Abstract
Background: Transcutaneous electrical nerve stimulation (TENS) has emerged as a significant intervention in rehabilitating spinal cord injury (SCI). It has shown promising results in restoring function and improving outcomes for individuals with SCI throughout the current evidence.
Objectives: To give a thorough recapitulation of the most recent systematic reviews (SRs) on the role of TENS in the rehabilitation of SCI.
Method: Several medical databases including MEDLINE (PubMed), Scopus, Web of Science, and Cochrane Library were searched systematically for citations published up until June 2023. Two reviewers thoroughly screened the research, gathered pertinent data, and assessed the caliber of the retrieved evidence. The quality of the SRs was evaluated using the JBI checklist for SRs.
Results: Following an initial screening of 736 articles, 16 satisfied the inclusion criteria. TENS showed promising effects for various rehabilitative purposes, including spasticity, pain, motor function, erectile function, and cognitive function with no major adverse effects in SCI patients.
Conclusion: This comprehensive overview of the literature outlines the currently available data for the use of TENS in SCI. However, to establish standardized procedures for improving the rehabilitation of SCI, further high-quality investigations are required.
Keywords
Spinal cord injury, Transcutaneous electrical nerve stimulation, TENS, Rehabilitation, Umbrella review, Systematic review
Introduction
Spinal cord injury (SCI) is damage to the spinal cord caused by disease, degeneration, or trauma (such as a car accident). The yearly incidence of SCI is believed to be between 40 and 80 cases per million people, while the world health organization (WHO) notes that a precise worldwide prevalence is unknown. Up to 90% of SCI cases have traumatic etiology, however the proportion of spinal cord injuries that are not traumatic seems to be increasing [1]. As a result of the disruption of the neuronal connections between the centers in the upper and lower central nervous system, SCI causes severe, long-lasting neurological damage that leads to including sensory and motor dysfunction, aberrant reflexes, and autonomic abnormalities [2,3]. Despite the high incidence, mortality, and severe outcomes in survivors, no definite treatment has been identified for this condition.
Further, SCI has a significant impact on the number of years spent with a disability, especially among younger people [4,5]. Hence, in order to enhance the functional results and quality of life (QoL) for people with SCI, it is crucial to find efficient rehabilitation methods [6,7]. The process of SCI rehabilitation is difficult and complex, necessitating creative thinking to support functional recovery per individual needs. Also, early rehabilitation is also essential to avoid bone loss, muscular atrophy, joint contractures, and to maintain healthy digestive and respiratory systems [8].
One well-known method that has been used extensively in rehabilitative medicine is transcutaneous electrical nerve stimulation (TENS) [9-14]. TENS consists of delivering electrical impulses to the nerves through the skin for modulating the neuronal activity and therapeutic purposes [15]. The stimulation protocol is modifiable in terms of rate delivery, intensity and duration [16]. This method has gained attention from researchers due to its non-invasiveness, relative cost-effectiveness, and benefits for a wide range of conditions. Application of TENS in SCI patients has been the subject of several studies for controlling different SCI-associated outcomes, including pain [17,18], motor function [19], spasticity [20,21], dysesthesias [22], bladder control and sexual function [23].
Umbrella reviews are a type of systematic review (SR) conducted with the intention of contrasting existing SRs and providing a comprehensive picture of a body of knowledge centered on a particular issue. Therefore, the strong degree of evidence provided by these studies can benefit both doctors and policymakers [24]. In the current study, we aimed to systematically review the current SRs regarding the application of TENS with rehabilitative purposes in SCI patients.
Methods
This umbrella review was conducted in accordance with the Joanna Briggs Institute (JBI) and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria [25].
Registration and protocol
The protocol was entered into PROSPERO/NHS, the International Prospective Register of Systematic Reviews (ID: CRD42023449882).
Review question
In order to establish the search strategy, the following research question was developed: “What evidence is currently available from SRs about the rehabilitative effects of TENS on SCI patients?”
Search strategy
A systematic search strategy was designed to retrieve the publications on the application of TENS on SCI patients. MEDLINE (via PubMed), Scopus, Web of Science, and Cochrane library were searched using English search terms from inception to June 2023.
The search strategy included the terms for target population (SCI), intervention (TENS), and study design (Systematic review). Different combinations of the terms were made using “OR” and “AND” to achieve a specific selection of literature (Table 1). To maximize retrieval of relevant studies, we did not include the "outcome" and "comparison" sections of the PICOS strategy.
|
Population |
Intervention |
Study design |
|
Spinal cord injury |
Transcutaneous electrical nerve stimulation |
Systematic review |
|
Paraplegia |
Transcutaneous electrostimulation |
Meta-analysis |
|
Quadriplegia |
Transcutaneous neuromodulation |
|
|
Tetraplegia |
Transcutaneous nerve stimulation |
|
|
Spinal cord transection |
Transcutaneous tibial nerve stimulation |
|
|
Spinal cord disease |
Transcutaneous spinal cord stimulation |
|
|
Myelopathy |
Functional electrical stimulation |
|
|
Myelitis |
Neuromuscular electrical stimulation |
|
|
Cord injury |
Electrostimulation |
|
|
SCI |
Electrical stimulation |
|
|
Neuromodulation |
||
|
TENS |
||
|
FES |
||
|
TSCS |
||
|
* Note: The OR Boolean operator was used between the terms in each column, while AND was used to combine the columns. |
||
All included studies' bibliographies were carefully searched, and authors of studies on electro-rehabilitation for SCI who had more publications detected during the search were manually located.
Inclusion and exclusion criteria
The following criteria were used for inclusion:
- Studies involving adult subjects (≥18 years old) who have full or partial lesions with a traumatic or nontraumatic etiology of SCI.
- Studies using any transcutaneously applied electrical stimulation, including transcutaneous spinal cord stimulation (TSCS) and transcutaneous tibial nerve stimulation (TTNS).
- Studies that reported the therapeutic (i.e. long-term) or orthotic (i.e. immediate) effects of electrical stimulation with rehabilitative purposes.
- SR studies, with or without a meta-analysis.
- Published in English.
The following were the exclusion criteria:
- Applying any other form of invasive or minimally invasive electrostimulation or other rehabilitation technique in the absence of TENS.
- Research on animal subjects and basic experiments, original publications, overviews, letters, summary of meetings, etc.
- Unpublished or duplicate literature.
- Unavailable full text.
Furthermore, SRs with mixed clinical populations were considered eligible if they specifically provided data for the SCI.
Selection process
After removing duplicate records, two unbiased reviewers evaluated the titles and abstracts of the articles (S.E, F.T). To determine eligibility, the whole texts of publications relevant to the study's objectives were then carefully examined. The same two reviewers had a discussion to settle any disagreements over the study selection.
