Abstract
Autism spectrum disorder is a multifactorial neurodevelopmental disorder with increasing prevalence worldwide. Given the heterogeneity of autism, it is highly unlikely to have a single effective therapy for autism. Adversities associated with current pharma-therapies in treating autism have prompted the emergence of alternative therapies including variety of behavioral and music interventions. Autistic children, who tend to show a strong preference for music, makes music therapy a promising intervention for autism. Music interventions have shown improved mental and physical health across multiple domains including autism. Previous clinical trials of music therapy versus traditional therapy for autistic children have shown encouraging but mixed results. In that regard, Indian classical music is postulated to exert enhanced benefits due to its melodic uniqueness. Current randomized controlled pilot study evaluated enhanced benefits of Indian classical music-Raga co-treatment added to the conventional standard care in 5-12 years old verbal autistic children. Participants were randomly assigned either to comprehensive standard care (Std) comprised of Applied Behavior Analysis (ABA) and occupational Sensory Integration Therapy (SIT), or to Raga-Music therapy added to the standard care. The comparative effectiveness of Raga-Music therapy add-on was evaluated over 8-weeks of treatment duration, as assessed by primary and secondary outcome measures. The primary outcome measure included the Autism Treatment Evaluation Checklist (ATEC) assessment that evaluated the progression of autism, while the secondary outcome measures included sensory processing assessment using Sensory Profile 2 (SP2) followed by Canadian Occupational Performance Measure (COPM) with satisfaction, before and after the treatment. Raga-Music co-treatment was found to accentuate the benefits of standard treatment in reducing the progression of autism along with improvement in sensory profile of verbal autistic children. Current study supports the notion of Raga-Music as an effective add-on for early intervention autism healthcare program while warranting further investigation.
Keywords
Autism spectrum disorder, Music therapy, Indian classical music, Raga, Emotional upliftment, Sensory profile, ABA, SP2, ATEC, COPM
Introduction
Autism spectrum disorder (ASD) or autism is a heterogeneous neurodevelopmental disorder encompassing a group of neurobehavioral conditions including autism, Asperger’s disorder, and pervasive developmental disorder [1,2], afflicting 1 in 31 children in United States [3] and 1 in 100 children worldwide [4]. Currently, there is no cure for ASD [5]. Ongoing pharmacological treatments of ASD include the use of psychostimulants, atypical antipsychotics, anti-depressants, and α-2 adrenergic receptor agonists, which provide only partial symptomatic relief and are associated with adverse side effects [6–10]. In that regard, non-pharmacological treatments with none/least adversities [11], such as cognitive behavioral therapy (CBT) [12], applied behavioral therapy (ABA) [13], social behavioral therapy (SBT) [14], and creative art [15–17] or music therapy (MT) [18–21] are increasingly being considered as effective therapeutic alternatives. The “spectrum” of autism embraces both challenges and strengths [22]. The challenges of ASD are characterized by persistent deficits in two core areas of functioning i.e. Social communication and interaction and restricted/repetitive patterns of behavior [23–25], along with other co-morbid psychotic conditions such as anxiety, depression, attention deficit, epilepsy, etc. [26,27]. Despite difficulties in such psychobehavioral domains, autistic children show strong visual/auditory learning and remarkable ability to excel in math, art, craft, and music [28–30]. There is a growing recognition in using music as a potential therapeutic intervention to improve psychological, motor and behavioral dysfunctions in people with neurological disorders including ASD [20,31–35]. Regardless of an inability to interpret other people’s nonverbal, facial, and bodily expressions of emotion(s), a strong preference for music is observed in autism [36–38], suggesting that music can be exploited as an effective alternative for treating ASD [39]. Early evidence suggests remarkable improvements across many clinical domains of ASD, paralleling with structural and functional corrections in the key brain-areas following music exposure [33,40,41]. One study showed statistically significant improvement in emotional responses of autistic children to musical stimuli [42]. Another report showed that compared to non-music controls, 8-12 weeks of music intervention improved parent-reported social communication and restored brain activity, as indicated by functional MRI after music intervention [41,43]. The social skills were significantly improved after improvisational music therapy in autistic children [44,45]. One study showed that listening to both “Happy” and “Sad” music activated cortical and subcortical brain regions involved in emotional processing and reward system, although “Happy” music effects were more pronounced than that of “Sad” music [46]. Effectiveness of music therapy may be attributed to its distinct ability to modify both the structural and functional brain connectivity via neuroplastic and neurophysiological mechanisms [47–49]. Moreover, the potential of music in reducing stress hormones while simultaneously elevating hormones involved in emotional/reward/cognitive processing and neuroplasticity, makes music-an effective choice [50–54]. Music affects functional brain connectivity and therefore is effective in treating neurological disorders [55–58] including ASD [28,41,59]. Music has a distinct ability to target ASD-specific core disabilities aimed at restructuring auditory-motor-cognitive connectivity leading to improvements in social communication, emotional and motor functions [37,41,42,60]. In that regard, Indian Classical Music (ICM) (Raga) may exert added advantage due to its structural uniqueness, defined rhythmicity and melodic framework of Raga, known to induce specific emotions/moods in a circadian-specific manner [61–64]. Studies have shown the ability of specific Ragas to elicit specific “Rasa” (emotion) aligning with corresponding brain physiology [65–69]. However, there are sparse reports showing Raga effects in autism except few [41,70]. This randomized controlled study explored the effects of Raga in treating autistic children. Current pilot study evaluated therapeutic feasibility of Indian Classical Music-Raga intervention (https://youtu.be/viL-8iubNpE; Owner, Mahesh Kale; One of the Authors), edited to remove extra-music components defined as “MUSIC”, in the verbal group autistic children between 5–12 years of age [71,72], at the Early Intervention Child Development Center, RUGRATS Cocoon, Ahmedabad, Gujarat, India. Since this is a preliminary pilot study aimed at assessing only the feasibility of MUSIC, and not a true 3-arm randomized controlled trial (RCT), it has not been registered in a clinical trials registry (ClinicalTrials.gov or CTRI). The primary objective of our study was to determine comparative effectiveness of MUSIC intervention in attenuating autism-progression in autistic children, as evaluated by the standardized Autism Treatment Evaluation Checklist (ATEC) assessment [73,74]. Since the abnormal sensory processing constitutes a characteristic feature of ASD [75–78], the secondary objective of our study was to examine the changes in sensory processing patterns in autistic children, before and after MUSIC intervention, using Winnie Dunn’s standardized sensory profile 2 (SP2) assessment [79–82], followed by Canadian Occupational Performance Measure (COPM) reflecting patients’ occupational functional improvement index and parent satisfaction [83,84].
Methods
Participants
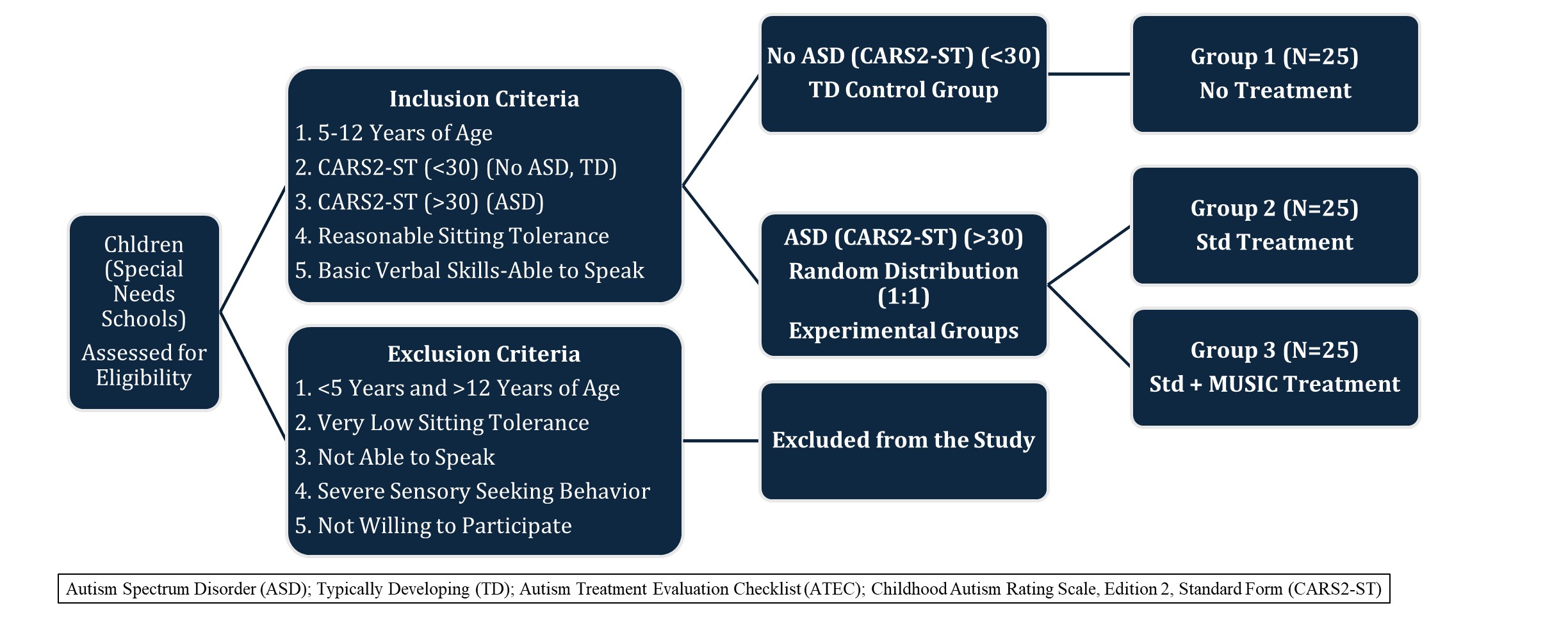
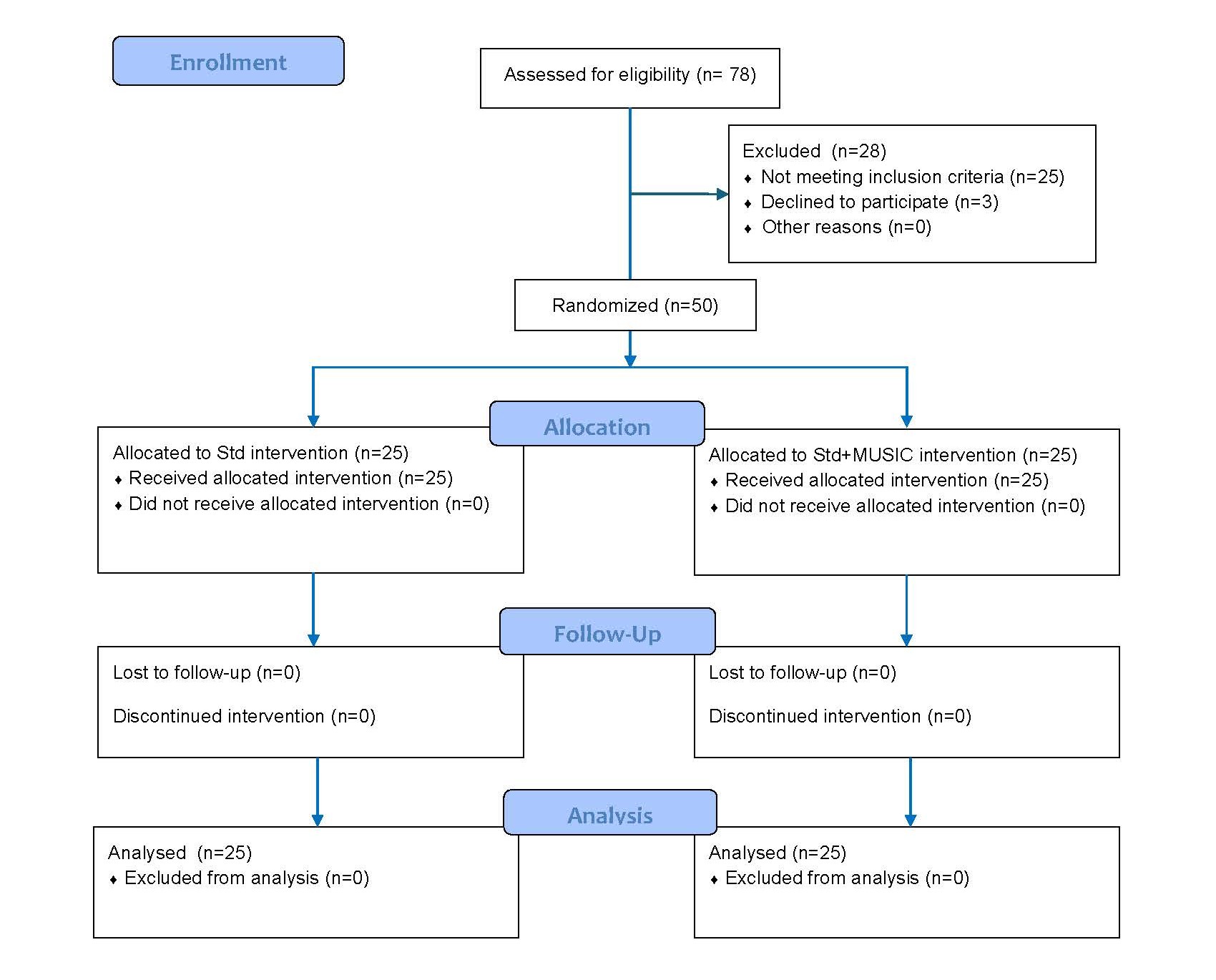
A total of 75 participants (5–12 years of age) were recruited who met the eligibility (inclusion criteria) after diagnosed for the presence of ASD using CARS2-ST scale [85,86] and randomly assigned to experimental groups (Figures 1 and 2). There was no attrition from the recruited participants.
