, genotoxic, and neurotoxic.
Based on the existing findings, mycotoxin exposures in Cameroon are a public health risk. Notwithstanding, Cameroon lacks a coordinated national surveillance system to routinely assess dietary intake and contamination levels. Most studies remain localized, offering only snapshots of the broader exposure landscape. A systematic approach that combines dietary surveys, food sampling, and health monitoring is urgently needed to guide public health policy. Strengthening food safety regulation, public awareness, and agricultural practices can significantly reduce mycotoxin exposure and its long-term health consequences [18].
Early-life susceptibility and dietary risk
Children and infants are very susceptible to the adverse health effects from exposure to environmental toxicants. The postnatal period is a critical period for the maturation of the immune and central nervous systems because at birth, these systems are still immature [45]. Exposure to toxins during the critical period can interfere with the brain development process, leading to cognitive deficits and other behavioral changes [6]. For the maturation of organs and systems, infants and children's bodies need energy, water, and oxygen, resulting in high food intake. Dietary mycotoxin exposure can be easily distributed within infants and children and can reach the CNS as the BBB is not fully developed. Also, due to the immaturity detoxification systems (enzymes), the presence of mycotoxins in their body will take time to be detoxified, and elimination of mycotoxins may be decreased as their glomerular filtration rate is less than 40% compared to adults and the function of the renal tubules is less mature [45,46].
A high food intake relative to body weight increases dietary exposure to contaminants, especially when the amount of food consumed is disproportionately high compared to body weight. In Cameroon, high food intake relative to body weight significantly elevates dietary exposure to mycotoxins, particularly among infants and young children. Due to rapid growth and higher metabolic demands, children consume more food per kilogram of body weight than adults, making them especially vulnerable to contaminants such as aflatoxins, fumonisins, and ochratoxins commonly found in staple crops like maize and groundnuts [14,17]. In some communities in Cameroon, where food diversity is limited and diets rely heavily on cereals susceptible to fungal contamination, young children may ingest mycotoxin levels far exceeding safety thresholds. For instance, a few studies have reported aflatoxin B1 concentrations, sometimes co-occurring with fumonisins and occasionally high levels, in maize, groundnuts, and their by-products, surpassing the EU regulatory limits [21,23], suggesting substantial exposure risks through daily consumption. The high ratio of food intake to body weight intensifies the dose-effect relationship, increasing the risk of adverse health outcomes such as growth impairment, immune dysfunction, and early-life liver toxicity [16,47]. These risks are further compounded by poor storage practices and limited regulatory enforcement, underscoring the urgency of targeted interventions in early childhood nutrition and food safety education in Cameroon [18].
Maternal dietary habits and breastfeeding may indirectly expose unborn babies and young breastfed children to food contaminants. This occurs especially when mycotoxins cross the placenta or are secreted into breast milk due to the mother's consumption of contaminated foods. In Cameroon, maternal dietary habits and breastfeeding practices may serve as significant indirect pathways for early-life mycotoxin exposure. Women of reproductive age often consume mycotoxin-contaminated staple foods such as maize, groundnuts, and their processed food products, which can lead to the transfer of these toxins to infants during pregnancy and lactation. Aflatoxins, particularly aflatoxin B1 and its metabolite aflatoxin M1, have been detected in breast milk [48,47], posing a serious risk to exclusively breastfed infants who’s immune and detoxification systems are still underdeveloped [45]. Two studies from Cameroon by Chuisseu et al. [48] and Tchana et al. [47] revealed high prevalence (38%, 16/42; and 2.8%, 3/62) of aflatoxins (mean: 7.4, range: 0.9-37 µg/L; and range: 0.005-0.625 µg/L) in breast milk respectively to be directly linked to maternal consumption patterns. This early exposure is associated with impaired immune responses, stunted growth, and increased vulnerability to infections during infancy [16,47,49,50]. The lack of food safety awareness and insufficient regulation of mycotoxins in local food systems further exacerbate this risk. Therefore, addressing maternal nutrition through public health education and improving food quality control are critical to reducing indirect exposure to mycotoxins in early childhood in Cameroon.