Data extraction
After data extraction, two reviewers worked separately to provide results on standardized spreadsheets. The overall and methodological characteristics of SRs were retrieved, which included the following items: Bibliographic information (First author, year of publication, and origin), design (SR +/- MA), searched databases, number and type of the included studies, type of the intervention, method of quality appraisal and whether they graded the evidence, outcomes, adverse events (AEs), and the main results.
Appraising the quality
Two independent reviewers (R.GH, S.S) evaluated the methodological quality of the included SRs using the JBI criteria for SRs (Appendix). Eleven questions are on this checklist to help in evaluating SRs. ‘Yes’, ‘no’, ‘unclear’, and not applicable ‘NA’ can be used to respond to each question. If there were 0-1, 2-3, or more than 3 no/unclear responses, the SRs were classified as high, moderate, or low quality, accordingly. A third reviewer was consulted in case there was any dispute.
Data synthesis
A quantitative data analysis was not feasible for this research due to the fact that the included SRs encompassed a wide range of intervention techniques and outcome measures. This heterogeneity made it challenging to perform a re-analysis and calculate summary effect sizes. A narrative synthesis method was utilized to analyze and interpret the data extracted from included SRs. This approach involved a comprehensive examination of the findings and key themes identified across the selected studies, allowing for a qualitative synthesis of the evidence.
Results
Search results
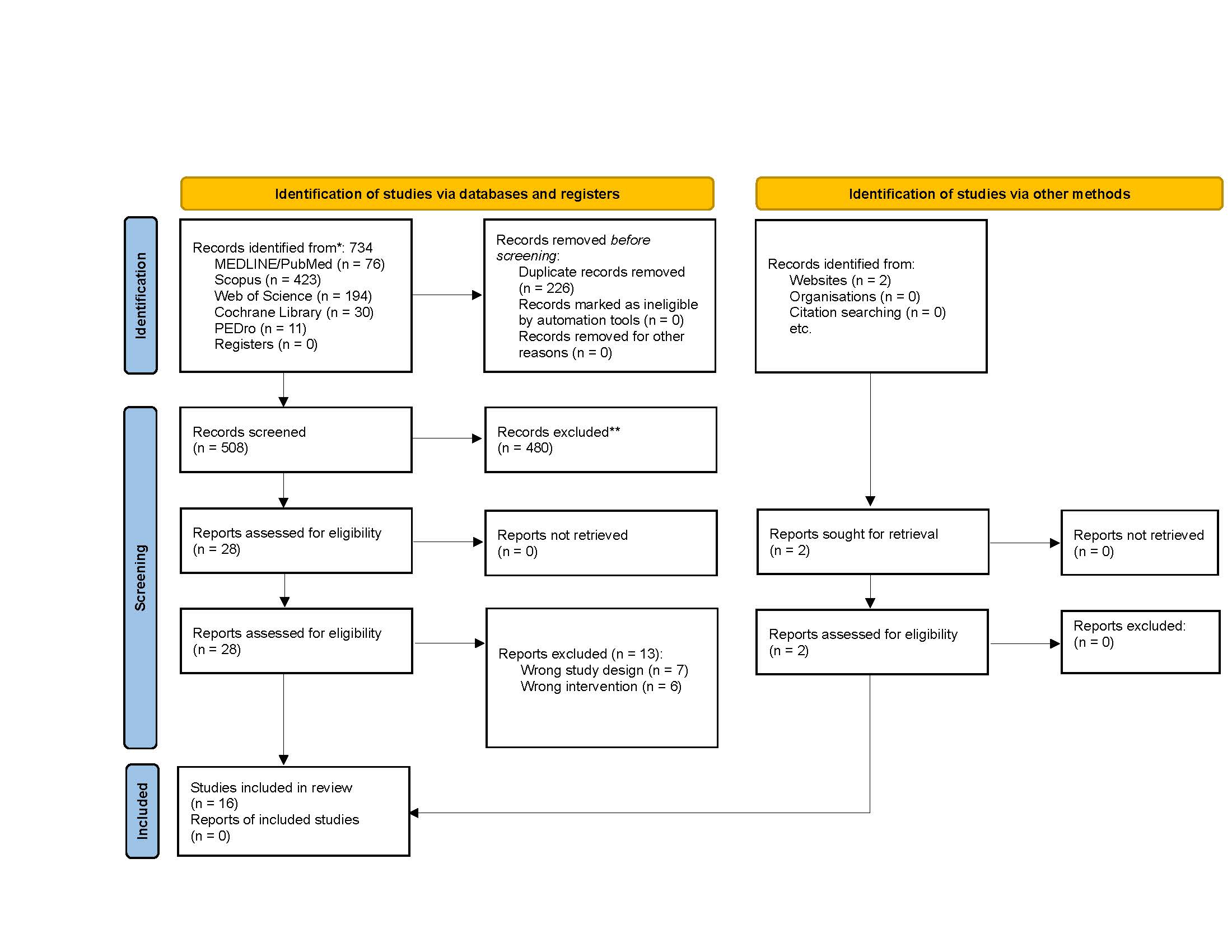
A total of 735 citations were retrieved through the systematic search of databases and 1 from the manual search, 510 citations remained after removing duplicates and were assessed based on their title and abstract. Of these, 30 articles were selected to undergo full evaluation. Finally, 16 SRs entered the study [19,26-40]. The selection process is demonstrated in Figure 1.
Figure 1. PRISMA 2020 flow diagram for new systematic reviews which included searches of databases, registers, and other sources.
Overall characteristics
Overall, meta-analysis was conducted by 6/16 SRs [30,32,35,36,39,40]. Studies were conducted from 2014 [34] to 2022 [32,37,40], originating from all over the globe, including the United States [34,37], Canada [33,35], Switzerland [26,29], Brazil [28,39], UK [32], Spain [19,27], Australia [30], Ireland [38], New Zealand [36], and China [31]. All the SRs reported the use of a form of TENS. TSCS was investigated through 4 SRs [19,32,37,38] and TTNS by 2 SRs [29,35].
There were no serious AEs, but a few SRs did report brief cutaneous irritation where the electrodes were placed, [19,33,34,36,38]. One patient in one of the trials did not tolerate stimulation and therefore exited the study [29]. One SR noted a little increase in tone 24 hours following treatment, unintentionally activating the micturition reflex and urinating while standing, experiencing pain during intense stimulation, and experiencing asymptomatic changes in blood pressure and pulse rate, in addition to modest side effects that might be caused by cervical stimulation, such as dizziness, flushing, seasickness, metallic taste, "sharp" breathing sensation, neck soreness, and sore throat [38].