Diagnosis of autism spectrum disorder-childhood autism rating scale, Std. 2nd Ed. (CARS2ST)
The childhood autism rating scale (CARS) is the most widely accepted diagnostic tool for diagnosing autism, originally developed by Schopler et al. [87,88], and later refined as CARS2, second edition with two versions namely CARS2-ST and CARS2-HF [89]. CARS2-ST, although similar to the original version, includes some modification(s) designed for children below 6 years of age, and a high function version (CARS2-HF) for those aged 6 years and older [90–92]. The diagnostic validity of CARS2-ST is high across Diagnostic and Statistical Manual of Mental Disorders (DSM)-IV/V criteria for diagnosing autism [90,93]. The CARS2-ST consists of 15-item questionnaire and is evaluated by a clinical expert based on personal interviews with the primary caregiver and direct observation of children [86]. Each question is rated on a scale of 1 (Normal at corresponding age) to 4 (Severely abnormal at corresponding age). Individual item scores are added to obtain an aggregate score ranging between 15–60 for each subject [85,86,92]. CARS2-ST score <30 indicates non-autism-Normal, the score >30 between 30–36 indicates mild-moderate ASD, and the score >36 indicates severe ASD [86,92,94–96]. The CARS2-ST assessment was conducted by a licensed and trained clinical professional and confirmed by the occupational therapist having many years of experience treating autistic children at the Early Intervention Child Development Center, RUGRATS Cocoon, Ahmedabad, Gujarat, India. All data were accurately recorded including age, demographics, date of test administration, CARS2-ST scores, and used for diagnostic identification of the presence or absence of ASD.
Inclusion/exclusion criteria
Inclusion criteria:
- Age between 5 - 12 years
- ASD score < 30 on CARS-2-ST scale—as Typically Developing (TD) Normal Control subjects
- ASD score > 30 on CARS-2-ST scale—as ASD experimental subjects
- Reasonable sitting tolerance (when evaluated during initial assessment)
- Basic verbal skills - able to speak for needs
Exclusion criteria
- Younger than 5 years and older than 12 years of age
- Not able to speak
- Very low sitting tolerance
- Severe sensory seeking behavior
- Not willing to participate in the study
Figure 1. Distribution of participants.
Recruitment and randomization
Based on the inclusion and exclusion criteria, eligible subjects between 5–12 years of age were recruited in the study and distributed among experimental groups (Figures 1 and 2). Eligible children with CARS2-ST score <30 were identified as non-autistic who served as typically developing (TD) normal controls (Group 1). Since this is only a pilot feasibility study to see if Raga may have any effect at all, TD group has been included as a non-randomized observational reference group. Eligible children with CARS2-ST score >30 were identified as ASD. The ASD subjects were randomized as follows. The randomization and recruitment was conducted by the healthcare professional staff at the Early Intervention Child Development Center, RUGRATS Cocoon, Ahmedabad, Gujarat, India. They enrolled participants by creating an unpredictable random list of participants to be assigned to different groups using a “Coin-Toss” simple randomization, while hiding the sequence of assignment from the enrolling participants. As mentioned earlier, a non-randomized TD group was included as an observational reference group (Group 1). Thus, the participants were randomly assigned to two experimental groups (1:1) based on the treatment regimen. ASD children to be treated only with standard therapy (Std) (Group 2) or to be treated with Std plus MUSIC therapy (Group 3) (Figures 1 and 2).
Group 1 (TD): Typically developing (TD) Non-ASD normal children, <30 CARS2-ST, (N=25)
Group 2 (ASD-Std): ASD children, CARS2-ST >30, treated with standard (Std) therapy only (N=25)
Group 3 (ASD-Std+MUSIC): ASD children, CARS2-ST >30, treated with Std plus MUSIC therapy (N=25)
MUSIC is defined as the Raga intervention (https://youtu.be/viL-8iubNpE (Owner, Mahesh Kale, One of the Authors)), edited to remove extra-music components (MUSIC). Although it appears unusual, in fact, there was no attrition from the recruited subjects for a short duration of 8 weeks. All assessments were conducted at pre- and post-treatment time-points. The TD controls, although not treated, underwent ATEC evaluation at the matching pre- and post-assessment time-points parallel to the experimental groups.
Figure 2. CONSORT 2010 flow diagram.
Sample size justification
The sample size of N=25 per group was determined based on the recommendation by the National Center for Complementary and Integrative Health (NCCIH), National Institute of Health (NIH), suggesting the sample size of N=25 per group to be practical for establishing feasibility rather than the consideration of statistical power [www.nccih.nih.gov]. Supporting reports in this regard confirmed that for the qualitative work (such as the current study) involving behavioral, emotional and psycho-social assessments, the sample size of N=30 or less is adequate to establish the feasibility [97–100]. A power analysis for sample-size estimation indicated that a range of 15–22 participants would be sufficient to detect a meaningful treatment-difference while testing feasibility [101]. A study aimed at evaluating feasibility and acceptability of music therapy used the sample size ranging between N=19–23 [98]. Another study that examined comparative effectiveness of music therapy involving pre- and post-test measurements used the sample size of N=20 [99]. With the recommended minimum sample size of N=12 to be used for the pilot trials as a rule of thumb [102], a sample size range of N=20–25 may be adequate to obtain meaningful group differences [103]. Since the main goal of current pilot study is to establish the platform to predict meaningful outcomes of MUSIC therapy to establish the feasibility for larger studies, a sample size of N=25 per group will be adequate without requiring a need for large sample size [98-100,104]. Thus, a sample size of N=25 per group used in the current feasibility pilot study is well-justified to determine comparative effectiveness of MUSIC therapy.
Study design
This feasibility pilot study design was aimed at testing whether the MUSIC co-treatment will enhance the benefits of standard therapy and possibly delay/attenuate the progression of ASD in autistic children. The study was conducted in verbal group children between 5–12 years of age [71,72], recruited after confirmed diagnosis of ASD or no ASD, as determined by CARS2-ST assessment [85,86]. The established CARS2-ST scale defines the score below 30 as normal, the scores >30 (30–36) as mild-moderate ASD, and the scores >36 as severe ASD [86,92,94,95]. As mentioned earlier, Children with CARS2-ST scores <30 were classified as typically developing (TD) non-ASD normal controls while the children with the CARS2-ST scores >30 were classified as ASD experimental subjects. The non-ASD TD controls did not receive any treatment while the ASD experimental subjects received standard treatment with or without MUSIC co-treatment. The ASD experimental group was divided into two treatment groups with 1:1 allocation ratio, including an experimental group treated with only standard (No MUSIC) (Std) intervention, and another experimental group treated with Std intervention combined with MUSIC intervention including a 15-min daily session of MUSIC therapy for 8 weeks. The primary outcome included ATEC assessment to monitor the progression of autism [73,74], while the secondary outcome assessments included SP2 [80–82] and COPM [83,84] to evaluate changes in the sensory processing and occupational performance, before and after the interventions. All evaluations were performed at the treatment-start base level (Pre-test) and at the treatment-end (Post-test) time points. ATEC evaluation was performed in all three groups, while SP2 and COPM assessments were conducted in both experimental groups (Figure 3).
Figure 3. Treatment regimen and outcome evaluation timeline.
Ethical aspects
This randomized feasibility pilot study was conducted in accordance with the approval (HSN/SAC Code: 998346; Date of Approval: January 12, 2024) granted by CONSCIENCE Independent Ethics Committee (CIEC), Ahmedabad, Gujarat, India. CIEC operates in compliance with the Indian Council of Medical Research (ICMR) Guidelines for Biomedical Research on Human Subjects, and WMA Declaration of Helsinki, Brazil, Oct 2013, 21 CFR Part 56 and 21 CFR Part 50, in governance with Good Clinical Practice. The process of obtaining informed consent was conducted in accordance with the requirements of CIEC Ethics Committee guidelines. The signed “Informed Consent” from each child/parent was registered before recruiting patients in the study. They were educated about the study set-up (Purpose, Procedure/Methods, Duration of the Project; Non-invasive risk-free nature of MUSIC intervention). They were assured that the session(s) will cease immediately if their child becomes distressed or uncomfortable for any reason. Participants were guaranteed that they have freedom to withdraw from the study at any time without the loss of benefits they are entitled to. This approved feasibility pilot study protocol has been registered at the Data Governance and Compliance Infrastructure (DGCI) (ECR/233/Indt/GJ/2015/RR21); Department of Health and Human Services (DHHS) (IORG0008391); and Office of Human Research Protections (OHRP) (IRB00010059).
Interventions
This study included 2 types of interventions:
- Standard intervention (Std)
- Indian Classical Music-Raga (https://youtu.be/viL-8iubNpE) intervention (MUSIC)
Standard intervention
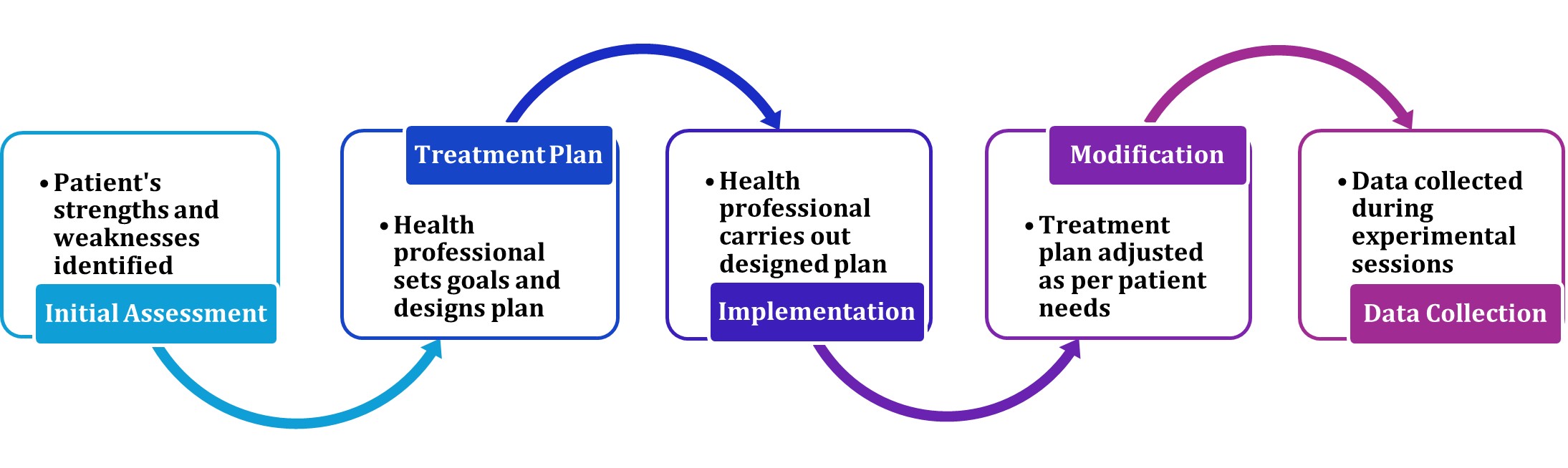
Standard intervention (Std) consisted of a comprehensive treatment plan including Applied behavioral Analysis (ABA) [13,105] combined with Occupational Sensory Integration Therapy (SIT) [106,107], addressing both the behavioral and sensory processing aspects [107,108] in treating verbal group autistic children. All interventions were delivered as “one-on-one” basis (Figure 4). Session compliance of all participants was confirmed.
Figure 4. Standard intervention flow chart.
Initial assessment
The initial assessment of participants randomized to the stated intervention(s) was carried out over an in person introductory ~1h session at the study site (Early Intervention Child Development Center, RUGRATS Cocoon, Ahmedabad, Gujarat, India), starting with an initial greeting-exchanges and welcoming. The comprehensive highlights of the project, including the purpose of the project, how the project to be conducted, what kind of treatments to be given (Std and MUSIC), duration of treatments, etc. all details were given. Then the designated healthcare professional discussed designing individualized occupational/behavioral treatment plan depending upon the weaknesses/strengths identified for each patient. Parents were assured about their child’s safety, comfort and freedom to withdraw from the study at any time for any reason. Following this introductory session, the signed “Informed Consent” was obtained from each child’s parent, after agreed for participating in the project.
Treatment plan, implementation/modification, and data collection
Applied behavior analysis (ABA) therapy (1h/Day, 5 Days/Week, 8 Weeks): Based on the observations made during Initial assessment, the therapist created a personalized ABA therapy-plan that aligned with the learner’s unique needs, by setting the goals to eliminate disruptive social behavior, such as throwing tantrums or inflicting self-injury, to encourage better, healthier social interactions.
The goals were modified depending on the age and ability of autistic children, along with individual factors:
- Learning abilities
- Social skills
- Self-care capabilities (e.g. bathing or brushing)
- Motor skills
- Communication skills
The parents/caregivers played a major role in ensuring the success of ABA therapy by actively enforcing desired behavior while the learners are outside of therapy-class. The therapist trained the parents/caregivers on how to respond to both desired and unacceptable social behavior. The therapist constantly evaluated the patient’s response and made changes as needed to ensure better outcomes.
Techniques used in implementing and modifying ABA
- Positive Reinforcement: This involved rewarding desired social behavior with favorite food right after a child exhibited the right behavior. This was to establish a positive correlation between the act and the reward, making them more likely to remember and repeat good behavior in the future.
- Negative Reinforcement: This was not intended to be confused with punishing for displaying inappropriate behavior. Instead, negative reinforcement involved taking away an object or activity after the instance involving undesired social behavior. Examples included—a child kicked his/her peer in class. To prevent such behavior, the child was engaged with his/her favorite activity and keep away from repeating bad behavior.
- Visual Cues: Both verbal prompts and visual gestures were effective at reinforcing the desired social behavior as long as they weren't accusatory or intimidating. Example included—directing the child to wash hands before meals or place his/her belongings neatly in a designated spot and repeating the directions frequently till they no longer need a cue/prompt.
- Verbal Cues: The therapist may teach the child with ASD to spell out certain words by singing and then use the same vocabulary to create entire sentences and stories.
- Analysis of Tasks: The instructor assigned tasks and observed the way they were completed to better analyze the child's abilities to create future tasks in line with their skills.
Sensory integration therapy (SIT) (45 min/Day, 5 Days/Week, 8 Weeks):
This therapy was aimed at providing controlled sensory experiences to help children better process and regulate their sensory input, involving a variety of play-based activities, deep pressure input, vestibular stimulation, and brushing protocols, all designed to address specific sensory processing needs.
Techniques used in implementing and modifying SIT
- Play-based activities: Play- as medium to engage children in activities that stimulated different sensory systems.
- Vestibular Stimulation: Activities like swinging, spinning, and balancing, which stimulated the vestibular system, responsible for balance and spatial awareness.
- Deep Pressure Input: Squeezing stress balls, using weighted blankets, applying gentle pressure can provide calming and grounding sensory experiences, improving body awareness and reducing anxiety.
- Tactile Play: Engaging with different textures, materials, to develop a more refined understanding of tactile sensations.
- Brushing Protocol: This technique involved using a soft brush to apply deep pressure to the skin, which can help regulate sensory input and improve tactile awareness.