Children in Cameroon are particularly vulnerable to the toxic effects of mycotoxins during critical developmental windows, namely the prenatal period, infancy, and early childhood (Figure 2). During pregnancy, mycotoxins such as aflatoxins can cross the placental barrier, exposing the fetus to harmful compounds that may disrupt cellular development and immune function [50,51]. In regions of Cameroon where dietary staples like maize and groundnuts are frequently contaminated, maternal exposure may potentially result in utero exposure to toxins such as aflatoxin B1 and fumonisins. After birth, infants remain at risk through contaminated breast milk, particularly aflatoxin M1, which is transferred from the mother’s diet [49,50]. This exposure continues into early childhood, a phase characterized by rapid growth and high food consumption relative to body weight. Children often consume large quantities of locally produced unregulated foodstuffs, further increasing their intake of mycotoxins [17]. Exposure during these vulnerable windows has been linked to growth faltering, immune suppression, and increased susceptibility to infections. Some key factors that may make children more vulnerable to mycotoxins are presented in Table 1. Protecting children in Cameroon requires early-life interventions focused on maternal nutrition, safe weaning foods, and improved food safety policies.
Figure 2. Pathways of early-life exposure to mycotoxins and their critical windows of neurodevelopmental vulnerability
|
Factor |
Description |
|
Immature detoxification systems |
Reduced capacity of liver enzymes to metabolize and excrete mycotoxins. |
|
High food intake per body weight |
Children consume more food relative to body mass, increasing toxin exposure. |
|
Frequent consumption of contaminated foods |
Maize-based and groundnut-based weaning foods are common sources of exposure. |
|
Underdeveloped gut barrier |
An incomplete gastrointestinal barrier may enhance systemic absorption. |
|
Limited dietary diversity |
Low-nutrient varieties may reduce the protective effects of antioxidants or fiber. |
|
Breastfeeding exposure pathway |
Mycotoxins can be transferred through breast milk if the maternal diet is contaminated. |
Neurodevelopmental outcomes: linking exposure to disorders
Dietary exposure to mycotoxins remains a threat to human health, with adverse effects on brain development. To study the possible link between mycotoxin exposure and neurodevelopmental health outcomes, various animal models (Table 2) have been used to observe the possible behavioral changes, like cognitive deficits, motor and learning impairment after acute or chronic mycotoxin exposure [52]. Rodents (mice and rats) and zebrafish are the common animal models used for in vivo studies regarding mycotoxin exposure. Additionally, zebrafish have been reported as an efficient animal model to study neurotoxicity in the field of food science [53]. The effect of mycotoxin exposure on the central nervous system depends on the type of mycotoxins, the duration of exposure, the dose, and individual susceptibility [36]. Due to their high affinity with the brain, aflatoxin B1, fumonisin B1, and ochratoxin A can reach the nerve cells, inducing neurotoxicity with effects on neurodevelopmental and functional abnormalities. Several studies using laboratory animals have shown that exposure to mycotoxins induced oxidative stress, impairment of antioxidant defense mechanisms, mitochondrial dysfunction, neuroinflammation, and inhibited neurotransmission systems leading to learning and memory impairment, impairment of locomotor, depression, and anxiety-like behavior [33,52,54]. AFB1 exposure in zebrafish embryos and larvae showed locomotor function impairment and disruption of neural development [55]. AFB1 exposure in mice and rats revealed the memory and learning deficits, motor function decline, anxious-depressive-like behavior with an increase of acetylcholinesterase (AChE) activity in the brain and monoamine oxidase, and a reduction of antioxidant system markers [54,56,57]. Another study with OTA showed an increase in glutathione peroxidase, glutathione-s-transferase, and glutathione reductase levels and a decrease in non-protein thiol levels in the zebrafish brain with behavior changes [58]. Additionally, OTA and FB1 exposure in mice revealed the alterations of antioxidant state, induction of lipid peroxidation and apoptosis, modulation of neurotransmitters with behavioral changes [59,60].