Further details are demonstrated in Table 2.
|
|
||||||||||
|
AUTHOR, YEAR |
Origin |
Design |
Searched databases |
Type of intervention |
Number and type of the included studies |
Quality appraisal |
GRADE |
Main outcome |
AE profile |
Overall results |
|
Megía Garcia et al. (2020) [19] |
Spain
|
SR
|
PubMed, CENTRAL, PEDro
|
TSCS |
13 (10 case report or series, 2 cross-over trials, 1 sham-controlled trial) |
CARE (Case Report Guidelines) scores
|
Yes |
Motor function
|
No apparent AE other than cutaneous irritation after repeated stimulation |
All studies reported an increase in motor response measured by recording surface EMG, voluntary movement, muscle strength, or function. |
|
Lu et al. (2015) [31] |
China |
SR |
Medline, Web of Science, EMBASE, CINAHL, PEDro |
Various, including TENS and FES |
16 studies: 4 ES: 2 TENS, 2 FES |
Downs and Black checklist |
No |
Upper limb muscle strength |
- |
Inconsistent results from TENS studies. |
|
Stampas et al. (2022) [37] |
USA
|
SR
|
PubMed, Scopus, Embase, CINAHL, CENTRAL, ClinicalTrials.gov
|
TSCS |
8 human trials and 9 animal studies
|
1. Human studies: Cochrane RoB Tool 2. Animal studies: SYRCLE’s
|
No |
Spasticity |
None |
TENS was the object of only one study which showed using several outcome measures, none of which achieved significant improvement. |
|
Massey et al. (2022) [32]
|
UK
|
SR + MA
|
PubMed, Web of Science, Scopus, and Cochrane Central Register of Controlled Trials databases
|
TENS, TSCS, FES |
Total: 29/ 10 TENS (9 for the lower limbs and 1 for the upper limbs); 6 TSCS, all targeting the lower limbs; 9 FES cycling; 3 used FES gait; and one assessed FES for the lower limbs |
PEDro
|
No |
Main: spasticity / Secondary: the Hoffman (H)- reflex,
|
- |
The use of TSCS and TENS, which did not induce a muscle contraction, indicate that activation of afferent fibers is at least required for non-invasive ES to reduce spasticity. |
|
Mills et al. (2016) [33]
|
Canada
|
SR
|
Medline, Embase, CENTRAL
|
Different TENS protocols for different locations including acupuncture points. |
14 RCTs, 3 on SCI
|
PEDro
|
Yes
|
Spasticity of ankle plantar flexors, spasm frequency scale, painful spasm scale, lower limb Ashworth score, clonus score, deep tendon reflex score, plantar stimulation response score
|
Mild skin irritation with erythema that resolved spontaneously in one study, no AE in other studies. |
There is level 1 and 2 evidence for its short-term effectiveness in the management of spasticity, in various neurologic etiologies, TENS can adjunct therapy to limb spasticity management with limited AEs. |
|
Harvey et al. (2016) [30] |
Australia
|
SR + MA
|
Medline, CINAHL, Embase, CENTRAL, PEDro
|
22 commonly administered
physiotherapy interventions; including TENS |
Total: 38 RCTs, TENS: 2
|
PEDro
|
Yes
|
Pain
|
- |
TENS was an effective treatment for managing pain based on two RCTs. |
|
Pacheco et al. (2021) [35] |
Canada
|
SR + MA
|
Medline, Central, Embase, Scopus, PsycINFO and PubMed
|
TTNS |
Total: 11 RCTs, TENS: 1
|
NIH tools
|
No
|
Main: cystometrogram parameters; Secondary: Cognition, safety
|
- |
TTNS is a safe and feasible modality that can be performed during inpatient rehabilitation of acute tSCI.
|
|
Afferi et al. (2020) [26] |
Switzerland |
SR |
PubMed, EMBASE |
All currently available treatment options for erectile dysfunction in spinal cord injury men; including TENS. |
Total: 47, TENS: 1 |
None |
No |
Erectile dysfunction |
- |
In the TENS groups, the IIEF-5 score was higher than the control group |
|
Garcia et al. (2019) [28] |
Brazil |
SR |
Scopus, PubMed, BVS, Google Scholar and BASE databases |
Somatosensory electrical stimulation including TENS |
Total: 10 TENS/SCI: 3 |
None |
No |
Spasticity |
- |
Positive effects in spastic spinal cord injured patients were reported. |
|
Yang et al. (2022) [40] |
China |
SR + MA |
PubMed, Embase, CENTRAL, CNKI, Wanfang, and Vip databases |
TENS |
6 RCTs |
Cochrane RoB tool |
No |
Pain |
- |
Based on limited evidence, TENS has some clinical therapeutic effects on SCI-associated pain.
|
|
Thomaz et al. (2019) [39]
|
Brazil |
SR + MA |
MEDLINE, CENTRAL, CINAHL, PEDro, PsycINFO, Embase |
TENS + FES |
7 RCTs |
PEDro |
No |
1. Muscle volume; 2. Spasticity |
- |
ES was found to be an effective method for increasing muscle volume in SCI patients, but had no effect on spasticity. |
|
Parittotokkaporn et al. (2021) [36] |
New Zealand |
SR + MA |
MEDLINE, Embase, WoS, Scopus, and Cochrane library |
TENS |
6 RCTs |
Cochrane RoB Tool |
No |
1. The primary outcomes: MCC and maximum detrusor pressure (Pdet). 2. Secondary outcome: urodynamic tests. 3. EMG |
Transient skin redness after surface electrodes in one study, none in others |
TENS may be an effective, safe intervention for neurogenic bladder dysfunction following SCI. |
|
Moreno-Duarte et al. (2014) [34] |
USA |
SR |
PubMed |
TENS, tDCS, rTMS, CES, SCS, and MCS |
8 clinical trials (1 TENS) |
Jaded scale |
No |
Chronic pain |
Skin burns and irritation |
According to 1 study, low frequency TENS has also shown to induce a release of enkephalins and endorphins to control pain. |
|
Gross et al. (2016) [29] |
Switzerland |
SR + MA |
Embase, Medline, CENTRAL, and Health Technology Assessment Database |
TENS (including TTNS) |
22 (2 RCTs, 14 prospective cohorts, 5 retrospective case series, 1 case report) |
Cochrane RoB for RCTs, self-defined criteria for non-RCTs |
No |
Efficacy of TENS in treating NLUTD; urodynamic evaluations, bladder diary |
One patient did not tolerate stimulation and stimulation had to be stopped. No other AEs have been reported |
The positive impact on urodynamic and bladder diary parameters as well as the favorable AE profile, indicates that TENS might be effective and safe for treating NLUTD. |
|
Taylor et al. (2021) [38] |
Ireland |
SR |
EMBASE, MEDLINE (Ovid), Web of Science |
TSCS |
25 |
Downs and Black Quality checklist |
Yes |
Reflex responses and motor recovery |
Limited reports; 4 studies, complete absence of AEs; 1 study: a modest increase in tone in the 24 hrs post-treatment, unintentional activation of the micturition reflex and voiding during standing, skin breakage, and transient redness, as well as discomfort during stimulation at high intensities, asymptomatic variations to heart rate and blood pressure and mild side effects possibly related to cervical stimulation including incidents of lightheadedness, feeling flushed, an sea, a metallic taste, a sensation of ‘sharp’ breathing, neck pain, and throat discomfort. |