- Proprioceptive Input: Activities like pushing, pulling, and engaging in resistive exercises can help improve body awareness and coordination by stimulating proprioceptors, which are located in muscles and joints.
- Visual and Auditory Stimuli: Activities like visual tracking exercises, using colorful lights, or playing with music can help improve visual processing, attention, and auditory processing skills.
- Oral Motor Activities: Chewing, blowing bubbles, and other oral motor activities can help improve oral sensory processing and support speech and feeding skills.
- Environmental Modifications: Adjustments to the environment, such as reducing noise levels, minimizing visual clutter, or providing sensory breaks, creating a more supportive and less overwhelming environment.
ABA therapy was conducted 1h/day, combined with Occupational Sensory Integration Therapy (SIT) 45 min/day.
ABA and SIT Therapies together were targeted at:
- Developing behavioral skills while simultaneously decreasing the severity of autism
- Encouraging acceptable social behavior
- Increasing attention and concentration
- Reducing self-harming behavior or throwing tantrums
- Developing effective communication to better interact with peers and parents
The data were collected using Autism Treatment Evaluation Checklist (ATEC), Winnie Dunn’s Sensory Profile 2 (SP2), and Canadian Occupational Performance Measure (COPM) along with Satisfaction Scoring evaluations before (Pre-) and after (Post-) the treatment(s).
MUSIC Intervention
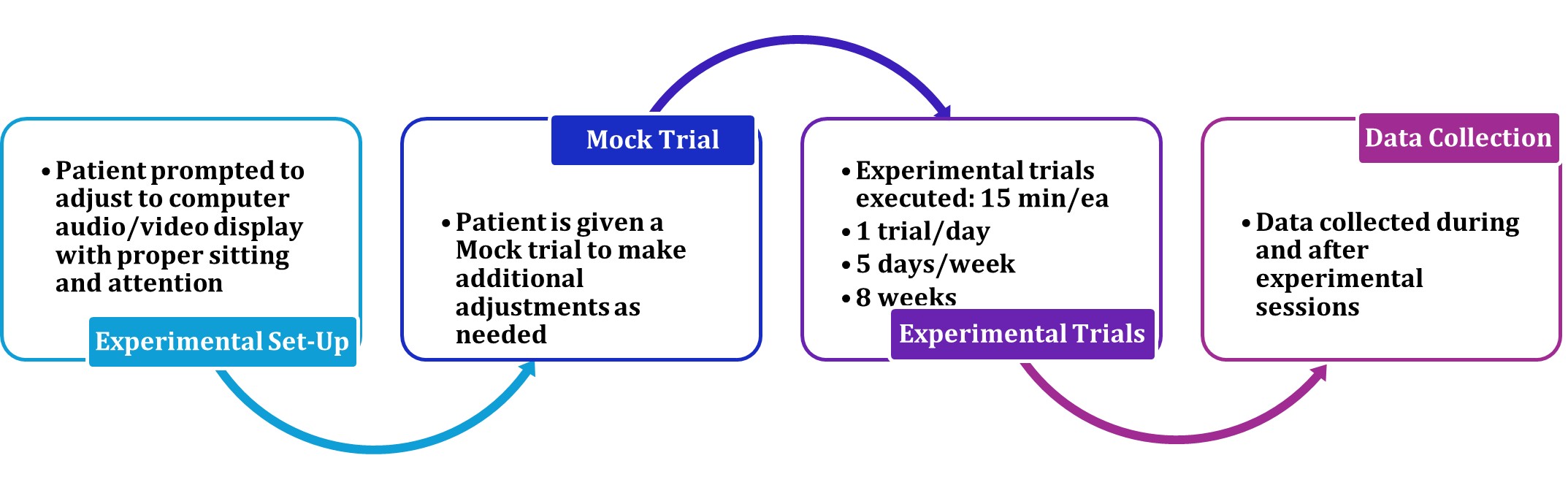
MUSIC Therapy (15 min/Day, 5 Days/Week, 8 Weeks): The MUSIC therapy was administered in the autistic children aged 5–12 years with one-on-one basis, at the Early Intervention Child Development Center, RUGRATS Cocoon, Ahmedabad, Gujarat, India, as outlined (Figure 5). Session compliance of all participants was confirmed.
Figure 5. MUSIC intervention flow chart.
Experimental set-up
The experimental set up consisted of a ~300 sq ft. isolated room within the facility, specifically dedicated to conduct MUSIC therapy, with the least sound disturbance, environmental controls of temperature and humidity, and optimum ambience. This room was large enough to house a dedicated computer-desk with comfortable chair set up, equipped with a large-screen computer monitor and a well-tested audio-visual system for an optimum display of MUSIC video, with plenty of walk-around free space. The room also contained sitting arrangements for the healthcare professional(s) in charge of conducting the MUSIC sessions.
Mock trial
A mock trial session was conducted prior to administering experimental sessions to ensure proper functioning of all technical devices to allow necessary adjustments such as computer-screen brightness, sharpness, audio-speakers, adjusting chair-computer height/distance, room environment and overall comfort adjustments, optimally ready to conduct experimental sessions.
Experimental trials
Each experimental session of 15 min duration was conducted by the licensed, well-trained and experienced professional using one on one settings. Each subject was stationed at designated computer-desk spot. Occasionally, the child was allowed to freely choose whether to stay on the carpeted floor or switch between chair and floor. The child was encouraged to communicate in one’s comfortable way including gestures, speech, hand-waving, using individual comfort level. Needed adjustments were made accordingly. All sessions were conducted in the same identical settings.
- Each 15 min Session; One Session/Day; Mon-Fri; 5 Sessions/Week, for 8 Weeks (Total 40 Sessions)
- Sat-Sun Week-end was used for “Missed” Session Make-Up, if any, in order to run parallel with the group
Data collection
The data were collected using Autism Treatment Evaluation Checklist (ATEC), Winnie Dunn’s Sensory Profile 2 Questionnaire and Canadian Occupational Performance Measure (COPM) Questionnaire along with Satisfaction Scoring before (Pre-) and after (Post-) the treatment. In addition to parent/care-giver reporting, the data for all participants was randomly re-assessed once/week by the healthcare professional in-charge at the facility.
Treatment fidelity
At every OT-SIT and MUSIC session(s), the healthcare professional(s) confirmed session compliance, and documented clinical notes including significant events, notable child behavior, confirmed appropriate administration of interventions including session-duration, child’s attention and/or intermittent participation such as singalong/gestures/hand-waving. All therapy and parent counselling sessions were videotaped to be re-assessed by independent raters and licensed occupational therapist, if needed, for additional confirmation.
Outcome measures
As mentioned earlier, the primary outcome measure included ATEC assessment [73,74], while the secondary outcome measures included SP2 [80–82], and COPM-Satisfaction [83,84] evaluations. All evaluations were performed at the treatment-start (Baseline/Pre-test) and at the treatment-end (Post-test) time-points. TD control group did not differ significantly between pre- and post-time points. TD controls, although untreated, were matched with the Pre- and Post-time points of experimental groups for ATEC assessments. ATEC evaluation was performed in all the groups, including non-ASD control group that did not receive any treatment, while SP2 and COPM-Satisfaction evaluations were conducted in experimental Groups, Group-2 that received only Std intervention and Group 3 that received Std plus MUSIC Co-treatment.
Primary outcome measure—autism treatment evaluation checklist (ATEC) assessment
The Autism Treatment Evaluation Checklist (ATEC) was originally developed by Rimland and Edelson to measure the severity of autism symptoms and to monitor or track changes over the duration of study/observation [109]. The primary use of ATEC evaluation is to measure changes in an individual's autism symptoms after interventions [74]. Researchers have used ATEC to measure the effectiveness of new treatments by measuring severity of ASD in response to treatment after comparing the baseline ATEC scores with the post-treatment ATEC scores [19,101,110]. ATEC is a one-page form designed to be completed by parents, teachers, or caretakers [73,74]. It consists of a total of 77 questionnaire items distributed in 4 subclasses representing 4 diagnostic domains: (i) Speech/Language Communication (14 items); (ii) Sociability (20 items); (iii) Sensory/ Cognitive Awareness (18 items); and (iv) Health/Physical/Behavior (25 items) [20,74]. Lower ATEC scores indicate lower ASD severity and efficient functioning while higher ATEC scores correlate with greater ASD severity [111].
Secondary outcome measure - winnie dunn sensory profile-2 (SP2) assessment
The Sensory Profile™ 2 (SP2) [112,113] is a standardized instrument to assess children’s sensory processing patterns which assesses the child’s response to sensory events throughout daily living situations. The SP2 has been widely used in many autism studies [75,79,106,114–116], and its reliability and validity has been very well established [82,117–119]. This instrument is based on Dunn’s four quadrant Sensory Processing Model (1997) [120] which theorizes the relationship between: (a) neurological construct (the functioning of a person’s nervous system) (neurological construct) and (b) self-regulatory strategies (behavioral construct)-the interaction of these neurological and behavioral constructs creates 4 basic patterns of sensory processing: (i) Seeking, (ii) Avoiding, (iii) Sensitivity, and (iv) Registration. These four quadrants of sensory responses are analyzed by SP2 in 9 sensory processing diagnostic domains: Auditory, Visual, Touch, Movement, Body Position, Oral, Conductual, Socio-emotional, and Attentional response(s) [121]. It comprises of 86 items which are scored on a 6-point scale (0-5): Not applicable (0), Seldom or never (1), Occasionally (2), Half of the times (3), Frequently (4), and Always or almost always (5). First, the raw scores are recorded for all 9 areas (Auditory, Visual, Touch, Movement, Body Position, Oral, Conductual, Socio-emotional, Attentional responses). These raw scores are then entered into the “Quadrant Grid”, appropriately distributed within the 4 basic sensory processing patterns (Seeking, Avoiding, Sensitivity, Registration), and total quadrant raw scores are obtained for each of the 4 sensory processing areas. These total quadrant raw scores are entered into the “Summary Score Sheet” to interpret the data according to the Quadrant Definitions. The Sensory Profile 2 (SP2) scores are interpreted by comparing a child's raw scores to a standard reference, which then categorizes their responses as "More than Others," "Less than Others," or "Just like the Majority of Others". It helps clinicians and researchers to understand a child's sensory processing patterns and how these patterns impact their daily activities and behavior. In general, lower the score, better the performance.
Canadian occupational performance measure (COPM) – satisfaction evaluation
Canadian Occupational performance Measure (COPM) is a client-centered outcome measure to assess an individual’s perception of their occupational performance in the areas such as selfcare, productivity and leisure [122]. It is an evidence-based outcome measure to capture client’s satisfaction about the treatment success level and satisfaction [81,84]. It is a useful tool for identifying occupational functional improvement and tracking progress [123], assessed within five problems in the activities of daily life having a proven reliability and validity [124]. The five occupational performance activities selected within the appropriate COPM domain(s): (a) Personal Care-(Bathing, Dressing, Personal Hygiene); (b) Productivity (Writing); (c) Leisure (Reading, Drawing, Swimming, Tavel, Socialization), were evaluated by a trained healthcare personnel on a 10-point scale ranging from 1 to 10. The COPM uses a 10-point scale for both performance and satisfaction, where 1 indicates "not able to do it at all" and 10 indicates "able to do it extremely well" or "extremely satisfied" [81,83]. Scoring for all items under Performance and Satisfaction categories was recorded and the aggregate score for each category was entered on the score sheet. A higher score generally indicates better occupational performance and satisfaction, and a greater change in scores suggests a greater improvement [125].
Data analysis
The raw data from all participant-records were first entered into the excel spreadsheets and cross-checked independently by two healthcare professional team members for accuracy. Data were subjected to statistical analysis using GraphPad Prism (Version 8.0.2, Boston, MA, USA). GraphPad Prism software is deigned/programmed for analyzing all data at 95% confidence intervals.
- Data were first analyzed for descriptive column statistics to obtain respective group means with standard deviation (SD) and Standard error of means (SEM) at 95% confidence intervals.
- Data were analyzed by parametric Paired t-test (that assumes normal distribution), to compare the measurements from the same individual/group at two different time-points, such as pre- and post-test time-points, with an intervention administered between two time-points [126–129].
- Data were also analyzed by non-parametric Wilcoxon signed-rank test (that does not assume normal distribution) to compare two related samples or repeated measurements on the same subjects, as an alternative to the Paired t-test when the data do not meet the assumptions of normality, assessing whether the median differences between paired observations are significant [128,130,131].
- The use of both parametric Paired t-test and non-parametric Wilcoxon signed-rank test, was to rule out the concerns about normal distribution-related outcome variability and sample size of <100 [132–135].
- The data were analyzed by Analysis of Covariance (ANCOVA) that combines the Analysis of Variance (ANOVA) and Regression to compare the means of a dependent variables (Post-test) across independent variables (Pre-test) for comparing Std treatment vs Std+MUSIC treatment.
- The data were also analyzed using Bonferroni post hoc test for multiple comparisons to determine which specific group comparison is driving the effect.
- A value of p<0.05 was considered statistically significant.
Results
Primary outcomes
MUSIC co-treatment maximally attenuated autism advancement in verbal autistic children: ATEC measurements were performed to assess the advancement and severity of autism [110,111]. All 3 groups were subjected to ATEC assessments before (Baseline/Pre-) and after (Post-) the intervention(s).