|
Mycotoxins |
Animal model |
Findings |
References |
|
AFB1 |
Wistar rats |
Neurobiochemical changes (generation of ROS/RNS, mitochondrial dysfunction, release of proinflammatory cytokines) Neurobehavioral changes (memory and learning deficits, motor function decline, anxious-depressive-like behavior) Histopathological alterations in some brain regions leading to neuronal structure and functional impairment |
[54,56,57] |
|
Zebrafish |
Induces apoptosis via mitochondrial dysfunction, generation of ROS Impairment of behavior and neurodevelopment during early embryonic stages |
[55] |
|
|
Ochratoxin A |
Mice/rats |
Alterations of the antioxidant system, modulation of neurotransmitters, neurobiohavioral changes (cognitive deficit, motor and learning impairment) |
[60] |
|
Zebrafish |
Locomotor impairment, alteration in the expression of genes associated with brain development |
[58] |
|
|
Fumonisins |
Mice/rats |
Disruption of neurotransmitter homeostasis and exacerbation of oxidative stress with impairment of spatial memory and learning ability. |
[59,65]
|
Cereal-based products are of great importance in terms of mycotoxin exposure as they are especially consumed by infants (from 4 months to 1 year of age) and children (from 1 year of age to 12 years) and are the most contaminated by these toxic compounds, especially AFs, FBs, and OTA. Dietary exposure to these mycotoxins can have an impact on growth, the immune system, as well as cognitive function, with possible outcomes of neurodevelopmental disorders such as autism spectrum disorder [61,62]. Epidemiological studies on humans from high-exposure regions about mycotoxin exposure and neurodevelopmental disorders are scarce. Most studies done in Cameroon focused on the detection of the presence of mycotoxins in foods [9,11,47,63,64] and human biomonitoring analysis on body fluids [14,22,44]. According to Tchana et al. [47], infant and young children’s exposure to AFs in their diets contributes to malnutrition and stuntedness in children.
Dietary risk assessment and health implications
Estimating children's dietary intake of mycotoxins in Cameroon is essential for assessing health risks and guiding mitigation strategies. Children are particularly vulnerable due to their higher food intake per kilogram of body weight and reliance on staple foods such as maize, groundnuts, and other mycotoxin-prone crops. They may also be exposed indirectly through their mothers during pregnancy or breastfeeding. Studies have shown that these staples and breast milk frequently exceed international safety thresholds, contributing to chronic exposure in early life. Without accurate intake estimation, it is difficult to assess whether exposure levels exceed safe limits (which varies per mycotoxins, matrix, exposed individual’s age, etc.) such as the tolerable daily intake (TDI) set by international food safety authorities. Only two studies exist on children's urinary mycotoxin exposure assessment by Ediage et al. [14] (n=220, AFM1 range: 0.00006-0.048 μg/L) and Tchana et al. (n=42, 48.8%, AFB1 range: 0.109–2.840 μg/L). Furthermore, other biological specimens, such as blood from children and complementary foods, can be used to estimate dietary exposures in children. Additionally, breast milk from breastfeeding mothers, such as those in the studies by Chuisseu et al. [48] and Tchana et al. [47] might be used if the consumption frequencies of the breastmilk and amount of breast milk intake per day by the children were recorded.
In dietary risk assessment, the estimated daily intake (EDI) is a key metric used to quantify exposure to food contaminants, including mycotoxins, based on consumption levels and contaminant concentrations in food. This is particularly important for vulnerable populations such as children in Cameroon, where staple foods like maize and groundnuts are frequently contaminated [14,47]. EDI values are often compared to health-based guidance values such as the TDI, which represents the amount of a contaminant that can be ingested daily over a lifetime without appreciable health risk [66]. For genotoxic and carcinogenic mycotoxins like aflatoxin B1, a TDI is not established; instead, the Margin of Exposure (MOE) is used. MOE is calculated by dividing a benchmark dose by the EDI. An MOE below 10,000 indicates a potential health concern [67,68]. In Cameroon, a few biomonitoring studies have shown that the EDI for children and adults often exceeds safe levels [14,22,44], and MOE values for aflatoxins frequently fall below the acceptable threshold [43,69], underscoring a concern for children and the urgent need for mitigation strategies. Despite the few studies cited above in children, dietary exposure assessments remain limited in Cameroon, emphasizing the need for routine surveillance, food safety education, and policy enforcement to protect children from ongoing mycotoxin risks in Cameroon. Furthermore, health risk assessment using different health risk indicators such as liver cancer risk [43], the margin of exposure [43,69], and hazard quotient, is still underrepresented in Cameroon.
Recommendations to reduce mycotoxin risks and improve policy relevance in Cameroon
Protecting the Cameroonian children from mycotoxin exposure requires coordinated national action across multi-sectors including health, agriculture, and food safety sectors, ensuring a full stakeholder analysis [18]. Current evidence in Cameroon shows high exposure risks in maize-, groundnut- and complementary-food–dependent communities, placing infants and young children at increased risk of impaired growth [14,47] and neurodevelopment [7,25]. Cameroon can adapt successful African strategies, such as Ghana’s National Aflatoxin Policy, the ECOWAS Aflatoxin Action Plan, and PACA’s continental strategy, combined with proven interventions (improved drying, hermetic storage) to reduce exposure rapidly and sustainably [70–74].