Limited evidence. |
|
Fernández-Tenorio et al., (2019) [27] |
Spain |
SR |
PubMed, PEDro, and CENTRAL |
TENS |
10, SCI: 2 |
PEDro scale |
No |
Spasticity |
None |
TENS is recommended as a potential treatment for spasticity due to its low cost, ease of use, and absence of AE. |
|
AE: Adverse Events; CARE score: Case Report Guidelines Scores; CENTRAL: Cochrane Central Register of Controlled Trials; CES: Cranial Electrotherapy Stimulation; CINAHL: Cumulative Index to Nursing and Allied Health Literature; CNKI: China National Knowledge Infrastructure; EMG: Electromyography; ES: Electrical Stimulation; FES: Functional Electrical Stimulation; IIEF-5: International Index of Erectile Function-5; MA: Meta-Analysis; MCC: Maximum Cystometric Capacity; MCS: Motor Cortex Stimulation; NIH: National Institutes of Health; NLUTD: Neurogenic Lower Urinary Tract Dysfunction; PEDro: The Physiotherapy Evidence Database; RCT: Randomized Controlled Trial; RoB: Risk of Bias; rTMS: Repetitive Transcranial Magnetical Stimulation; SCI: Spinal Cord Injury; SCS: Spinal Cord Stimulation; SR: Systematic Review; SYRCLE: Systematic Review Centre For Laboratory Animal Experimentation; tDCS: Transcranial Direct Current Stimulation; TENS: Transcutaneous Electrical Nerve Stimulation; tSCI: Traumatic Spinal Cord Injury; TSCS: Transcutaneous Spinal Cord Stimulation; TTNS: Transcutaneous Tibial Nerve Stimulation; WoS: Web of Science |
||||||||||
Application of TENS for various rehabilitative purposes
Spasticity: Six SRs investigated the use of TENS for managing spasticity [27,28,32,33,37,39]. Garcia et al. [28] investigated the use of somatosensory electrical stimulation for relieving spasticity with various etiologies. According to the 3 studies that investigated the SCI patients, an overall improvement was reported by these studies. One of these studies reported that TENS failed to modulate Hoffman (H)-reflex. Further, one trial showed that the efficacy of TENS on spasticity depends on repeated sessions.
Mills and Dossa [33] summarized the randomized controlled trials (RCTs) reporting the effects of TENS for managing limb spasticity with different etiologies (SCI in 3/14 RCTs). Three categories were created by them to group intervention protocols: 1) TENS vs no TENS or placebo TENS, 2) TENS versus another TENS treatment or another spasticity management intervention, and 3) TENS as an adjuvant to another spasticity management strategy. In the first group, according to only one double-blinded RCT, the use of The International Classification of Functioning (ICF) body structure and function domain showed better outcome measures after a single 60-minute TENS treatment over the common peroneal nerve of the limb with dominant spasticity, but not in the Achilles tendon jerks or ankle clonus [21]. The results of one RCT in the second group, which compared 15 daily consecutive treatments of 15 minutes of TENS on the tibial nerve to baclofen, revealed equal outcomes in all end measures, but not in the Hmax/Mmax ratio, H-reflex delay, clonus score, or the plantar stimulation response score [20]. In the final group, 60 minutes of TENS before 15 sessions of standardized physical therapy interventions decreased immediate and short-term Composite Spasticity Scale scores compared to physical therapy alone, per one high-quality RCT on people with SCI-induced lower limb spasticity [41].
Massey et al. [32] investigated the use of different electrostimulation techniques, including TENS and TSCS, on SCI-induced spasticity. Of the 29 research, 10 examined the impact of TENS on spasticity—9 focusing on the lower limbs and 1 on the upper limbs—while 6 utilized TSCS, all of which targeted the lower limbs. The other studies employed various forms of functional electrical stimulation (FES). Their findings indicate that non-invasive electrostimulation to reduce spasticity requires, at minimum, afferent fiber activation, as demonstrated by the application of TSCS and TENS, which did not result in a muscular contraction.
Stampas et al. [37] evaluated the rehabilitative methods for the prevention of spasticity in SCI. They gathered evidence from both human and animal trials. In human trials, only 1/8 of the study examined spasticity as the primary endpoint with a sample size big enough to test the theory. In this study, compared to controls, neuromodulation of the spinal cord using electric stimulation of the common peroneal nerve decreased stiffness in the lower limbs. Other trials covered this trial as well [41].
Two TENS trials were included in Thomaz et al. SR's [39] investigation of the impact of electrical stimulation on skeletal muscle volume and spasticity in SCI. However, both of these trials were already covered by other SRs [20,41]. Similarly, Fernández-Tenorio et al. study investigated the effects of application of TENS for spasticity with different etiologies, including two trials on SCI patients [27]. Both of these trials were covered by the previous publications [20,21].
Pain: Applicating TENS for managing pain in SCI was mentioned in 3 SRs [30,34,40]. In Moreno-Duarte et al. [34] SR, different electrical and magnetic neural stimulation techniques for treating chronic pain in SCI were examined. They included only one crossover, unblinded, clinical trial that applied high frequency (HF)-TENS vs low frequency (LF)-TENS for two weeks. The results of these trials suggest that with no discernible difference between HF and LF, TENS deserves investigation as an adjunctive therapy in SCI patients suffering from neuropathic pain [42]. The goal of Harvey et al. [30] was to evaluate the efficacy of 22 frequently used physiotherapy methods for SCI patients. TENS was reported in 2 of their included trials [17,43]. In one, TENS was found to significantly reduce pain in people with SCI [17]; while in the other, LF-TENS was proposed as a potential treatment for SCI pain [43]. Yang et al. [40] conducted a meta-analysis of 6 RCTs with 165 SCI patients. The visual analog scale (VAS) and short-form McGill pain questionnaire ratings were lower in the experimental group as compared to the control, according to the results of a meta-analysis (Mean difference [MD] = −1.52, 95%CI, −2.44 to −0.60, P = 0.001, MD = −0.70, 95% CI, −1.03 to −0.25, P = 0.002, respectively).
Motor function: Three SRs reported outcomes related to the rehabilitation of motor function in SCI [28,31,32] which can be categorized into upper and lower motor function.