Data for all three groups were analyzed and plotted representing:
- Individual ATEC score(s) derived from a total of all four diagnostic domains (Figure 6)
- Data representing group means for each diagnostic domain(s) of ATEC evaluation (Figure 7)
- ANCOVA Analysis comparing ATEC Domains of Std vs Std+MUSIC groups (Figure 8)
- Bonferroni Multiple Group Comparison (Table 1)
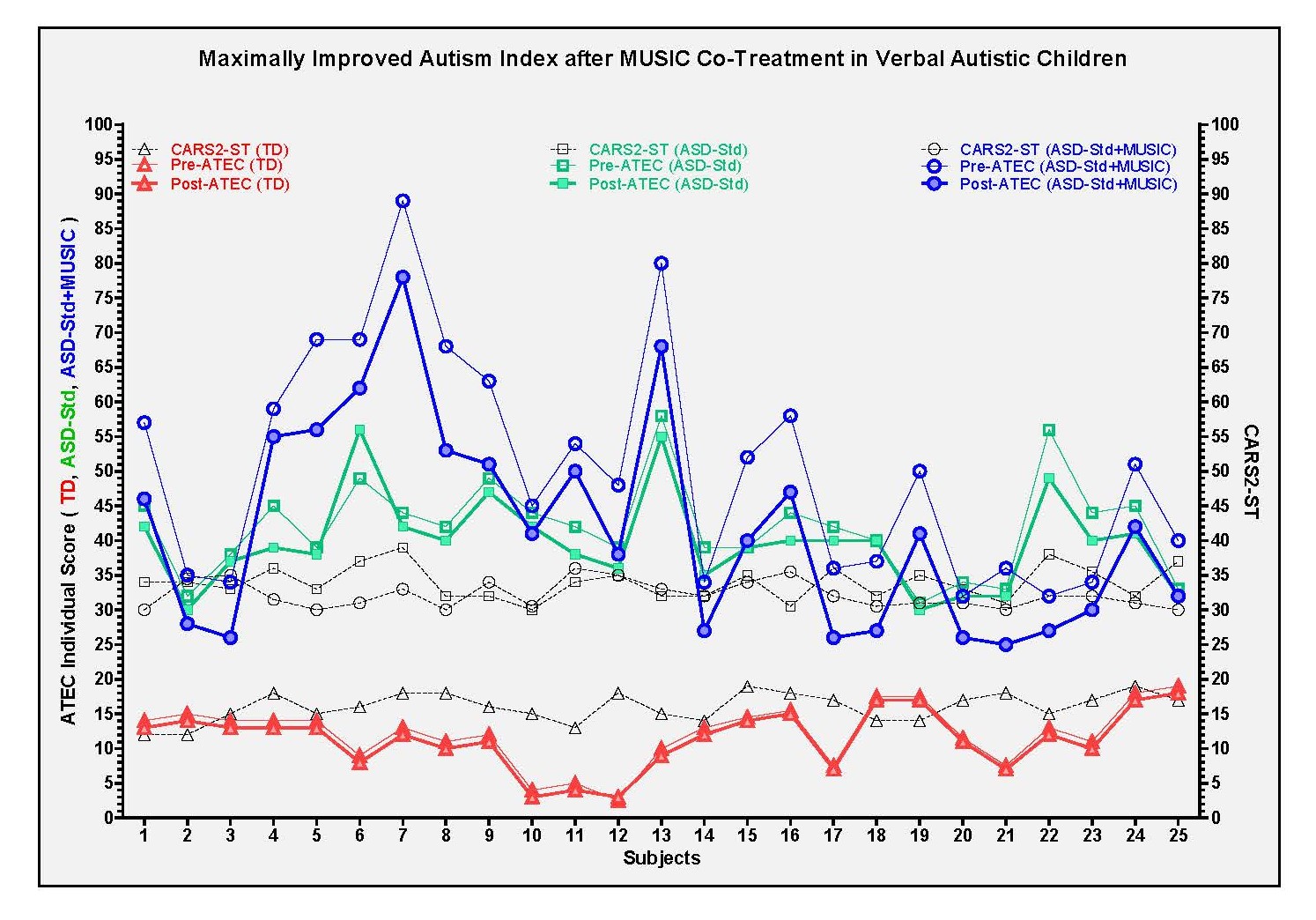
- Individual aggregate ATEC index with reference to CARS2-ST: Individual aggregate ATEC evaluation derived from 4 diagnostic domains for all groups, plotted with reference to respective CARS2-ST scores, showed remarkable group differences (Figure 6). As expected, typically developing (TD) untreated controls, in absence of ASD with <30 CARS2-ST scores (Open black triangles), did not exhibit presence of autism and/or its progression. The pre- and post-ATEC index was observed to be overlapping with no significant difference between pre-(Open bold green triangles) and post-(Filled bold green triangles) evaluation time points (Figure 6). On the other hand, ASD subjects with >30 CARS2-ST score (Open black squares with dotted line) that received only Std treatment, showed moderately improved ATEC score as indicated by reduced post-treatment ATEC values indicating improvement (Filled red squares) than the pre-treatment ATEC values (Open red squares). Such an improvement was found to be enhanced after MUSIC co-treatment in ASD subjects with >30 CARS2-ST score (Open black circles), showing remarkably reduced ATEC measure indicating maximum improvement (Filled blue circles) than the pre-treatment values of MUSIC-co-treated ASD group (Open blue circles) (Figure 6).
Figure 6. Individual ATEC index with reference to CARS2-ST in control and experimental groups.
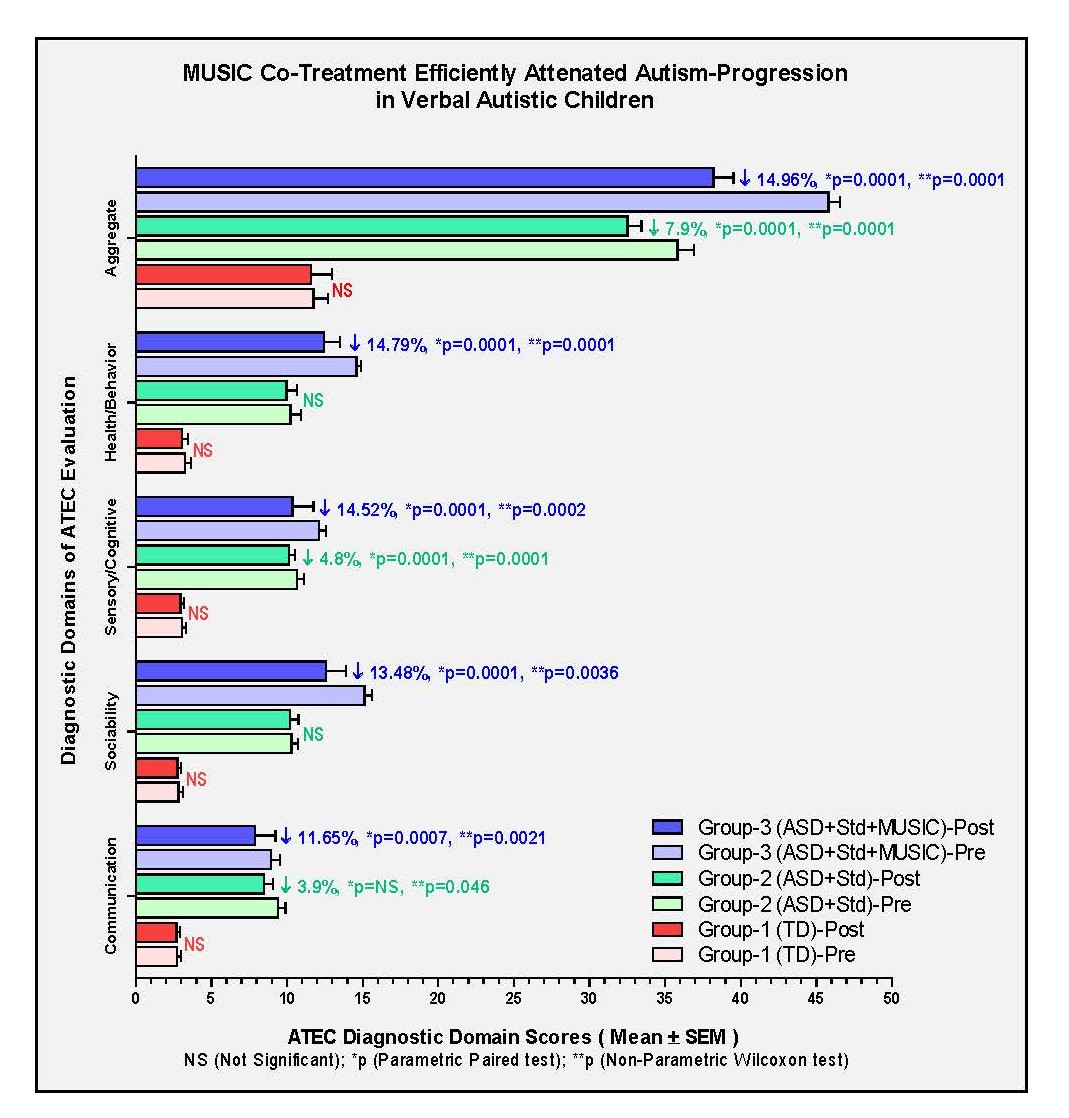
(b) Data representing group means for each diagnostic domain(s) of ATEC evaluation: Following the comparison of individual aggregate ATEC data, detailed analysis of each of the ATEC-diagnostic domains i.e. Speech/Language/Communication (Communication), Sociability, Sensory/Cognitive Awareness (Sensory/Cognitive), Health/Physical/Behavior (Health/Behavior) was conducted in all groups (Figure 7). Overall observations showed non-significant changes in all ATEC-diagnostic domains in untreated TD group (Figure 7, red bars). ASD group with Std treatment showed ~4% moderate improvement in only 2 diagnostic domains i.e. Communication and Sensory/Cognitive categories, with the overall improvement of ~8% (Figure 7, green bars). While ASD group that received Std plus MUSIC treatment showed improvement in all diagnostic domains ranging between ~11–15%, with maximum improvement of ~15% (*p/**p=0.0001) (Figure 7, blue bars).
Figure 7. ATEC diagnostic domains in control and experimental groups.
These observation indicate that MUSIC co-treatment enhanced the benefits of Std treatment in reducing the symptoms of autism in verbal autistic children.
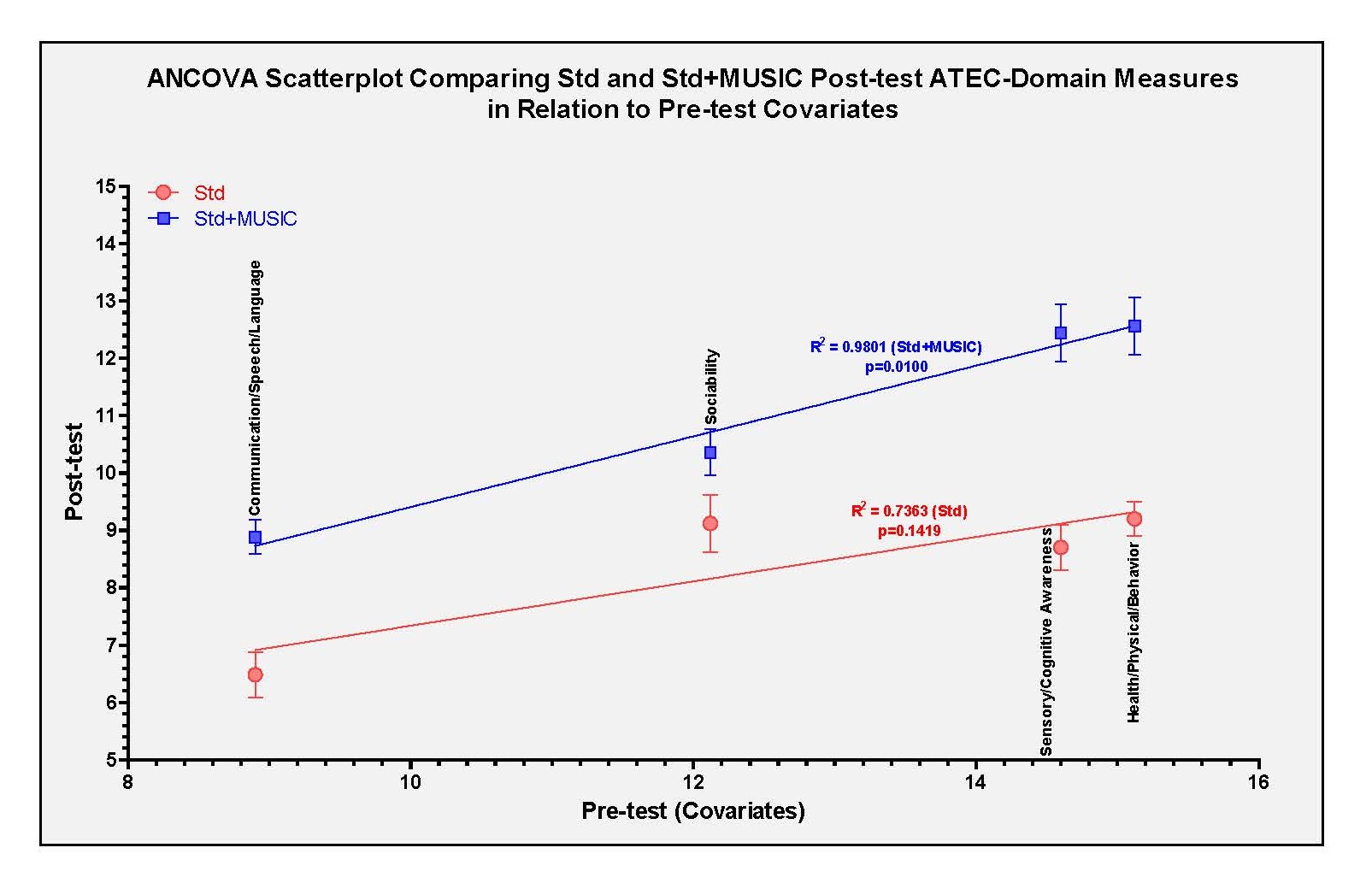
(c) ANCOVA comparing ATEC-Domains of Std vs Std+MUSIC Groups: Following the comparison of individual aggregate ATEC data, analysis of covariance (ANCOVA) for each of the ATEC-diagnostic domains was conducted at 95% confidence interval, where Std and Std+MUSIC post-test mean values as dependent variables compared with the independent pre-test covariates. The results showed group-specific effects in all diagnostic domain of ATEC assessment after ANCOVA in both experimental groups (Std treatment, Std+MUSIC treatment) (Figure 8). R2 values in this scatterplot indicated the proportion of variance in the dependent variables (Post-test measures) relative to Pre-test covariates (Figure 8). An observed R2 value of R2 = 0.7363 in Std group indicated ~70% of the variation in this dependable variable group (Std), while an observed R2 value of R2 = 0.9801 in Std+MUSIC group indicated >90% of the variation in this dependable variable group (Std+MUSIC) (Figure 8). The results indicate that despite moderately high R2 value (R2 = 0.7363), Std treatment did not reach statistical significance (p=0.1419), while Std+MUSIC treatment was found to be highly significant both with R2 (R2 = 0.9801) and p value (p=0.0100) (Figure 8). Overall, these results suggest the effectiveness of MUSIC add-on to Std treatment may be more advantageous in reducing ATEC-symptoms.
Figure 8. ANCOVA analysis of ATEC diagnostic domains in Std and Std+MUSIC experimental groups.