Some priority actions include:
• Surveillance and Data Systems: Establish sentinel biomonitoring for children and pregnant women; build an integrated health-agriculture dashboard to track hotspots and guide response.
• Stronger Regulation and Market Control: Adopt/update national maximum limits aligned with Codex; enforce risk-based inspection, particularly for foods consumed by infants and children.
• Maternal and Child Nutrition Interventions: Integrate exposure-reduction messaging into Antenatal Care/Postnatal Care (ANC/PNC); promote dietary diversity and safe complementary feeding practices.
• Public Health Education: Use community radio, schools, women’s groups and health workers to promote low-cost mitigation such as proper drying and airtight storage.
• Agricultural and Post-Harvest Solutions: Scale proven drying and hermetic storage technologies; pilot biological control in high-risk zones.
• Research and Capacity Investment: Strengthen laboratory capacity, develop child-specific exposure models, and evaluate intervention effectiveness.
In all, monitoring success will focus on reduced contamination in foods and biomarkers, improved regulation compliance, and increased adoption of safer storage practices.
Conclusion
This review underscores an urgent yet underrecognized public health issue in Cameroon: children’s dietary exposure to neurotoxic mycotoxins and their potential role in neurodevelopmental disorders (NDDs). Common toxins like aflatoxin B1, fumonisin B1, and ochratoxin A, prevalent in staple foods such as maize and groundnuts, threaten early brain development, especially during prenatal, infant, and early childhood periods. Children’s heightened vulnerability is due to immature detoxification systems, underdeveloped blood-brain barriers, and maternal transfer during pregnancy and breastfeeding. These mycotoxins impair neurodevelopment through oxidative stress, mitochondrial dysfunction, neuroinflammation, and disrupted neurotransmission. Animal studies confirm associations with cognitive deficits, motor impairments, and behavioral changes resemble conditions like ASD and ADHD. In Cameroon, the situation is worsened by poor food storage, weak regulatory oversight, and low dietary diversity. However, national-level data on exposure and health effects remain scarce, and most studies focus narrowly on food contamination rather than holistic risk assessments. To reduce the impact of mycotoxins on child health, a coordinated response is needed, including: (1) establishing national biomonitoring systems; (2) conducting child-specific dietary risk assessments; (3) strengthening food safety regulation; and (4) educating the public on maternal and child nutrition and safe food handling. Strengthening these interconnected systems is essential to safeguard child health and food integrity.
Figure 3. Conceptual framework illustrating the pathways of mycotoxin exposure in Cameroon, the resulting neurodevelopmental risks in children, and the critical gaps in surveillance, regulation, maternal nutrition, and public health education.
References
2. Cainelli E, Bisiacchi P. Neurodevelopmental disorders: past, present, and future. Children (Basel). 2022 Dec 24;10(1):31.
3. Doi M, Usui N, Shimada S. Prenatal environment and neurodevelopmental disorders. Front Endocrinol (Lausanne). 2022 Mar 15;13:860110.
4. Hadders-Algra M. Early Diagnostics and Early Intervention in Neurodevelopmental Disorders-Age-Dependent Challenges and Opportunities. J Clin Med. 2021 Feb 19;10(4):861.
5. Wylie AC, Short SJ. Environmental toxicants and the developing brain. Biol Psychiatry. 2023 May 15;93(10):921–33.
6. Song C, Wang Z, Cao J, Dong Y, Chen Y. Neurotoxic mechanisms of mycotoxins: Focus on aflatoxin B1 and T-2 toxin. Environ Pollut. 2024 Sep 1;356:124359.
7. Abia WA, Foupouapouognigni Y, Nfombouot HP, Ngoungoure LV, Ntungwe EN, Salah-Abbès JB, et al. A scoping review on mycotoxin-induced neurotoxicity. Discov. toxicol. 2025 Feb 17;2(1):1.
8. Dai C, Tian E, Li H, Gupta SD, Hao Z, Wang Z, et al. Molecular mechanisms of aflatoxin neurotoxicity and potential neuroprotective agents. Food Sci Hum Wellness. 2024 Sep 1;13(5):2445–55.