Among the included studies on upper motor function, the results were conflicted; in one research, two patients with traumatic spinal cord injuries (tSCIs) who received TENS in addition to their usual rehabilitation therapy exhibited improvements in hand function, motor control, and finger muscle strength [44]. The other reported that applying TENS modulated cervical spinal circuitry to improve volitional control of upper motor function in tetraplegic individuals [45]. Another study found that giving SCI patients a 12-week TENS treatment over their wrist extensor muscles boosts their fatigue resistance regardless of their exercise regimen. Nevertheless, only the high-resistance protocol increased muscle strength and was shown to improve muscle aerobic metabolism after training compared to the low-resistance protocol [46]. Nevertheless, a different RCT revealed that tetraplegic people's voluntary wrist strength does not enhance when TENS is added to progressive resistance training programs [47].
Regarding lower motor function, in one trial the subjects executed two types of ankle movements: irregular ankle movements along a predefined trajectory with and without tonic TSCS, and rhythmic ankle movements (plantar and dorsiflexion) at a predefined pace. The effects of TSCS on aided over-ground walking were investigated in a subgroup of eight individuals. The outcomes demonstrated that ankle movement coordination remained unaltered throughout stimulation. The measured polysynaptic spinal reflex showed a considerable reduction of pathogenic activity unique to SCI, indicating that TSCS at 30 Hz regulated spinal reflex activity. In the three with the lowest walking function scores as well as the one with the highest, the subgroup showed positive stimulation effects, such as increased maximum walking speed or more continuous and faster stepping at a self-selected speed [48]. For self-assisted standing following chronic motor and sensory full paralysis with no AEs, TSCS modified lumbosacral spinal networks in a study including fifteen SCI patients [49].
Neurogenic lower urinary tract dysfunction (NLUTD): NLUTD was addressed by 2 SRs [29,36]. Gross et al. SR [29] reported use of TENS for NLUTD, originated from different conditions, including SCI. Their research suggests that TENS is a safe and efficient therapeutic option for those with NLUTD. The other SR by Parittotokkaporn [36] explicitly investigated the SCI-induced NLUTD. Through the meta-analysis of 6 RCTs with 353 individuals, they showed that TENS significantly increased maximum cystometric capacity (standardized mean difference [SMD]: 1.11, 95% confidence interval [CI]: 0.08–2.14, p = 0.03, I2 = 54%) in acute SCI with no AE. However, it did not affect the maximum detrusor pressure.
Erectile dysfunction: Only one SR was retrieved in this category, which included one trial regarding the application of TENS in the management of post-SCI erectile dysfunction [26]. According to this trial, for patients with partial suprasacral SCI, TENS of the pelvic floor muscles is a promising, cost-efficient, safe, and effective physical therapy approach [50]. Although this trial reported measures of urinary function, it was not included in the SRs regarding NLUTD, mentioned in the previous category.
Cognition: Same as the previous category, cognitive function was addressed by one SR in one trial. The impact of therapies for post-injury cognitive impairment in people with tSCI was analyzed by Pacheco et al. SR [35]. In one study, Stampas et al. [37] looked at the effectiveness of TTNS in patients with acute SCI who also had NLUTD. Regarding the safety of TTNS, one of the main findings of this study was cognition. At the time of inpatient rehabilitation release, which occurred on average 16 days after admission, there were no statistically significant changes in cognitive scores between patients who got TTNS therapy and those who did not.
Quality assessment for included studies
As shown in Table 3, based on the JBI quality assessment tool, 9/16 SRs had a low quality [19,27,28,30-32,34,37,40], 5/16 had moderate quality [26,29,33,38,39], and only 2/16 SRs were high quality [35,36]. The main category in which SRs lacked in quality was the assessment of the publications bias which was only reported by 3/16 [36,39,40].
|
Citation |
Q1** |
Q2 |
Q3 |
Q4 |
Q5 |
Q6 |
Q7 |
Q8 |
Q9 |
Q10 |
Q11 |
Overall quality |
|
Moreno-Duarte et al. (2014) [34] |
Y |
Y |
N |
N |
Y |
U |
Y |
Y |
N |
U |
Y |
Low |
|
Lu (2015) [31] |
Y |
N |
Y |
Y |
Y |
U |
U |
Y |
N |
Y |
N |
Low |
|
Gross et al. (2016) [29] |
Y |
Y |
U |
Y |
N |
U |
Y |
Y |
N |
Y |
Y |
Moderate |
|
Mills et al. (2016) [33] |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
N |
U |
N |
Moderate |
|
Harvey et al. (2016) [30] |
U |
Y |
N |
Y |
Y |
Y |
Y |
Y |
N |
U |
Y |
Low |
|
Garcia et al. (2019) [28] |
Y |
U |
Y |
N |
N |
N |
N |
N |
N |
U |
N |
Low |
|
Thomaz et al. (2019) [39] |
Y |
Y |
N |
Y |
Y |
U |
Y |
Y |
Y |
Y |
N |
Moderate |
|
Fernández-Tenorio et al. (2019) [27] |
Y |
Y |
U |
Y |
Y |
U |
U |
Y |
N |
N |
Y |
Low |
|
Afferi et al. (2020) [26] |
Y |
Y |
U |
N |
N |
N |
U |
Y |
N |
U |
Y |
Moderate |
|
Megia Garcia et al. (2020) [19] |
Y |
U |
N |
Y |
Y |
U |
U |
Y |
N |
U |
Y |
Low |
|
Pacheco et al. (2021) [35] |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
N |
Y |
Y |
High |
|
Taylor et al. (2021) [38] |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
N |
N |
Y |
Moderate |
|
Parittotokkaporn et al. (2021) [36] |
Y |
Y |
U |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
High |
|
Yang et al. (2022) [40] |
Y |
Y |
U |
Y |
Y |
U |
U |
Y |
Y |
Y |
N |
Low |
|
Massey et al. (2022) [32] |
Y |
Y |
N |
Y |
Y |
N |
U |
Y |
N |
Y |
Y |
Low |
|
Stampas et al. (2022) [37] |
Y |
N |
N |
Y |
Y |
Y |
U |
Y |
N |
N |
N |
Low |
|
* JBI critical appraisal checklist questions: Q1. Is the review question clearly and explicitly stated? Q2. Were the inclusion criteria appropriate for the review question? Q3. Was the search strategy appropriate? Q4. Were the sources and resources used to search for studies adequate? Q5. Were the criteria for appraising studies appropriate? Q6. Was critical appraisal conducted by two or more reviewers independently? Q7. Were there methods to minimize errors in data extraction? Q8. Were the methods used to combine studies appropriate? Q9. Was the likelihood of publication bias assessed? Q10. Were recommendations for policy and/or practice supported by the reported data? Q11. Were the specific directives for new research appropriate? **Abbreviations: Q: Question Y: Yes N: No U: Unclear |
||||||||||||
Discussion
The main output of the current umbrella reviews is a systematic and uniform assessment of all the evidence, available through SRs and MAs, on the application of TENS in the rehabilitation of SCI.