(d) Bonferroni multiple comparison of ATEC data between Std and Std+MUSIC groups: The Bonferroni multiple comparison test was conducted to determine which treatment-means are significantly different from each other, despite the fact that Bonferroni correction helps reduce Type I errors (false positives), but it can make it harder to find significant results, increasing the likelihood of Type II errors (false negatives). To determine which means are significantly different, Bonferroni multiple comparison test was conducted using GraphPad Prism, comparing all pairs between Std treatment (Pre and Post) and Std+MUSIC treatment (Pre & Post) with the mean-difference adjusted at the 95% confidence interval (Table 1).
|
Bonferroni's Multiple Comparison |
Mean Difference |
t Distribution |
Significance (p<0.05) |
Adjusted Difference Mean 95% CI |
|
Std (Pre) vs Std (Post) |
2.160 |
0.5275 |
Not Significant |
-8.872 to 13.19 |
|
Std (Pre) vs Std+MUSIC (Pre) |
-11.76 |
2.872 |
Not Significant |
-22.79 to -0.7283 |
|
Std (Pre) vs Std+MUSIC (Post) |
-2.840 |
0.6935 |
P=0.003 |
-13.87 to 8.192 |
|
Std (Post) vs Std+MUSIC (Pre) |
-13.92 |
3.399 |
P=0.002 |
-24.95 to -2.888 |
|
Std (Post) vs Std+MUSIC (Post) |
-5.000 |
1.221 |
Not Significant |
-16.03 to 6.032 |
|
Std+MUSIC (Pre) vs Std+MUSIC (Post) |
8.920 |
2.178 |
P=0.0001 |
-2.112 to 19.95 |
The results showed that the comparison between the Std (Pre) group vs Std (Post) or Std+MUSIC (Post) groups were non-significant. Similarly, the comparison between Std (Post) vs Std+MUSIC (Post) also was non-significant. On the other hand, the comparisons between Std (Pre) vs Std+MUSIC (Post) (p=0.003), Std (Post) vs Std+MUSIC (Pre) (p=0.002) were observed to be moderately significant, while the comparison between Std+MUSIC (Pre) vs Std+MUSIC (Post) reached the highest significance (p=0.0001). These results reinforce the ANCOVA findings showing the effectiveness of MUSIC add-on in reducing ATEC-symptoms.
Secondary outcomes
Based on the fact that abnormal sensory processing and atypical hypersensitivity to sensory input constitute a cardinal feature of ASD [79,115,136], sensory profile assessment was performed as a secondary outcome measure in the current study. Secondly, sensory challenges in ASD are known to impair perceptual and cognitive skills [83,137], and interfere with the occupational performance including activities of daily living [138]. Therefore, occupational performance using COPM and Parent Satisfaction were included in addition to SP2 assessment as a client-centered outcome measure, to verify the effects of intervention [83].
MUSIC co-treatment significantly restored sensory profile to normalcy in verbal autistic children
Sensory profile-2 (SP2) assessment was conducted in experimental groups with or without MUSIC intervention. ASD children typically score very high in almost all sensory profile diagnostic domains than TD children [79,136], and therefore lower sensory scoring is interpreted as interventional effectiveness towards normalcy [136]. Current observations are consistent with this concept where SP2 diagnostic domain scores before the treatment (Pre-test) are higher than (or unchanged) after the treatment (Post-test) (Figure 9). Results show that Std treatment combined with MUSIC treatment exhibited significant improvement in all diagnostic domains of SP2 (Figure 9). Specifically, Attention, Conduct and Auditory domains showed improvement <10%, while all other domains showed highly significant improvements ranging between 10%–20% (Figure 9).
Figure 9. SP2 diagnostic domains in experimental groups.
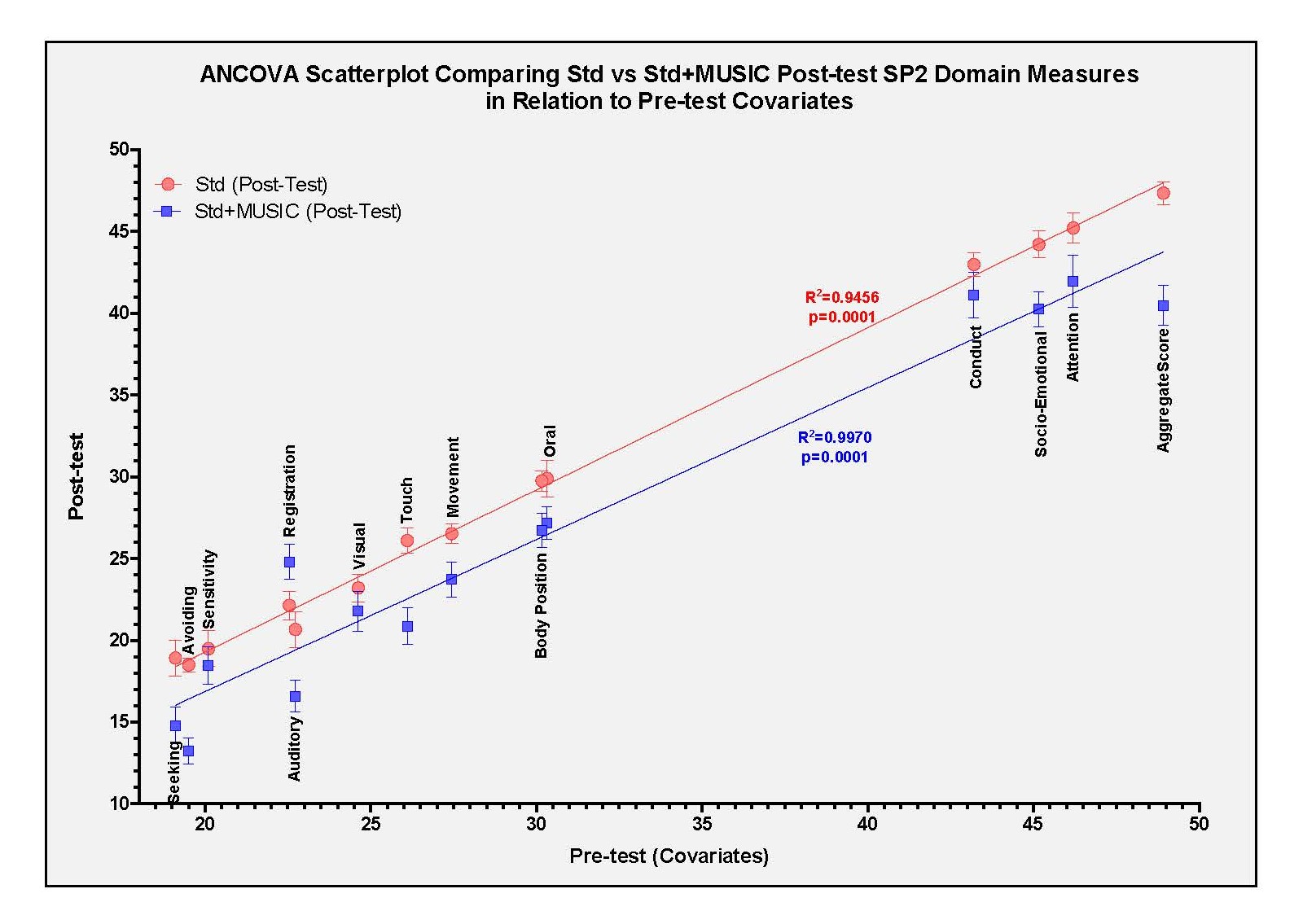
On the other hand, although Std treatment-effects were not found to reach the significant level, there was an observed trend towards marginal improvement in all SP2 diagnostic domains (Table 2). As observed in Figure 10, all diagnostic domain of SP2 evaluation showed group-specific effects after ANCOVA analysis in Std treatment group and in Std+MUSIC treatment group. The scatterplot showed R2 values indicating the proportion of variance in all dependent variables (Post-test measures) relative to Pre-test covariates (Figure 10). An observed R2 value of R2 = 0.9456 in Std group indicated >90% of variation in the Std treatment dependable variable, while an observed R2 value of R2 = 0.9970 in Std+MUSIC group indicated almost 100% of the variation in this dependable variable group (Std+MUSIC) (Figure 10). The results show that both Std treatment and Std+MUSIC treatment have exerted highly significant effects (P=0.0001), with slightly higher edge after MUSIC add-on treatment (Figure 10). These results support added advantage of MUSIC co-treatment in improving sensory profile in verbal autistic children. As observed for ATEC data analysis, SP2 data also showed that the comparison between Std (Pre) group vs Std (Post) or Std+MUSIC (Post) groups were non-significant (Table 3). Similarly, the comparison between Std (Post) vs Std+MUSIC (Pre) also was non-significant. On the other hand, the comparison between the Std (Pre) vs Std+MUSIC (Post) (p=0.002), and Std (Post) vs Std+MUSIC (Post) (p=0.005) were observed to be moderately significant, while the comparison between Std+MUSIC (Pre) vs Std+MUSIC (Post) was highly significant (p=0.0001) (Table 3). These results emphasize previous observations indicating the effectiveness of MUSIC add-on in improving sensory profile in verbal autistic children.
Figure 10. ANCOVA analysis of SP2 diagnostic domains in Std and Std+MUSIC experimental groups.
|
Sensory Profile 2 Diagnostic Domains |
Pre-SP2 (Mean ± SEM) (N=25) |
Post-SP2 (Mean ± SEM) (N=25) |
% Change |
Parametric Paired t-Test (*p Value) |
Non-Parametric Wilcoxon Signed Rank Test (**p Value) |
|
Seeking |
45.16±0.82 |
44.2±0.82 |
↓ 2.13% |
0.975 (NS) |
1.0 (NS) |
|
Avoiding |
46.2±0.48 |
45.2±0.92 |
↓ 2.16% |
0.657 (NS) |
0.625 (NS) |
|
Sensitivity |
43.2±0.50 |
42.98±0.72 |
↓ 0.62% |
0.467 (NS) |
0.365 (NS) |
|
Registration |
48.92±1.1 |
47.34±1.1 |
↓ 3.23% |
0.245 (NS) |
0.217 (NS) |
|
Auditory |
23.62±0.64 |
23.2±0.65 |
↓ 1.78% |
0.0542 (NS) |
0.0589 (NS) |
|
Visual |
19.5±0.47 |
18.5±0.43 |
↓ 4.13% |
0.795 (NS) |
0.821 (NS) |
|
Touch |
20.1±1.15 |
19.5±1.1 |
↓ 2.98% |
0.541 (NS) |
0.455 (NS) |
|
Movement |
22.52±0.51 |
21.88±0.58 |
↓ 2.84% |
0.196 (NS) |
0.167 (NS) |
|
Body Position |
19.1±0.58 |
18.92±0.63 |
↓ 0.94% |
0.449 (NS) |
0.561 (NS) |
|
Oral |
26.1±0.46 |
26.1± 0.58 |
↓ 0.00% |
0.455 (NS) |
0.609 (NS) |
|
Conduct |
22.54±0.56 |
22.14±0.56 |
↓ 1.77% |
0.837 (NS) |
0.866 (NS) |
|
Socio-Emotional |
30.32±1.56 |
29.89±1.6 |
↓ 1.42% |
0.271 (NS) |
0.286 (NS) |
|
Attention |
27.44±0.26 |
26.52±0.41 |
↓ 3.35% |
0.811 (NS) |
1.0 (NS) |
|
Aggregate Rank |
30.16±1.43 |
29.74±1.14 |
↓ 1.39% |
0.171 (NS) |
0.094 (NS) |
|
1 Sensory Profile 2 (SP2), 2 Autism Spectrum Disorder (ASD), 3 Not Significant (NS). |
|||||
|
Bonferroni's Multiple Comparison |
Mean Difference |
T Distribution |
Significance (p<0.05) |
Adjusted Difference Mean 95% CI |
|
Std (Pre) vs Std (Post) |
0.7136 |
1.206 |
Not Significant |
-0.9314 to 2.359 |
|
Std (Pre) vs Std+MUSIC (Pre) |
-0.4071 |
0.6880 |
Not Significant |
-2.052 to 1.238 |
|
Std (Pre) vs Std+MUSIC (Post) |
3.779 |
6.385 |
P= 0.002 |
2.134 to 5.424 |
|
Std (Post) vs Std+MUSIC (Pre) |
-1.121 |
1.894 |
Not Significant |
-2.766 to 0.5243 |
|
Std (Post) vs Std+MUSIC (Post) |
3.065 |
5.179 |
P=0.005 |
1.420 to 4.710 |
|
Std+MUSIC (Pre) vs Std+MUSIC (Post) |
4.186 |
7.073 |
P=0.0001 |
2.541 to 5.831 |
MUSIC co-treatment efficiently improved COPM-Satisfaction in verbal autistic children
Children suffering from ASD are known to express serious problems with sensory processing which affects their occupational performance in selfcare and productivity [83,137,138]. COPM is conventionally used to assess occupational performance of ASD and to see the effectiveness of therapeutic intervention [81,84,123]. COPM has two main scoring categories i.e. Performance and Satisfaction. A score between 1 (Not able to do at all) and 10 (able to do extremely well) [124] within the COPM domain(s). For both categories, a lower score indicates low performance and satisfaction while higher scores indicate occupational functional improvements and the impact of autism treatment success level and satisfaction [125]. Results show that although both Std and Std plus MUSIC interventions improved both categories of COPM assessment i.e. Performance and Satisfaction, more pronounced results were evident with MUSIC co-treatment.