9. Njumbe Ediage E, Hell K, De Saeger S. A comprehensive study to explore differences in mycotoxin patterns from agro-ecological regions through maize, peanut, and cassava products: A case study, Cameroon. J Agric Food Chem. 2014 May 21;62(20):4789–97.
10. Kana JR, Gnonlonfin BG, Harvey J, Wainaina J, Wanjuki I, Skilton RA, et al. Assessment of aflatoxin contamination of maize, peanut meal and poultry feed mixtures from different agroecological zones in Cameroon. Toxins (Basel). 2013 Apr 29;5(5):884–94.
11. Abia WA, Warth B, Sulyok M, Krska R, Tchana AN, Njobeh PB, et al. Determination of multi-mycotoxin occurrence in cereals, nuts and their products in Cameroon by liquid chromatography tandem mass spectrometry (LC-MS/MS). Food Control. 2013 Jun 1;31(2):438–53.
12. Fandohan P, Zoumenou D, Hounhouigan DJ, Marasas WF, Wingfield MJ, Hell K. Fate of aflatoxins and fumonisins during the processing of maize into food products in Benin. Int J Food Microbiol. 2005 Feb 15;98(3):249–59.
13. Wagacha JM, Muthomi JW. Mycotoxin problem in Africa: current status, implications to food safety and health and possible management strategies. Int J Food Microbiol. 2008 May 10;124(1):1–2.
14. Njumbe Ediage E, Diana Di Mavungu J, Song S, Sioen I, De Saeger S. Multimycotoxin analysis in urines to assess infant exposure: a case study in Cameroon. Environ Int. 2013 Jul;57-58:50–9.
15. Alvito P, Pereira-da-Silva L. Mycotoxin exposure during the first 1000 days of life and its impact on children’s health: A clinical overview. Toxins (Basel). 2022 Mar 4;14(3):189.
16. Turner PC, Moore SE, Hall AJ, Prentice AM, Wild CP. Modification of immune function through exposure to dietary aflatoxin in Gambian children. Environ Health Perspect. 2003 Feb;111(2):217–20.
17. Gong Y, Hounsa A, Egal S, Turner PC, Sutcliffe AE, Hall AJ, et al. Postweaning exposure to aflatoxin results in impaired child growth: a longitudinal study in Benin, West Africa. Environ Health Perspect. 2004 Sep;112(13):1334–8.
18. Abia WA. Aflatoxins: Probing the Multifaceted Impacts and Innovative Solutions for Agrifood Security and Safety, Economic Stability, Trade and Public Health in Cameroon. J Environ Toxicol Res. 2024;108(2):1–9.
19. Awuor AO, Wambura G, Ngere I, Hunsperger E, Onyango C, Bigogo G, et al. A mixed methods assessment of knowledge, attitudes and practices related to aflatoxin contamination and exposure among caregivers of children under 5 years in western Kenya. Public Health Nutr. 2023 Dec;26(12):3013–22.
20. Ayele M, Haile D, Alonso S, Sime H, Abera A, Balcha KH, et al. Aflatoxin exposure among children of age 12–59 Months in Butajira District, South-Central Ethiopia: A community based cross-sectional study. BMC Pediatr. 2022 Jun 2;22(1):326.
21. Khan R. Mycotoxins in food: Occurrence, health implications, and control strategies-A comprehensive review. Toxicon. 2024 Sep 1;248:108038.
22. Abia WA, Warth B, Sulyok M, Krska R, Tchana A, Njobeh PB, et al. Bio-monitoring of mycotoxin exposure in Cameroon using a urinary multi-biomarker approach. Food Chem Toxicol. 2013 Dec 1;62:927–34.
23. Abia WA, Warth B, Ezekiel CN, Sarkanj B, Turner PC, Marko D, et al. Uncommon toxic microbial metabolite patterns in traditionally home-processed maize dish (fufu) consumed in rural Cameroon. Food Chem Toxicol. 2017 Sep 1;107:10–9.
24. Pei X, Zhang W, Jiang H, Liu D, Liu X, Li L, et al. Food‐Origin Mycotoxin‐Induced Neurotoxicity: Intend to Break the Rules of Neuroglia Cells. Oxid Med Cell Longe. 2021;2021(1):9967334.