The rehabilitation procedure for SCI patients must consider various aspects of the disease that reflect the diverse needs of patients. For instance, age and condition, mechanism of injury, spinal cord lesion site and extent, etc. must all be taken into account when strategizing the rehabilitation plans [8]. In addition to physical rehabilitation, several studies discuss the importance of psychological rehabilitation approaches which have secondary effects on health, social participation, and QoL [51-53]. Use of electrical neuromodulation for rehabilitative purposes in SCI individuals has been the subject of many studies. James et al. [54] discussed the use of neuromodulation techniques to activate isolated neuronal circuitry below the injury site. This approach aims to compensate for the loss of supraspinal input and facilitate the transmission of sensory information for processing by higher centers. They also highlighted the range of approaches in neuromodulation for restoring function after spinal cord injury, including electrical stimulation of peripheral muscles, spinal cord, and brain. Gerasimenko et al. [55] described a novel technique of TSCS that neuromodulates the physiological state of the injured spinal cord. In those with motor-incomplete SCI, this method has demonstrated potential in regulating central excitability during voluntary treadmill stepping. Furthermore, Hofstoetter et al. [56] presented initial data on the use of non-invasive TSCS to augment voluntary locomotor activity in motor-incomplete SCI individuals. The study demonstrated the potential of TSCS in modulating the central state of excitability during voluntary stepping.
The promising effects of electrical stimulation in the context of SCI can be investigated in the context of both the intervention and the condition. To begin with, there is evidence of persistent supraspinal connections below the damage site after SCI. This indicates that the sub-lesioned neuronal circuitry continues to receive supraspinal inputs, but these residual inputs are inadequate to induce function. Hence, due to this preserved connection, it is possible that it is affected by external stimulation [57]. Furthermore, electrical stimulation can also affect the neuronal regeneration post-SCI [58,59]. For instance, in a study by Singh et al. [60] low-frequency electrical stimulation improved axon regeneration and recovery of motor and sensory function following sciatic nerve damage.
It is noteworthy to mention that this rehabilitative strategy comes with several limitations. First, while electrical stimulation can activate isolated neuronal circuitry below the injury site, it may not be able to precisely target specific muscles or movements. This lack of specificity can reduce the efficacy of electrical stimulation in obtaining intended functional results [54]. Another limitation is that the effectiveness of this modality can vary widely among the patient based on individual factors such as the severity and location of the injury. As a result, it is difficult to anticipate and maximize the consequences of electrical stimulation therapy. Also, while some studies have shown positive effects of electrical stimulation on voluntary movement and standing in individuals with SCI [61], others have found limited efficacy in preventing bone loss or improving bone density [62]. Therefore, further research is needed to better understand the mechanisms underlying the effects of electrical stimulation and to optimize its long-term benefits.
Conclusion
Rehabilitation plays a vital role in the recovery process following injury. By maximizing function and minimizing secondary problems, it promotes patients' independence and improves their overall quality of life. According to current available SRs, TENS holds promise for the rehabilitation of different SCI-associated conditions, including spasticity, pain management, and NLUTD. However, This conclusion is primarily based on low- to moderate-quality SRs. In addition, the application of TENS in SCI routine rehabilitation is bound to several limitations that can be addressed by future research, including lack of specificity, variability in individual responses, limited understanding of long-term effects, and practical challenges of implementation.
Acknowledgements
We would like to thank the Clinical Research Development Unit of Tabriz Valiasr Hospital, Tabriz University of Medical Sciences, Tabriz, Iran for their assistance in this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Deputy for Research of Tabriz University of Medical Sciences (Number: 72628).
Research Involving Human Participants and/or Animals
All the material used in the conduction of this study was already published and no direct human or animal were included.
Data Availability Statement
The data that support the findings of this study is retrieved from previously published materials and available from the corresponding author upon reasonable request.
Ethics of Approval Statement
The protocol of this study was approved by the Tabriz University of Medical Sciences (Code: 72628)
Patient Consent Statement
All the material used in the conduction of this study was already published and no direct human or animal were included.
Permission to Reproduce Material from Other Sources
All the material used in the conduction of this study was already published.
Clinical Trial Registration
This study followed an umbrella review methodology; hence, this is not applicable.
Author Contributions
F.T: Conceptualization, Data Curation, Funding Acquisition, Investigation, Methodology, Project Administration, Visualization, Writing, Original Draft Preparation, Review & Editing.
S. GH: Investigation, Data Curation, Visualization, Writing.
S. S: Investigation, Visualization, Validation, Writing.
A. RM: Conceptualization, Investigation, Methodology, Project Administration, Supervision, Visualization, Writing: Original Draft Preparation, Review & Editing.
References
2. Ehrmann C, Mahmoudi SM, Prodinger B, Kiekens C, Ertzgaard P. Impact of spasticity on functioning in spinal cord injury: an application of graphical modelling. J Rehabil Med. 2020 Mar 31;52(3):jrm00037.
3. Ong B, Wilson JR, Henzel MK. Management of the Patient with Chronic Spinal Cord Injury. Med Clin North Am. 2020 Mar;104(2):263-78.
4. Hall OT, McGrath RP, Peterson MD, Chadd EH, DeVivo MJ, Heinemann AW, et al. The Burden of Traumatic Spinal Cord Injury in the United States: Disability-Adjusted Life Years. Arch Phys Med Rehabil. 2019 Jan;100(1):95-100.
5. Middleton JW, Dayton A, Walsh J, Rutkowski SB, Leong G, Duong S. Life expectancy after spinal cord injury: a 50-year study. Spinal Cord. 2012 Nov;50(11):803-11.
6. Barclay L, Callaway L, McDonald R, Farnworth L, Brown T, Broom L. Time use following spinal cord injury: an examination of the literature. British Journal of Occupational Therapy. 2011 Dec;74(12):573-80.
7. Stevens SL, Caputo JL, Fuller DK, Morgan DW. Physical activity and quality of life in adults with spinal cord injury. J Spinal Cord Med. 2008;31(4):373-8.
8. Nas K, Yazmalar L, Şah V, Aydın A, Öneş K. Rehabilitation of spinal cord injuries. World J Orthop. 2015 Jan 18;6(1):8-16.
9. Tahmasbi F, Ghaderpanah R, Sadrian S, Heris RM, Salehi-Pourmehr H. Effects of transcutaneous electrical nerve stimulation (TENS) on chronic pain in older adults: A systematic review and meta-analysis. Current Physical Medicine and Rehabilitation Reports. 2023 Jun;11(2):242-53.
10. Tahmasbi F, Hosseini S, Hajebrahimi S, Heris RM, Salehi-Pourmehr H. Efficacy of Tibial Nerve Stimulation in Neurogenic Lower Urinary Tract Dysfunction Among Patients with Multiple Sclerosis: A Systematic Review and Meta-analysis. Urol Res Pract. 2023 Mar;49(2):100-11.