Effect of Std and Std plus MUSIC co-treatment on COPM performance: ASD subjects treated only with Std treatment showed non-significant improvement in selfcare, productivity and one of the leisure activity (Reading) diagnostic domains of COPM, while rest of the two leisure categories (Sports, Socialization) were non-significantly improved (Table 4). Nonetheless, there was an overall significant improvement in COPM after Std treatment (Table 4). Compared to Std treatment (Table 4), when ASD subjects were treated with MUSIC add-on Co-treatment, there was significant improvement in all diagnostic domains of COPM (Table 5).
|
COPM1 Diagnostic Domains |
Pre-COPM1 (Mean ± SEM2) (N=25) |
Post-COPM1 (Mean ± SEM2) (N=25) |
% Improvement |
Parametric Paired t-Test (*p Value) |
Non-Parametric Wilcoxon Signed Rank t-Test (**p Value) |
|
Selfcare (Bathing, Dressing) |
3.03±0.38 |
4.34±0.43 |
á 43.4 % |
0.0001 |
0.0001 |
|
Productivity (Writing) |
2.8±0.33 |
3.67±0.35 |
á 31 % |
0.0035 |
0.0051 |
|
Leisure (Reading/Crafts) |
3.28±0.35 |
4.06±0.39 |
á 20.7 % |
0.0101 |
0.0053 |
|
Leisure (Sports/Travel) |
2.85±0.32 |
3.0±0.32 |
á 5.5 % |
0.4725 (NS)3 |
0.3410 (NS)3 |
|
Leisure (Socialization) |
3.06±0.38 |
3.22±0.43 |
á 5.3 % |
0.4205 (NS)3 |
0.3997 (NS)3 |
|
Aggregate Rank |
15.1±1.27 |
18.18±1.29 |
á 21.2 % |
0.0001 |
0.0001 |
|
1 Canadian Occupational Performance (COPM), 2 Standard Error of mean (SEM), 3 Not Significant (NS) |
|||||
|
COPM1 Diagnostic Domains |
Pre-COPM1 (Mean ± SEM2) (N=25) |
Post-COPM1 (Mean ± SEM2) (N=25) |
% Improvement |
Parametric Paired t-Test (*p Value) |
Non-Parametric Wilcoxon Signed Rank t-Test (**p Value) |
|
Selfcare (Bathing, Dressing) |
2.72±0.29 |
4.36±0.28 |
á 60.29 % |
0.0001 |
0.0001 |
|
Productivity (Writing) |
1.44±0.13 |
2.52±0.14 |
á 75.23 % |
0.0001 |
0.0001 |
|
Leisure (Reading/Crafts) |
1.8±0.19 |
2.76±0.29 |
á 53.33 % |
0.0001 |
0.0001 |
|
Leisure (Sports/Travel) |
1.48±0.12 |
2.2±0.14 |
á 48.64 % |
0.0001 |
0.0007 |
|
Leisure (Socialization) |
1.52±0.14 |
1.92±0.36 |
á 26.32 % |
0.0093 |
0.0192 |
|
Aggregate Rank |
8.88±0.56 |
13.76±0.62 |
á 54.95 % |
0.0001 |
0.0001 |
|
1 Canadian Occupational Performance (COPM), 2 Standard Error of mean (SEM) |
|||||
Effect of Std and Std plus MUSIC co-treatment on satisfaction: Satisfaction index in ASD subjects that received Std treatment showed significant improvements for selfcare area, whereas there were moderate improvements observed for Sports and Reading Leisure activity domains. While Writing and Socialization categories did not exhibit significant improvements, there was a significantly higher level of satisfaction when aggregate rank was considered (Table 6). The MUSIC co-treatment along with the Std treatment resulted in significantly enhanced Satisfaction levels in all diagnostic domains, along with overall highly significant improvement in aggregate score (Table 7). In Summary, significant differences were observed for ATEC assessment both after Std treatment and after MUSIC co-treatment, with greater improvements observed in MUSIC co-treatment group. With regard to sensory profile, SP2 scores did not reach the significance although the trend was towards improvement after Std treatment but significantly improved for after MUSIC co-treatment. Similar trend was observed for COPM-Satisfaction assessment showing overall greater impact of MUSIC co-treatment than Std treatment in verbal autistic children.
|
Satisfaction Diagnostic Domains |
Pre-Satisfaction (Mean ± SEM2) (N=25) |
Post-Satisfaction (Mean ± SEM2) (N=25) |
% Improvement |
Parametric Paired t-Test (*p Value) |
Non-Parametric Wilcoxon Signed Rank t-Test (**p Value) |
|
Selfcare (Bathing, Dressing) |
2.64±0.31 |
3.58±0.43 |
á 35.6 % |
0.0043 |
0.0036 |
|
Productivity (Writing) |
2.16±0.29 |
2.53±0.32 |
á 7.3% |
0.1132 (NS)3 |
0.1251 (NS)3 |
|
Leisure (Reading/Crafts) |
2.56±0.2 |
3.33±0.34 |
á 29.94 % |
0.021 |
0.0174 |
|
Leisure (Sports/Travel) |
2.48±0.32 |
2.74±0.33 |
á 10.5 % |
0.0396 |
0.0568 (NS)3 |
|
Leisure (Socialization) |
2.46±0.29 |
2.52±0.35 |
á 3.7 % |
0.6772 (NS)3 |
0.6658 (NS)3 |
|
Aggregate Rank |
12.4±1.17 |
14.44±1.19 |
á 16.45 % |
0.0001 |
0.0001 |
|
1 Canadian Occupational Performance (COPM), 2 Standard Error of mean (SEM), 3 Not Significant (NS) |
|||||
|
Satisfaction Diagnostic Domains |
Pre- Satisfaction1 (Mean ± SEM2) (N=25) |
Post-Satisfaction1 (Mean ± SEM2) (N=25) |
% Improvement |
Parametric Paired t-Test (*p Value) |
Non-Parametric Wilcoxon Signed Rank t-Test (**p Value) |
|
Selfcare (Bathing, Dressing) |
1.36±0.09 |
2.24±0.16 |
á 64.7 % |
0.0001 |
0.0001 |
|
Productivity (Writing) |
1.21±0.08 |
1.84±0.12 |
á 53.34% |
0.0006 |
0.0020 |
|
Leisure (Reading/Crafts) |
1.56±0.18 |
2.12±0.34 |
á 35.89 % |
0.0198 |
0.0236 |
|
Leisure (Sports/Travel) |
1.36±0.09 |
1.84±0.12 |
á 35.29 % |
0.0152 |
0.0197 |
|
Leisure (Socialization) |
1.4±0.22 |
1.8±0.29 |
á 28.57 % |
0.0093 |
0.0083 |
|
Aggregate Rank |
6.84±0.44 |
9.84±0.44 |
á 43.86 % |
0.0001 |
0.0001 |
|
1Satisfaction, 2 Standard Error of mean (SEM) |
|||||
Discussion
This randomized controlled pilot trial tested the hypothesis that multifold effects of Indian classical Raga (MUSIC) may be more effective in enhancing the outcomes of conventional therapy in treating verbal autistic children. Music per se, has shown promising results in maintaining physical and mental well-being in general [139–141]. Especially in case of ASD, therapeutic use of music is even further magnified since preference for music constitutes one of the strengths of autism [38,39,142]. Moreover, music intervention primarily focuses on developing social, emotional and other sensory processing abilities, and impairments in these abilities are central to ASD [143]. One of the earliest study reported effects of music therapy on enhancing social skills in children with autism [101]. Sharda et al. have shown that 8–12 weeks of music exposure restored auditory-motor intrinsic connectivity and improved parent-reported outcomes in social communication [41]. Another study showed clinical improvement in Aberrant Behavior Checklist (ABC) and Clinical Global Impression (CGI) outcomes in children with ASD [142]. A pilot study conducted by Feng et al. showed improved fine motor skills in autistic children after music therapy [144]. A recent randomized trial showed that music therapy improved engagement and intellectual disability in autistic children [60]. Currently, there are 26 clinical trials completed/ongoing involving the use of music intervention in treating ASD (Clinicaltrials.gov). Most of these are aimed at treating autism-progression and sensory processing abnormalities. Most clinical trials have not posted their results/outcomes, however, some of the findings do show reduced autism-symptoms and improved sensory profiles (Clinicaltrials.gov). To our knowledge, this is one of the pioneering studies in reporting effectiveness of Indian classical Raga (MUSIC) in alleviating autism-symptoms. Current findings are consistent with the previous reports but show added advantage of Indian classical Raga. The enhanced potential of MUSIC that used Raga Yaman (https://youtu.be/viL-8iubNpE) may be attributed to the multifold mechanisms of Raga Yaman. The Predominance of Shuddha Swaras (pure notes) of Raga Yaman makes the Raga “Happy” and “Cheerful”, and the presence of Tivra “Ma” intensifies the inherent emotions of accompanying Swara(s) [61,66,145,146]. This effect may activate emotional processing and reward system [46] by elevating hormones involved in emotional/reward/cognitive processing and neuroplasticity while simultaneously reducing stress hormones [50–54], promoting functional brain connectivity leading to improvements in social communication, emotional and motor functions [37,41,42,60]. Thus, the intensified happy/cheerfulness elicited by Raga Yaman will stimulate joy, uplift emotions, remove “isolation”, and promote sociability and communication in autistic kids. Moreover, the “Sampurna” Nature of Raga is considered to activate all energy centers (Chakras) [68,147,148], exposing individual to a full range of the frequency spectrum can “open up” a person to wholeness [149]. Thus, Raga Yaman will help focus wandering attention and open up to wholeness in autism. Autism is a neurodevelopmental disorder. Swadhishthana Chakra governs creativity, pre- and post-natal development, emotions/mood, anxiety [68,150]. Therefore, in addition to above-mentioned effects, Raga Yaman, can also boost self-esteem and control aggression in autistic kids. Currently observed “enhancing” effects of MUSIC due to its pleiotropy arising from the multifold mechanisms discussed above justify proposed hypothesis in support of using MUSIC as an effective adjuvant in treating ASD.
Conclusions
Current study suggests effectiveness of MUSIC intervention as an effective add-on in treating autism spectrum disorder. These findings warrant future research in testing Raga co-treatment for larger trials. We hope that autistic children and their families will be most impacted by the results of our study and may help them.
Author Contributions
All authors contributed equally.
Conflict of Interests
All authors declare no conflict of interests, including the author (MK) who has permitted one of his videos used for MUSIC intervention in this study.
Funding Disclosure
This work was funded by the support provided by the Fulbright U.S. Scholar Program, U.S. Department of State and United States-India Educational Foundation (USIEF) Fulbright Commission to Neelima Chauhan (PI) (Fulbright-Nehru Academic and Professional Excellence Awardee; Grant #APE 2023-24 USIEF).
Acknowledgements
Authors gratefully acknowledges the support provided by the U.S. Department of State and United States-India Educational Foundation (USIEF) Fulbright Commission, India. Its contents are solely the responsibility of the author and do not represent official views of Fulbright Program, USIEF. Authors also acknowledge the support provided by the Department of Pediatrics, University of Illinois at Chicago, Chicago, IL, USA.
Data Availability
The data are not publicly available due to privacy restrictions and ethical regulations. The data presented in the study can be made available upon request from the corresponding author only for scientific/academic purposes.
References
2. Ostrowski J, Religioni U, Gellert B, Sytnik-Czetwertyński J, Pinkas J. Autism Spectrum Disorders: Etiology, Epidemiology, and Challenges for Public Health. Med Sci Monit. 2024 Jun 4;30:e944161.
3. Maenner MJ, Warren Z, Williams AR, Amoakohene E, Bakian AV, Bilder DA, et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years - Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill Summ. 2023 Mar 24;72(2):1–14.
4. Zeidan J, Fombonne E, Scorah J, Ibrahim A, Durkin MS, Saxena S, et al. Global prevalence of autism: A systematic review update. Autism Res. 2022 May;15(5):778–90.
5. Saral D, Olcay S, Ozturk H. Autism Spectrum Disorder: When There is no Cure, There are Countless of Treatments. J Autism Dev Disord. 2023 Dec;53(12):4901–16.
6. Hellings J. Pharmacotherapy in autism spectrum disorders, including promising older drugs warranting trials. World J Psychiatry. 2023 Jun 19;13(6):262–77.
7. Turner M. The role of drugs in the treatment of autism. Aust Prescr. 2020 Dec;43(6):185–90.
8. Shamabadi A, Karimi H, Arabzadeh Bahri R, Motavaselian M, Akhondzadeh S. Emerging drugs for the treatment of irritability associated with autism spectrum disorder. Expert Opin Emerg Drugs. 2024 Mar;29(1):45–56.
9. Fieiras C, Chen MH, Escobar Liquitay CM, Meza N, Rojas V, Franco JVA, et al. Risperidone and aripiprazole for autism spectrum disorder in children: an overview of systematic reviews. BMJ Evid Based Med. 2023 Feb;28(1):7–14.
10. Maniram J, Karrim SBS, Oosthuizen F, Wiafe E. Pharmacological Management of Core Symptoms and Comorbidities of Autism Spectrum Disorder in Children and Adolescents: A Systematic Review. Neuropsychiatr Dis Treat. 2022 Aug 7;18:1629–44.
11. Yu Z, Zhang P, Tao C, Lu L, Tang C. Efficacy of nonpharmacological interventions targeting social function in children and adults with autism spectrum disorder: A systematic review and meta-analysis. PLoS One. 2023 Sep 19;18(9):e0291720.
12. Wang X, Zhao J, Huang S, Chen S, Zhou T, Li Q, et al. Cognitive Behavioral Therapy for Autism Spectrum Disorders: A Systematic Review. Pediatrics. 2021 May;147(5):e2020049880.
13. Lepri A. Psychoeducational and Behavioral Interventions in Autism Spectrum Disorder: is the ABA Method Really the Most Effective? Psychiatr Danub. 2024 Sep;36(Suppl 2):203–04.
14. Elliott SJ, Marshall D, Morley K, Uphoff E, Kumar M, Meader N. Behavioural and cognitive behavioural therapy for obsessive compulsive disorder (OCD) in individuals with autism spectrum disorder (ASD). Cochrane Database Syst Rev. 2021 Sep 3;9(9):CD013173.
15. Martínez-Vérez V, Gil-Ruíz P, Domínguez-Lloria S. Interventions through Art Therapy and Music Therapy in Autism Spectrum Disorder, ADHD, Language Disorders, and Learning Disabilities in Pediatric-Aged Children: A Systematic Review. Children (Basel). 2024 Jun 7;11(6):706.
16. Khalil R, Demarin V. Creative therapy in health and disease: Inner vision. CNS Neurosci Ther. 2024 Mar;30(3):e14266.
17. Bernier A, Ratcliff K, Hilton C, Fingerhut P, Li CY. Art Interventions for Children With Autism Spectrum Disorder: A Scoping Review. Am J Occup Ther. 2022 Sep 1;76(5):7605205030.
18. Ke X, Song W, Yang M, Li J, Liu W. Effectiveness of music therapy in children with autism spectrum disorder: A systematic review and meta-analysis. Front Psychiatry. 2022 Oct 6;13:905113.
19. Zhou Z, Zhao X, Yang Q, Zhou T, Feng Y, Chen Y, et al. A randomized controlled trial of the efficacy of music therapy on the social skills of children with autism spectrum disorder. Res Dev Disabil. 2025 Mar;158:104942.