25. Ngoungoure LV, Abia WA, Owona BV, Foupouapouognigni Y, Elombo FK, Nfombouot HP, et al. Neurodevelopmental Outcomes of Mycotoxins Exposure and Effect on Brain Development in Infants and Young Children. Dev Neurobiol. 2025 Oct;85(4):e23000.
26. Li M, Tang S, Peng X, Sharma G, Yin S, Hao Z, et al. Lycopene as a therapeutic agent against aflatoxin b1-related toxicity: Mechanistic insights and future directions. Antioxidants (Basel). 2024 Apr 11;13(4):452.
27. Doi K, Uetsuka K. Mechanisms of mycotoxin-induced neurotoxicity through oxidative stress-associated pathways. Int J Mol Sci. 2011 Aug 15;12(8):5213–37.
28. Penalva-Olcina R, Juan C, Fernández-Franzón M, Juan-García A. Cell cycle and enzymatic activity alterations induced by ROS production in human neuroblastoma cells SH-SY5Y exposed to Fumonisin B1, Ochratoxin A and their combination. Toxicol In Vitro. 2023 Dec 1;93:105670.
29. Zuo M, Ye M, Lin H, Liao S, Xing X, Liu J, et al. Mitochondrial dysfunction in environmental toxicology: mechanisms, impacts, and health implications. Chem Res Toxicol. 2024 Nov 1;37(11):1794–806.
30. Nourbakhsh F, Tajbakhsh E. Neurotoxicity mechanism of Ochratoxin A. Qual Assur Saf Crops Foods. 2021 Apr 27;13(2):34–45.
31. Aytulun A, Ünal D. Neuroinflammation and autism: what have we learned so far? Int J Dev Disabil. 2024 Jun 12:1–21.
32. Mehrzad J, Malvandi AM, Alipour M, Hosseinkhani S. Environmentally relevant level of aflatoxin B1 elicits toxic pro-inflammatory response in murine CNS-derived cells. Toxicol Lett. 2017 Sep 5;279:96–106.
33. Adedara IA, Owumi SE, Oyelere AK, Farombi EO. Neuroprotective role of gallic acid in aflatoxin B1‐induced behavioral abnormalities in rats. J Biochem Mol Toxicol. 2021 Mar;35(3):e22684.
34. Penalva-Olcina R, Juan C, Fernández-Franzón M, Juan-García A. Involvement of pro-inflammatory mediators and cell cycle disruption in neuronal cells induced by gliotoxin and ochratoxin A after individual and combined exposure. Toxicol Lett. 2024 Mar 1;393:24–32.
35. Nimgampalle M, Chakravarthy H, Sharma S, Shree S, Bhat AR, Pradeepkiran JA, et al. Neurotransmitter systems in the etiology of major neurological disorders: Emerging insights and therapeutic implications. Ageing Res Rev. 2023 Aug 1;89:101994.
36. Kuć-Szymanek A, Kubik-Machura D, Kościelecka K, Męcik-Kronenberg T, Radko L. Neurotoxicological Effects of Some Mycotoxins on Humans Health and Methods of Neuroprotection. Toxins (Basel). 2025 Jan 6;17(1):24.
37. Richard-PhD SA. The novel neurotoxic and neuroimmunotoxic capabilities of aflatoxin B1 on the nervous system: a review. Adv Biosci Clin Med. 2020 Jan 1–8.
38. Kadry H, Noorani B, Cucullo L. A blood-brain barrier overview on structure, function, impairment, and biomarkers of integrity. Fluids Barriers CNS. 2020 Nov 18;17(1):69.
39. Obafemi BA, Adedara IA, Rocha JB. Neurotoxicity of ochratoxin A: Molecular mechanisms and neurotherapeutic strategies. Toxicology. 2023 Oct 1;497:153630.
40. Elangovan A, Singh A, Iyer M, Kumar SM, Kinoshita M, Krishnan J, et al. Unravelling the Mechanistic Approach of Aflatoxin Contaminated Food on Neurodegenerative Diseases-A Novel Approach. J Appl Toxicol. 2025 Oct;45(10):1947-67.
41. Monono EY, Abia WA, Mokake ND, Afanga YA, Monono EN, Ntane OA, et al. Aflatoxins in Cameroon: Occurrence, Potential Health Risks and the Way Forward in Implementing the Partnership for Aflatoxin Control in Africa (PACA) Country-Led Model. J Food Nutr Sci. 2024;9(4):222–33.