11. Tahmasbi F, Mosaddeghi-Heris R, Soleimanzadeh F, Ghaderpanah R, Sadrian S, Hajebrahimi S, et al. Effects of Posterior Tibial Nerve Stimulation on Fecal Incontinence: An Umbrella Review. Neuromodulation. 2024 Feb;27(2):229-242.
12. Tahmasbi F, Salehi-Pourmehr H, Naseri A, Ghaderi S, Javadi-Farid F, Hajebrahimi S, et al. Effects of posterior tibial nerve stimulation (PTNS) on lower urinary tract dysfunction: An umbrella review. Neurourol Urodyn. 2024 Feb;43(2):494-515.
13. Tahmasbi F, Sanaie S, Eslami S, Rahimi-Mamaghani A. Application of Transcutaneous Electrical Acupoint Stimulation (TEAS) for Management of Postoperative Pain After Gynecological Surgeries: A Meta-Analysis. Pain Manag Nurs. 2025 Feb;26(1):111-124.
14. Tahmasbi F, Sanaie S, Salehi-Pourmehr H, Ghaderi S, Rahimi-Mamaghani A. The role of transcutaneous electrical nerve stimulation (TENS) in rehabilitation of cerebral palsy: a systematic review. Dev Neurorehabil. 2025 Jan;28(1):52-61.
15. American Physical Therapy Association. Guide to physical therapist practice. Phys Ther. 2001;81:9-746.
16. Johnson MI, Bjordal JM. Transcutaneous electrical nerve stimulation for the management of painful conditions: focus on neuropathic pain. Expert Review of Neurotherapeutics. 2011 May 1;11(5):735-53.
17. Bi X, Lv H, Chen BL, Li X, Wang XQ. Effects of transcutaneous electrical nerve stimulation on pain in patients with spinal cord injury: a randomized controlled trial. J Phys Ther Sci. 2015 Jan;27(1):23-5.
18. Zeb A, Arsh A, Bahadur S, Ilyas SM. Effectiveness of transcutaneous electrical nerve stimulation in management of neuropathic pain in patients with post traumatic incomplete spinal cord injuries. Pak J Med Sci. 2018 Sep-Oct;34(5):1177-80.
19. Megía García A, Serrano-Muñoz D, Taylor J, Avendaño-Coy J, Gómez-Soriano J. Transcutaneous Spinal Cord Stimulation and Motor Rehabilitation in Spinal Cord Injury: A Systematic Review. Neurorehabil Neural Repair. 2020 Jan;34(1):3-12.
20. Aydin G, Tomruk S, Keleş I, Demir SO, Orkun S. Transcutaneous electrical nerve stimulation versus baclofen in spasticity: clinical and electrophysiologic comparison. Am J Phys Med Rehabil. 2005 Aug;84(8):584-92.
21. Ping Ho Chung B, Kam Kwan Cheng B. Immediate effect of transcutaneous electrical nerve stimulation on spasticity in patients with spinal cord injury. Clin Rehabil. 2010 Mar;24(3):202-10.
22. Nishi Y, Ikuno K, Minamikawa Y, Igawa Y, Osumi M, Morioka S. A novel form of transcutaneous electrical nerve stimulation for the reduction of dysesthesias caused by spinal nerve dysfunction: A case series. Front Hum Neurosci. 2022 Aug 24;16:937319.
23. Samejima S, Shackleton C, McCracken L, Malik RN, Miller T, Kavanagh A, et al. Effects of non-invasive spinal cord stimulation on lower urinary tract, bowel, and sexual functions in individuals with chronic motor-complete spinal cord injury: Protocol for a pilot clinical trial. PLoS One. 2022 Dec 13;17(12):e0278425.
24. Hunt H, Pollock A, Campbell P, Estcourt L, Brunton G. An introduction to overviews of reviews: planning a relevant research question and objective for an overview. Syst Rev. 2018 Mar 1;7(1):39.
25. Aromataris E, Fernandez R, Godfrey CM, Holly C, Khalil H, Tungpunkom P. Summarizing systematic reviews: methodological development, conduct and reporting of an umbrella review approach. Int J Evid Based Healthc. 2015 Sep;13(3):132-40.
26. Afferi L, Pannek J, Louis Burnett A, Razaname C, Tzanoulinou S, Bobela W, et al. Performance and safety of treatment options for erectile dysfunction in patients with spinal cord injury: A review of the literature. Andrology. 2020 Nov;8(6):1660-73.
27. Fernández-Tenorio E, Serrano-Muñoz D, Avendaño-Coy J, Gómez-Soriano J.Transcutaneous electrical nerve stimulation for spasticity: A systematic review. Neurologia (Engl Ed). 2019 Sep;34(7):451-60.
28. Garcia MAC, Vargas CD. Is somatosensory electrical stimulation effective in relieving spasticity? A systematic review. J Musculoskelet Neuronal Interact. 2019 Sep 1;19(3):317-25.
29. Gross T, Schneider MP, Bachmann LM, Blok BF, Groen J, Hoen LA, et al. Transcutaneous Electrical Nerve Stimulation for Treating Neurogenic Lower Urinary Tract Dysfunction: A Systematic Review. Eur Urol. 2016 Jun;69(6):1102-11.
30. Harvey LA, Glinsky JV, Bowden JL. The effectiveness of 22 commonly administered physiotherapy interventions for people with spinal cord injury: a systematic review. Spinal Cord. 2016 Nov;54(11):914-23.
31. Lu X, Battistuzzo CR, Zoghi M, Galea MP. Effects of training on upper limb function after cervical spinal cord injury: a systematic review. Clin Rehabil. 2015 Jan;29(1):3-13.
32. Massey S, Vanhoestenberghe A, Duffell L. Neurophysiological and clinical outcome measures of the impact of electrical stimulation on spasticity in spinal cord injury: Systematic review and meta-analysis. Front Rehabil Sci. 2022 Dec 16;3:1058663.
33. Mills PB, Dossa F. Transcutaneous Electrical Nerve Stimulation for Management of Limb Spasticity: A Systematic Review. Am J Phys Med Rehabil. 2016 Apr;95(4):309-18.
34. Moreno-Duarte I, Morse LR, Alam M, Bikson M, Zafonte R, Fregni F. Targeted therapies using electrical and magnetic neural stimulation for the treatment of chronic pain in spinal cord injury. Neuroimage. 2014 Jan 15;85 Pt 3:1003-13.
35. Pacheco N, Mollayeva S, Jacob B, Colantonio A, Mollayeva T. Interventions and cognitive functioning in adults with traumatic spinal cord injuries: a systematic review and meta-analysis. Disabil Rehabil. 2021 Apr;43(7):903-19.
36. Parittotokkaporn S, Varghese C, O'Grady G, Lawrence A, Svirskis D, O'Carroll SJ. Transcutaneous Electrical Stimulation for Neurogenic Bladder Dysfunction Following Spinal Cord Injury: Meta-Analysis of Randomized Controlled Trials. Neuromodulation. 2021 Oct;24(7):1237-46.