20. Shi Z, Wang S, Chen M, Hu A, Long Q, Lee Y. The effect of music therapy on language communication and social skills in children with autism spectrum disorder: a systematic review and meta-analysis. Front Psychol. 2024 May 7;15:1336421.
21. Gao X, Xu G, Fu N, Ben Q, Wang L, Bu X. The effectiveness of music therapy in improving behavioral symptoms among children with autism spectrum disorders: a systematic review and meta-analysis. Front Psychiatry. 2025 Jan 14;15:1511920.
22. Braconnier ML, Siper PM. Neuropsychological Assessment in Autism Spectrum Disorder. Curr Psychiatry Rep. 2021 Jul 30;23(10):63.
23. Lai MC, Lombardo MV, Baron-Cohen S. Autism. Lancet. 2014 Mar 8;383(9920):896–910.
24. Genovese A, Butler MG. The Autism Spectrum: Behavioral, Psychiatric and Genetic Associations. Genes (Basel). 2023 Mar 9;14(3):677.
25. Saure E, Laasonen M, Kylliäinen A, Hämäläinen S, Lepistö-Paisley T, Raevuori A. Social communication and restricted, repetitive behavior as assessed with a diagnostic tool for autism (ADOS-2) in women with anorexia nervosa. J Clin Psychol. 2024 Aug;80(8):1901–16.
26. Hirota T, King BH. Autism Spectrum Disorder: A Review. JAMA. 2023 Jan 10;329(2):157–68.
27. Bougeard C, Picarel-Blanchot F, Schmid R, Campbell R, Buitelaar J. Prevalence of Autism Spectrum Disorder and Co-Morbidities in Children and Adolescents: A Systematic Literature Review. Focus (Am Psychiatr Publ). 2024 Apr;22(2):212–28.
28. LaGasse B, Yoo GE, Hardy MW. Rhythm and music for promoting sensorimotor organization in autism: broader implications for outcomes. Front Integr Neurosci. 2024 Jul 8;18:1403876.
29. Jaschke AC, Howlin C, Pool J, Greenberg YD, Atkinson R, Kovalova A, et al. Study protocol of a randomized control trial on the effectiveness of improvisational music therapy for autistic children. BMC Psychiatry. 2024 Sep 27;24(1):637.
30. Courchesne V, Langlois V, Gregoire P, St-Denis A, Bouvet L, Ostrolenk A, Mottron L. Interests and Strengths in Autism, Useful but Misunderstood: A Pragmatic Case-Study. Front Psychol. 2020 Oct 6;11:569339.
31. Brancatisano O, Baird A, Thompson WF. Why is music therapeutic for neurological disorders? The Therapeutic Music Capacities Model. Neurosci Biobehav Rev. 2020 May;112:600–15.
32. Dan Y, Xiong Y, Xu D, Wang Y, Yin M, Sun P, et al. Potential common targets of music therapy intervention in neuropsychiatric disorders: the prefrontal cortex-hippocampus -amygdala circuit (a review). Front Hum Neurosci. 2025 Feb 3;19:1471433.
33. Gassner L, Geretsegger M, Mayer-Ferbas J. Effectiveness of music therapy for autism spectrum disorder, dementia, depression, insomnia and schizophrenia: update of systematic reviews. Eur J Public Health. 2022 Feb 1;32(1):27–34.
34. Huang X, Luo X. Music therapy for cognitive deficits of neuropsychiatric disorders. World J Psychiatry Ment Health Res. 2024;7(1):1039. Epub 2024 Apr 17.
35. Lin TH, Liao YC, Tam KW, Chan L, Hsu TH. Effects of music therapy on cognition, quality of life, and neuropsychiatric symptoms of patients with dementia: A systematic review and meta-analysis of randomized controlled trials. Psychiatry Res. 2023 Nov;329:115498.
36. Molnar-Szakacs I, Heaton P. Music: a unique window into the world of autism. Ann N Y Acad Sci. 2012 Apr;1252:318–24.
37. LaGasse AB. Social outcomes in children with autism spectrum disorder: a review of music therapy outcomes. Patient Relat Outcome Meas. 2017 Feb 20;8:23–32.
38. Goris J, Brass M, Cambier C, Delplanque J, Wiersema JR, Braem S. The Relation Between Preference for Predictability and Autistic Traits. Autism Res. 2020 Jul;13(7):1144–54.
39. Jaschke AC, Howlin C, Pool J, Greenberg YD, Atkinson R, Kovalova A, et al. Correction: Study protocol of a randomized control trial on the effectiveness of improvisational music therapy for autistic children. BMC Psychiatry. 2024 Nov 15;24(1):815
40. Geretsegger M, Elefant C, Mössler KA, Gold C. Music therapy for people with autism spectrum disorder. Cochrane Database Syst Rev. 2014 Jun 17;2014(6):CD004381.
41. Sharda M, Tuerk C, Chowdhury R, Jamey K, Foster N, Custo-Blanch M, et al. Music improves social communication and auditory-motor connectivity in children with autism. Transl Psychiatry. 2018 Oct 23;8(1):231.
42. Quintin EM. Music-Evoked Reward and Emotion: Relative Strengths and Response to Intervention of People With ASD. Front Neural Circuits. 2019 Sep 18;13:49.
43. Alayidh M, Alawad F, Alanazy WB, Al-Harbi FO, Alotaibi AM, Al Mohammed QAet al. Music Therapy for People With Autism Spectrum Disorder: A Systematic Review of Randomized Clinical Trials. Cureus. 2025 Mar 28;17(3):e81361.
44. Ghasemtabar SN, Hosseini M, Fayyaz I, Arab S, Naghashian H, Poudineh Z. Music therapy: An effective approach in improving social skills of children with autism. Adv Biomed Res. 2015 Jul 27;4:157.
45. Mössler K, Schmid W, Aßmus J, Fusar-Poli L, Gold C. Attunement in Music Therapy for Young Children with Autism: Revisiting Qualities of Relationship as Mechanisms of Change. J Autism Dev Disord. 2020 Nov;50(11):3921–34.
46. Caria A, Venuti P, de Falco S. Functional and dysfunctional brain circuits underlying emotional processing of music in autism spectrum disorders. Cereb Cortex. 2011 Dec;21(12):2838–49.
47. Chen WG, Iversen JR, Kao MH, Loui P, Patel AD, Zatorre RJet al. Music and Brain Circuitry: Strategies for Strengthening Evidence-Based Research for Music-Based Interventions. J Neurosci. 2022 Nov 9;42(45):8498–507.
48. Zatorre RJ. Hemispheric asymmetries for music and speech: Spectrotemporal modulations and top-down influences. Front Neurosci. 2022 Dec 20;16:1075511.
49. Sharma SR, Gonda X, Tarazi FI. Autism Spectrum Disorder: Classification, diagnosis and therapy. Pharmacol Ther. 2018 Oct;190:91–104.
50. Poquérusse J, Azhari A, Setoh P, Cainelli S, Ripoli C, Venuti P, et al. Salivary α-amylase as a marker of stress reduction in individuals with intellectual disability and autism in response to occupational and music therapy. J Intellect Disabil Res. 2018 Feb;62(2):156–63.
51. Salimpoor VN, Benovoy M, Larcher K, Dagher A, Zatorre RJ. Anatomically distinct dopamine release during anticipation and experience of peak emotion to music. Nat Neurosci. 2011 Feb;14(2):257–62.
52. Ferreri L, Mas-Herrero E, Cardona G, Zatorre RJ, Antonijoan RM, Valle M, et al. Dopamine modulations of reward-driven music memory consolidation. Ann N Y Acad Sci. 2021 Oct;1502(1):85–98.
53. Mas-Herrero E, Ferreri L, Cardona G, Zatorre RJ, Pla-Juncà F, Antonijoan RM, et al. The role of opioid transmission in music-induced pleasure. Ann N Y Acad Sci. 2023 Feb;1520(1):105–14.
54. Caria A, Ciringione L, Falco S. Morphofunctional Alterations of the Hypothalamus and Social Behavior in Autism Spectrum Disorders. Brain Sci. 2020 Jul 8;10(7):435.
55. Speranza L, Pulcrano S, Perrone-Capano C, di Porzio U, Volpicelli F. Music affects functional brain connectivity and is effective in the treatment of neurological disorders. Rev Neurosci. 2022 Mar 24;33(7):789–801.
56. Chatterjee D, Hegde S, Thaut M. Neural plasticity: The substratum of music-based interventions in neurorehabilitation. NeuroRehabilitation. 2021;48(2):155–66.
57. Ramaswamy M, Philip JL, Priya V, Priyadarshini S, Ramasamy M, Jeevitha GC, et al. Therapeutic use of music in neurological disorders: A concise narrative review. Heliyon. 2024 Aug 8;10(16):e35564.
58. Kunikullaya U K, Pranjić M, Rigby A, Pallás-Ferrer I, Anand H, Kunnavil R, et al. The molecular basis of music-induced neuroplasticity in humans: A systematic review. Neurosci Biobehav Rev. 2025 Aug;175:106219.
59. Bharathi G, Jayaramayya K, Balasubramanian V, Vellingiri B. The potential role of rhythmic entrainment and music therapy intervention for individuals with autism spectrum disorders. J Exerc Rehabil. 2019 Apr 26;15(2):180–6.
60. Yum YN, Poon K, Lau WK, Ho FC, Sin KF, Chung KM, et al. Music therapy improves engagement and initiation for autistic children with mild intellectual disabilities: A randomized controlled study. Autism Res. 2024 Dec;17(12):2702–22.
61. Valla JM, Alappatt JA, Mathur A, Singh NC. Music and Emotion-A Case for North Indian Classical Music. Front Psychol. 2017 Dec 19;8:2115.
62. Sharma S, Sasidharan A, Marigowda V, Vijay M, Sharma S, Mukundan CS, et al. Indian classical music with incremental variation in tempo and octave promotes better anxiety reduction and controlled mind wandering-A randomised controlled EEG study. Explore (NY). 2021 Mar-Apr;17(2):115–21.
63. Ganguli KK, Rao P. On the perception of raga motifs by trained musicians. J Acoust Soc Am. 2019 Apr;145(4):2418.
64. Babel S, Baral S, Srivastava A. Impact of Listening to Indian Classical Music, or Rāgas, on the Electroencephalogram: A Meta-Analysis. Cureus. 2023 Nov 28;15(11):e49592.
65. Bardekar AA, Gurjar AA. Empirical Study of Indian Classical Ragas Structure and its Emotional Influence on Human Body For Music Therapy. Journal of Management Engineering and Information Technology. 2016;3(4).
66. Bardekar AA. Analysis of Indian Classical Raga Yaman Influence on Human Brain Waves. International Journal of Creative Research Thoughts (IJCRT). 2018;6(2):655–9.
67. Sarkar J, Biswas U. An effect of Raga Therapy on our human body. Int J Humanit Soc Sci Res. 2015 Nov;1(1):40–3.
68. Chauhan NB, Kale M, Gujar-Kale P. Raga: The connecting link between psyche and soma. The Neuroscience Chronicles. 2024 Aug 31;4(1):31–5.
69. Chauhan N, Kale M, Naik N. Raga Therapy for Autism. Medp Neurol Neurosurg. 2022;3(1):1–7.
70. Sravanti L, Kommu JV, Suswaram S, Yadav AS. Musical preferences of Indian children with autism spectrum disorder and acceptability of music therapy by their families: An exploratory study. Industrial psychiatry journal. 2023 Jan 1;32(1):176–86.
71. Tager‐Flusberg H, Kasari C. Minimally verbal school‐aged children with autism spectrum disorder: The neglected end of the spectrum. Autism research. 2013 Dec;6(6):468–78.
72. Brignell A, Williams KJ, Reilly S, Morgan AT. Language growth in verbal autistic children from 5 to 11 years. Autism Research. 2024 Oct;17(10):1994-2003.
73. Al Backer NB. Correlation between Autism Treatment Evaluation Checklist (ATEC) and Childhood Autism Rating Scale (CARS) in the evaluation of autism spectrum disorder. Sudan J Paediatr. 2016;16(1):17–22.
74. Netson R, Schmiedel Fucks A, Schmiedel Sanches Santos A, Poloni LEP, Nacano NN, Fucks E, et al. A Comparison of Parent Reports, the Mental Synthesis Evaluation Checklist (MSEC) and the Autism Treatment Evaluation Checklist (ATEC), with the Childhood Autism Rating Scale (CARS). Pediatr Rep. 2024 Mar 11;16(1):174–89.
75. Fabbri-Destro M, Maugeri F, Ianni C, Corsini S, Di Stefano E, Scatigna S, et al. Early Sensory Profile in Autism Spectrum Disorders Predicts Emotional and Behavioral Issues. J Pers Med. 2022 Sep 27;12(10):1593.
76. Loh SY, Ee SI, Marret MJ. Sensory processing and its relationship to participation among childhood occupations in children with autism spectrum disorder: exploring the profile of differences. Int J Dev Disabil. 2021 Jul 16;69(2):226–37.
77. Kilroy E, Aziz-Zadeh L, Cermak S. Ayres Theories of Autism and Sensory Integration Revisited: What Contemporary Neuroscience Has to Say. Brain Sci. 2019 Mar 21;9(3):68.
78. Sakamoto N, Oe M, Ozone M. An Exploratory Study of the Associations Between Sensory Processing Patterns and Emotional/Behavioral Problems in Children with Autism Spectrum Disorder. Kurume Med J. 2024 Jul 2;70(1.2):47–52.
79. Tomchek SD, Dunn W. Sensory processing in children with and without autism: a comparative study using the short sensory profile. Am J Occup Ther. 2007 Mar-Apr;61(2):190–200.
80. Brown NB, Dunn W. Relationship between context and sensory processing in children with autism. Am J Occup Ther. 2010 May-Jun;64(3):474–83.
81. Kim Y, Kim M, Park C, You JSH. Effects of Integrative Autism Therapy on Multiple Physical, Sensory, Cognitive, and Social Integration Domains in Children and Adolescents with Autism Spectrum Disorder: A 4-Week Follow-Up Study. Children (Basel). 2022 Dec 15;9(12):1971.
82. Little LM, Dean E, Tomchek S, Dunn W. Sensory Processing Patterns in Autism, Attention Deficit Hyperactivity Disorder, and Typical Development. Phys Occup Ther Pediatr. 2018 Aug;38(3):243–54.