42. Nguegwouo E, Tchuenchieu A, Tene HM, Fokou E, Nama GM, Saeger SD, et al. Mycotoxin contamination of food and associated health risk in Cameroon: a 25-years review (1993-2018). Eur J Nutr Food Saf. 2019 Jan 21;9(1):52–65.
43. Aseneck MA, Mouafo HT, Tchana A, Dimala SC, Kamal HA, Kolawole O. AFLATOXINS CONTAMINATION IN NNAM OWONDO (A LOCAL GROUNDNUT-BASED FOOD), AND CONSUMERS’DIETARY EXPOSURES AND SAFETY LEVELS IN YAOUNDE, CENTRE REGION OF CAMEROON. Food Environ Saf. 2024 Sep 6;23(3).
44. Abia WA, Tchana AN, Djonabaye D, Šarkanj B, Mfopou EY, Ezekiel CN, et al. Assessment of urinary biomarkers of mycotoxin exposure in adults from Cameroon. World Mycotoxin J. 2024 Nov 22;17(3-4):191–203.
45. Sly PD, Flack F. Susceptibility of children to environmental pollutants. Ann NY Acad Sci. 2008 Oct;1140(1):163–83.
46. Tesfamariam K, De Boevre M, Kolsteren P, Belachew T, Mesfin A, De Saeger S, et al. Dietary mycotoxins exposure and child growth, immune system, morbidity, and mortality: a systematic literature review. Crit Rev Food Sci Nutr. 2020;60(19):3321–41.
47. Tchana AN, Moundipa PF, Tchouanguep FM. Aflatoxin contamination in food and body fluids in relation to malnutrition and cancer status in Cameroon. Int J Environ Res Public Health. 2010 Jan;7(1):178–88.
48. Chuisseu PD, Abia WA, Zibi B, Simo KN, Ngantchouko NC, Tambo E, et al. Safety of breast milk vis-a-vis common infant formula and complementary foods from western and centre regions of Cameroon from mycotoxin perspective. Rec Adv Food Scie. 2018 Apr;1:23–31.
49. Polychronaki N, West RM, Turner PC, Amra H, Abdel-Wahhab M, Mykkänen H, et al. A longitudinal assessment of aflatoxin M1 excretion in breast milk of selected Egyptian mothers. Food and Chemical Toxicology. 2007 Jul 1;45(7):1210–5.
50. Turner PC. The molecular epidemiology of chronic aflatoxin driven impaired child growth. Scientifica (Cairo). 2013;2013(1):152879.
51. Wild CP, Gong YY. Mycotoxins and human disease: a largely ignored global health issue. Carcinogenesis. 2010 Jan 1;31(1):71-82.
52. Furian AF, Fighera MR, Royes LF, Oliveira MS. Recent advances in assessing the effects of mycotoxins using animal models. Curr Opin Food Sci. 2022 Oct 1;47:100874.
53. Hou Y, Liu X, Qin Y, Hou Y, Hou J, Wu Q, et al. Zebrafish as model organisms for toxicological evaluations in the field of food science. Compr Rev Food Sci Food Saf. 2023 Sep;22(5):3481–505.
54. Del Fabbro L, Sari MH, Ferreira LM, Furian AF. Natural compounds mitigate mycotoxins-induced neurotoxicity by modulating oxidative tonus: in vitro and in vivo insights–a review. Food Addit Contam Part A. 2024 Apr 2;41(4):438–59.
55. Wu TS, Cheng YC, Chen PJ, Huang YT, Yu FY, Liu BH. Exposure to aflatoxin B1 interferes with locomotion and neural development in zebrafish embryos and larvae. Chemosphere. 2019 Feb 1;217:905–13.
56. Souto NS, Dassi M, Braga AC, Rosa ÉV, Fighera MR, Royes LF, et al. Repeated co‐exposure to aflatoxin B1 and aspartame disrupts the central nervous system homeostasis: behavioral, biochemical, and molecular insights. J Food Sci. 2023 Apr;88(4):1731–42.
57. Owumi S, Chimezie J, Salami MO, Ishaya JA, Onyemuwa CV, Nnamdi M, et al. Lutein and Zeaxanthin abated neurobehavioral, neurochemical and oxido-inflammatory derangement in rats intoxicated with Aflatoxin B1. Toxicon. 2025 Apr 1;258:108345.