37. Stampas A, Hook M, Korupolu R, Jethani L, Kaner MT, Pemberton E, et al. Evidence of treating spasticity before it develops: a systematic review of spasticity outcomes in acute spinal cord injury interventional trials. Ther Adv Neurol Disord. 2022 Feb 17;15:17562864211070657.
38. Taylor C, McHugh C, Mockler D, Minogue C, Reilly RB, Fleming N. Transcutaneous spinal cord stimulation and motor responses in individuals with spinal cord injury: A methodological review. PLoS One. 2021 Nov 18;16(11):e0260166.
39. Thomaz SR, Cipriano G Jr, Formiga MF, Fachin-Martins E, Cipriano GFB, Martins WR, et al. Effect of electrical stimulation on muscle atrophy and spasticity in patients with spinal cord injury - a systematic review with meta-analysis. Spinal Cord. 2019 Apr;57(4):258-66.
40. Yang Y, Tang Y, Qin H, Xu J. Efficacy of transcutaneous electrical nerve stimulation in people with pain after spinal cord injury: a meta-analysis. Spinal Cord. 2022 May;60(5):375-81.
41. Oo WM. Efficacy of addition of transcutaneous electrical nerve stimulation to standardized physical therapy in subacute spinal spasticity: a randomized controlled trial. Arch Phys Med Rehabil. 2014 Nov;95(11):2013-20.
42. Norrbrink C. Transcutaneous electrical nerve stimulation for treatment of spinal cord injury neuropathic pain. J Rehabil Res Dev. 2009;46(1):85-93.
43. Celik EC, Erhan B, Gunduz B, Lakse E. The effect of low-frequency TENS in the treatment of neuropathic pain in patients with spinal cord injury. Spinal Cord. 2013 Apr;51(4):334-7.
44. Perdan J, Kamnik R, Ceru B, Bajd T, Savrin R, Jelenc J, et al. Comparison of four evaluation approaches in transcutaneous electrical nerve stimulation treatment in two incomplete tetraplegic subjects. Neuromodulation. 2010 Jul;13(3):238-45.
45. Freyvert Y, Yong NA, Morikawa E, Zdunowski S, Sarino ME, Gerasimenko Y, et al. Engaging cervical spinal circuitry with non-invasive spinal stimulation and buspirone to restore hand function in chronic motor complete patients. Sci Rep. 2018 Oct 19;8(1):15546.
46. Hartkopp A, Harridge SD, Mizuno M, Ratkevicius A, Quistorff B, Kjaer M, et al. Effect of training on contractile and metabolic properties of wrist extensors in spinal cord-injured individuals. Muscle Nerve. 2003 Jan;27(1):72-80.
47. Glinsky J, Harvey L, van Es P, Chee S, Gandevia SC. The addition of electrical stimulation to progressive resistance training does not enhance the wrist strength of people with tetraplegia: a randomized controlled trial. Clin Rehabil. 2009 Aug;23(8):696-704.
48. Meyer C, Hofstoetter US, Hubli M, Hassani RH, Rinaldo C, Curt A, et al. Immediate Effects of Transcutaneous Spinal Cord Stimulation on Motor Function in Chronic, Sensorimotor Incomplete Spinal Cord Injury. J Clin Med. 2020 Nov 2;9(11):3541.
49. Sayenko DG, Rath M, Ferguson AR, Burdick JW, Havton LA, Edgerton VR, et al. Self-Assisted Standing Enabled by Non-Invasive Spinal Stimulation after Spinal Cord Injury. J Neurotrauma. 2019 May 1;36(9):1435-50.
50. Shendy WS, El Semary MM, Battecha KH, Abdel-Azim MS, Mourad HS, El Gohary AM. Efficacy of transcutaneous electrical nerve stimulation versus biofeedback training on bladder and erectile dysfunction in patients with spinal cord injury. Egyptian Journal of Neurology, Psychiatry & Neurosurgery. 2015 Jul 1;52(3):194-200.
51. Nooijen CF, Stam HJ, Sluis T, Valent L, Twisk J, van den Berg-Emons RJ. A behavioral intervention promoting physical activity in people with subacute spinal cord injury: secondary effects on health, social participation and quality of life. Clin Rehabil. 2017 Jun;31(6):772-80.
52. Ottomanelli L, Lind L. Review of critical factors related to employment after spinal cord injury: implications for research and vocational services. J Spinal Cord Med. 2009;32(5):503-31.
53. Price P, Stephenson S, Krantz L, Ward K. Beyond my front door: The occupational and social participation of adults with spinal cord injury. OTJR: Occupation, Participation and Health. 2011 Mar;31(2):81-8.
54. James ND, McMahon SB, Field-Fote EC, Bradbury EJ. Neuromodulation in the restoration of function after spinal cord injury. Lancet Neurol. 2018 Oct;17(10):905-17.
55. Gerasimenko Y, Gorodnichev R, Moshonkina T, Sayenko D, Gad P, Reggie Edgerton V. Transcutaneous electrical spinal-cord stimulation in humans. Ann Phys Rehabil Med. 2015 Sep;58(4):225-31.
56. Hofstoetter US, Krenn M, Danner SM, Hofer C, Kern H, McKay WB, et al. Augmentation of Voluntary Locomotor Activity by Transcutaneous Spinal Cord Stimulation in Motor-Incomplete Spinal Cord-Injured Individuals. Artif Organs. 2015 Oct;39(10):E176-86.
57. Edgerton VR, Tillakaratne NJ, Bigbee AJ, de Leon RD, Roy RR. Plasticity of the spinal neural circuitry after injury. Annu Rev Neurosci. 2004;27:145-67.
58. Ahlborn P, Schachner M, Irintchev A. One hour electrical stimulation accelerates functional recovery after femoral nerve repair. Exp Neurol. 2007 Nov;208(1):137-44.
59. Asensio-Pinilla E, Udina E, Jaramillo J, Navarro X. Electrical stimulation combined with exercise increase axonal regeneration after peripheral nerve injury. Exp Neurol. 2009 Sep;219(1):258-65.
60. Singh B. Impact and Mechanisms of Extracellular Electrical Stimulation on Peripheral Nerve Regeneration. Doctoral thesis, University of Calgary, Calgary, Canada; 2013.
61. Harkema S, Gerasimenko Y, Hodes J, Burdick J, Angeli C, Chen Y, et al. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. Lancet. 2011 Jun 4;377(9781):1938-47.
62. Clark JM, Jelbart M, Rischbieth H, Strayer J, Chatterton B, Schultz C, et al. Physiological effects of lower extremity functional electrical stimulation in early spinal cord injury: lack of efficacy to prevent bone loss. Spinal Cord. 2007 Jan;45(1):78-85.