58. Valadas J, Sachett A, Marcon M, Bastos LM, Piato A. Ochratoxin A induces locomotor impairment and oxidative imbalance in adult zebrafish. Environ Sci Pollut Res Int. 2023 Feb;30(8):21144–55.
59. Krishnaswamy K, Manasa V, Khan MT, Serva Peddha M. Apocynin exerts neuroprotective effects in fumonisin b1–induced neurotoxicity via attenuation of oxidative stress and apoptosis in an animal model. J Food Sci. 2024 Feb;89(2):1280–93.
60. Bhat PV, Anand T, Manu TM, Khanum F. Restorative effect of l-Dopa treatment against Ochratoxin A induced neurotoxicity. Neurochem Int. 2018 Sep 1;118:252–63.
61. Sá SV, Monteiro C, Fernandes JO, Pinto E, Faria MA, Cunha SC. Emerging mycotoxins in infant and children foods: A review. Crit Rev Food Sci Nutr. 2023 May 7;63(12):1707–21.
62. Adaku Chilaka C, Mally A. Mycotoxin occurrence, exposure and health implications in infants and young children in Sub-Saharan Africa: A review. Foods. 2020 Nov 1;9(11):1585.
63. Bouelet Ntsama IS, Frazzoli C, Pouokam GB, Colizzi V. Occurrence and dietary risk assessment of mycotoxins in most consumed foods in cameroon: Exploring current data to understand futures challenges. Foods. 2023 Apr 20;12(8):1713.
64. Njobeh PB, Dutton MF, Koch SH, Chuturgoon AA, Stoev SD, Mosonik JS. Simultaneous occurrence of mycotoxins in human food commodities from Cameroon. Mycotoxin Res. 2010 Feb;26(1):47–57.
65. Yang D, Ye Y, Huang Y, Huang H, Sun J, Wang JS, et al. Effects of FB1 and HFB1 on autonomous exploratory and spatial memory and learning abilities in mice. J Agric Food Chem. 2023 Oct 11;71(44):16752–62.
66. WHO Food Additives Series. Safety evaluation of certain contaminants in food: prepared by the eighty-third meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). Geneva: WHO Food Additives Series; 2018. avialable from https://www.who.int/publications/i/item/9789241660747
67. European Food Safety Authority (EFSA). Opinion of the scientific panel on contaminants in the food chain [CONTAM] related to the potential increase of consumer health risk by a possible increase of the existing maximum levels for aflatoxins in almonds, hazelnuts and pistachios and derived products. EFSA J. 2007 Mar;5(3):446.
68. EFSA CONTAM Panel (EFSA Panel on Contaminants in the Food Chain). Scientific opinion–Risk assessment of aflatoxins in food. EFSA J. 2020;18(3).
69. Dimala S. Charlette, Aseneck A. Marchel, Kamal A. Hassan, Achidi U. Aduni, Tchana N. Angele, and Wilfred A. Abia (2024). Food Security, Dietary Intake, Foodways, and Aflatoxins-Food Safety Levels amongst the Elderly Population (50 Years and Above) of Obam Ongola Community in Yaoundé, Cameroon, J. Nutrition and Food Processing, 7(11);
70. Government of Ghana. National Policy for Aflatoxin Control in Food and Feed. First Edition. ACCRA, Ghana; 2022. pp. 1–137.
71. Pretari A, Hoffmann V, Tian L. Post-harvest practices for aflatoxin control: Evidence from Kenya. J Stored Prod Res. 2019 Jun;82:31–9.
72. Economic Community of West African States (ECOWAS). Aflatoxin Control Action Plan for ECOWAS Member States (ECOACAP). Abuja: Economic Community of West African States (ECOWAS); 2014. Accessed on November 4, 2025. Available from: https://www.scribd.com/document/730586176/ECOWAS-Aflatoxin-Action-Control-Plan-FINAL-DRAFT.
73. Partnership for Aflatoxin Control in Africa (PACA). PACA Strategy 2013–2022. African Union Commission; 2013. Accessed November 4, 2025. Available from: https://archives.au.int/handle/123456789/5553.
74. Baoua IB, Amadou L, Ousmane B, Baributsa D, Murdock LL. PICS bags for post-harvest storage of maize grain in West Africa. J Stored Prod Res. 2014 Jul 1;58:20–8.