83. Beheshti SZ, Hosseini SS, Maroufizadeh S, Almasi-Hashiani A. Occupational performance of children with autism spectrum disorder and quality of life of their mothers. BMC Res Notes. 2022 Jan 15;15(1):18.
84. Ghahramani S, Hassani Mehraban A, Alizadeh Zarei M, Ghahramani S. Occupational Therapy Outcome Measures in Preschool Children With Autism Spectrum Disorders: A Scoping Review. OTJR (Thorofare N J). 2024 Oct;44(4):568–76.
85. Papavasiliou AS, Nikaina I, Rizou J, Alexandrou S. The effect of a psycho-educational program on CARS scores and short sensory profile in autistic children. Eur J Paediatr Neurol. 2011 Jul;15(4):338–44.
86. Ji SI, Park H, Yoon SA, Hong SB. A Validation Study of the CARS-2 Compared With the ADOS-2 in the Diagnosis of Autism Spectrum Disorder: A Suggestion for Cutoff Scores. J Korean Acad Child Adolesc Psychiatry. 2023 Jan 1;34(1):45–50.
87. Schopler E, Reichler RJ, DeVellis RF, Daly K. Toward objective classification of childhood autism: Childhood Autism Rating Scale (CARS). J Autism Dev Disord. 1980 Mar;10(1):91–103.
88. Renner ESRJRBR. The childhood autism rating scale (CARS) for diagnostic screening and classification of autism. New York: Irvington;1986.
89. Schopler E vBM, Wellman GJ, Love SR. Childhood Autism Rating Scale-Second Edition (CARS-2). Western Psychological Services, Los Angeles; 2010.
90. Dawkins T, Meyer AT, Van Bourgondien ME. The Relationship Between the Childhood Autism Rating Scale: Second Edition and Clinical Diagnosis Utilizing the DSM-IV-TR and the DSM-5. J Autism Dev Disord. 2016 Oct;46(10):3361–8.
91. Huang CY, Chen KS, Lee KY, Lin CH, Chen KL. Different autism measures targeting different severity levels in children with autism spectrum disorder. Eur Arch Psychiatry Clin Neurosci. 2024 Feb;274(1):27–33.
92. Samadi SA, Mahmoodizadeh A, Foladgar M, Moradi SB, Lotfi B, McConkey R. The Childhood Autism Rating Scale Second Edition (CARS2) and Its Applicability in an Iranian Sample. Autism Res. 2025 Mar;18(3):541–52.
93. Moon SJ, Hwang JS, Shin AL, Kim JY, Bae SM, Sheehy-Knight J, et al. Accuracy of the Childhood Autism Rating Scale: a systematic review and meta-analysis. Dev Med Child Neurol. 2019 Sep;61(9):1030–8.
94. Huang CY, Chen KS, Lee KY, Lin CH, Chen KL. Correction: Different autism measures targeting different severity levels in children with autism spectrum disorder. Eur Arch Psychiatry Clin Neurosci. 2024 Jun;274(4):1021.
95. Park HS, Yi SY, Yoon SA, Hong SB. Comparison of the Autism Diagnostic Observation Schedule and Childhood Autism Rating Scale in the Diagnosis of Autism Spectrum Disorder: A Preliminary Study. Soa Chongsonyon Chongsin Uihak. 2018 Oct 1;29(4):172–7.
96. Randall M, Egberts KJ, Samtani A, Scholten RJ, Hooft L, Livingstone Net al. Diagnostic tests for autism spectrum disorder (ASD) in preschool children. Cochrane Database Syst Rev. 2018 Jul 24;7(7):CD009044.
97. Whitehead AL, Julious SA, Cooper CL, Campbell MJ. Estimating the sample size for a pilot randomised trial to minimise the overall trial sample size for the external pilot and main trial for a continuous outcome variable. Stat Methods Med Res. 2016 Jun;25(3):1057–73.
98. Fachner J, Maidhof C, Murtagh D, De Silva D, Pasqualitto F, Fernie Pet al. Music therapy, neural processing, and craving reduction: an RCT protocol for a mixed methods feasibility study in a Community Substance Misuse Treatment Service. Addict Sci Clin Pract. 2023 May 27;18(1):36.
99. Shin JH, Jeong E. Virtual reality-based music attention training for acquired brain injury: A protocol for randomized cross-over trial. Front Neurol. 2023 Aug 8;14:1192181.
100. Kunselman AR. A brief overview of pilot studies and their sample size justification. Fertil Steril. 2024 Jun;121(6):899–901.
101. LaGasse AB. Effects of a music therapy group intervention on enhancing social skills in children with autism. J Music Ther. 2014 Fall;51(3):250–75.
102. Julious SA. Sample size of 12 per group rule of thumb for a pilot study. Pharmaceutical Statistics: The Journal of Applied Statistics in the Pharmaceutical Industry. 2005 Oct;4(4):287–91.
103. Hertzog MA. Considerations in determining sample size for pilot studies. Res Nurs Health. 2008 Apr;31(2):180–91.
104. Williams TI, Loucas T, Sin J, Jeremic M, Aslett G, Knight M, et al. A randomised controlled feasibility trial of music-assisted language telehealth intervention for minimally verbal autistic children-the MAP study protocol. Pilot Feasibility Stud. 2021 Oct 1;7(1):182.
105. Anderson LK. Autistic experiences of applied behavior analysis. Autism. 2023 Apr;27(3):737–50.
106. Kashefimehr B, Kayihan H, Huri M. The Effect of Sensory Integration Therapy on Occupational Performance in Children With Autism. OTJR (Thorofare N J). 2018 Apr;38(2):75–83.
107. Schoen SA, Lane SJ, Mailloux Z, May-Benson T, Parham LD, Smith Roley S, et al. A systematic review of ayres sensory integration intervention for children with autism. Autism Res. 2019 Jan;12(1):6–19.
108. Randell E, Wright M, Milosevic S, Gillespie D, Brookes-Howell L, Busse-Morris M, et al. Sensory integration therapy for children with autism and sensory processing difficulties: the SenITA RCT. Health Technol Assess. 2022 Jun;26(29):1–40.
109. Rimland B, Edelson SM. Autism treatment evaluation checklist. Journal of Intellectual Disability Research. 1999.
110. Li YQ, Sun YH, Liang YP, Zhou F, Yang J, Jin SL. Effect of probiotics combined with applied behavior analysis in the treatment of children with autism spectrum disorder: a prospective randomized controlled trial. Zhongguo dang dai er ke za zhi= Chinese journal of contemporary pediatrics. 2021 Nov 1;23(11):1103–10.
111. Mahapatra S, Khokhlovich E, Martinez S, Kannel B, Edelson SM, Vyshedskiy A. Longitudinal Epidemiological Study of Autism Subgroups Using Autism Treatment Evaluation Checklist (ATEC) Score. J Autism Dev Disord. 2020 May;50(5):1497–1508.
112. Brown C, Dunn W. Sensory Profile™ Adolescent/Adult Sensory Profile® Adolescent/Adult Sensory Profile Summary Report. Pearson, San Antonio, TX, USA; 2006.
113. Dunn W. Sensory Profile 2: User's Manual: Psych Corporation; 2014.
114. Panda PK, Sharawat IK, Saha S, Gupta D, Palayullakandi A, Meena K. Efficacy of oral folinic acid supplementation in children with autism spectrum disorder: a randomized double-blind, placebo-controlled trial. European Journal of Pediatrics. 2024 Nov;183(11):4827–35.
115. Marcilla-Jorda M, Grande C, Coelho V, Rubio-Belmonte C, Moro-Ipola M. Sensory Profile-2 in Autism Spectrum Disorder: An Analysis within the International Classification of Functioning, Disability and Health Framework. Journal of Autism and Developmental Disorders. 2025 May;55(5):1824–39.
116. Simpson K, Adams D, Alston-Knox C, Heussler HS, Keen D. Exploring the Sensory Profiles of Children on the Autism Spectrum Using the Short Sensory Profile-2 (SSP-2). J Autism Dev Disord. 2019;49(5):2069–79.
117. Dean EE, Little L, Tomchek S, Wallisch A, Dunn W. Prevalence Models to Support Participation: Sensory Patterns as a Feature of All Children's Humanity. Front Psychol. 2022;13:875972.
118. Dunn W, Brown C, Breitmeyer A, Salwei A. Construct Validity of the Sensory Profile Interoception Scale: Measuring Sensory Processing in Everyday Life. Front Psychol. 2022;13:872619.
119. Brown T, Leo M, Austin DW. Discriminant validity of the Sensory Profile in Australian children with autism spectrum disorder. Phys Occup Ther Pediatr. 2008;28(3):253–66.
120. Kientz MA, Dunn W. A comparison of the performance of children with and without autism on the Sensory Profile. Am J Occup Ther. 1997;51(7):530–7.
121. Metz AE, Boling D, DeVore A, Holladay H, Liao JF, Vlutch KV. Dunn's Model of Sensory Processing: An Investigation of the Axes of the Four-Quadrant Model in Healthy Adults. Brain Sci. 2019;9(2).
122. Enemark Larsen A, Rasmussen B, Christensen JR. Enhancing a Client-Centred Practice with the Canadian Occupational Performance Measure. Occup Ther Int. 2018;2018:5956301.
123. Larsen AE, Law M. Canadian Occupational Performance Measure the state of the art - a review. Scand J Occup Ther. 2025;32(1):2473045.
124. Ohno K, Tomori K, Sawada T, Seike Y, Yaguchi A, Kobayashi R. Measurement Properties of the Canadian Occupational Performance Measure: A Systematic Review. Am J Occup Ther. 2021;75(6).
125. Kangarani-Farahani M, Thompson-Hodgetts S, Zwicker JG. Effectiveness of Cognitive Orientation to daily Occupational Performance for autistic children with developmental coordination disorder. Dev Med Child Neurol. 2025;67(2):216–22.
126. Xu M, Fralick D, Zheng JZ, Wang B, Tu XM, Feng C. The Differences and Similarities Between Two-Sample T-Test and Paired T-Test. Shanghai Arch Psychiatry. 2017;29(3):184–8.
127. Proudfoot JA, Lin T, Wang B, Tu XM. Tests for paired count outcomes. Gen Psychiatr. 2018;31(1):e100004.
128. Hazra A, Gogtay N. Biostatistics Series Module 3: Comparing Groups: Numerical Variables. Indian J Dermatol. 2016;61(3):251–60.
129. Hedberg EC, Ayers S. The power of a paired t-test with a covariate. Soc Sci Res. 2015;50:277–91.
130. Li H, Johnson T. Wilcoxon's signed-rank statistic: what null hypothesis and why it matters. Pharm Stat. 2014;13(5):281–5.
131. Rosner B, Glynn RJ, Lee ML. The Wilcoxon signed rank test for paired comparisons of clustered data. Biometrics. 2006;62(1):185–92.
132. Politi MT, Ferreira JC, Patino CM. Nonparametric statistical tests: friend or foe? J Bras Pneumol. 2021;47(4):e20210292.
133. Ryan RL, Gianneschi G. Retrospective case control study of microcurrent therapy in autism spectrum disorder: Behavioral outcomes and dose-response analysis. Explore (NY). 2025;21(3):103156.
134. Raja R, Sharma A, Ramesh B, Ramassamy E, Esai Amudhan N, Prasath NH, et al. Comparative Evaluation of Music and Movement Therapy and Visual Pedagogy as Interventional Tools in Promoting Oral Health in Children With Autism Spectrum Disorder: A Randomized Controlled Trial. Spec Care Dentist. 2025;45(2):e70015.
135. Sawicki CM, Duran P, Hestehave S, Khanna R, Wade SD. Green light exposure in children with autism spectrum disorder: a pilot study. J Clin Pediatr Dent. 2024;48(4):99-107.
136. Narzisi A, Fabbri-Destro M, Crifaci G, Scatigna S, Maugeri F, Berloffa S, et al. Sensory Profiles in School-Aged Children with Autism Spectrum Disorder: A Descriptive Study Using the Sensory Processing Measure-2 (SPM-2). J Clin Med. 2022;11(6).
137. Liu J, Yao L, Zhang W, Xiao Y, Liu L, Gao X, et al. Gray matter abnormalities in pediatric autism spectrum disorder: a meta-analysis with signed differential mapping. Eur Child Adolesc Psychiatry. 2017;26(8):933–45.
138. Mische Lawson L, Foster L, Hamner K, Wright L. Exploring Effects of Sensory Garments on Participation of Children on the Autism Spectrum: A Pretest-Posttest Repeated Measure Design. Occup Ther Int. 2022;2022:3540271.
139. Borzoo T, Tafazoli M, Ebrahimzadeh M, Kazemi-Arpanahi H, Tabahfar R, Kamyari N, et al. The effect of aromatherapy and music therapy on blood pressure and heart rate of nursing students before taking the exam. Sci Rep. 2025;15(1):12783.
140. Sukumar M, Thanneeru SK, Sutar RF, Sahu A, Agrawal A. Effect of Music Therapy on Quality of Life in Geriatric Population: A Systematic Review and Meta-Analysis. Indian J Palliat Care. 2024;30(4):296-303.
141. Quan Y, Lo CY, Wolff L, Wang J, Olsen KN, Thompson WF. Cognitive benefits of music in aerobic exercise: Evidence from a Bayesian network meta-analysis in adults with mild cognitive impairment. Arch Gerontol Geriatr. 2025;134:105848.
142. Rabeyron T, Robledo Del Canto JP, Carasco E, Bisson V, Bodeau N, Vrait FX, et al. A randomized controlled trial of 25 sessions comparing music therapy and music listening for children with autism spectrum disorder. Psychiatry Res. 2020;293:113377.
143. Geretsegger M, Holck U, Gold C. Randomised controlled trial of improvisational music therapy's effectiveness for children with autism spectrum disorders (TIME-A): study protocol. BMC Pediatr. 2012;12:2.
144. Feng H, Mahoor MH, Dino F. A Music-Therapy Robotic Platform for Children With Autism: A Pilot Study. Front Robot AI. 2022;9:855819.
145. Mathur A, Vijayakumar SH, Chakrabarti B, Singh NC. Emotional responses to Hindustani raga music: the role of musical structure. Front Psychol. 2015;6:513.
146. Kaufmann W. Rasa, rāga-mālā and performance times in North Indian rāgas. Ethnomusicology. 1965;9(3):272–91.
147. Deepa K. Aesthetics of music—a detailed study on ragas therapy. JETIR. 2020;7(4):1823–9.
148. Veerabrahmachar R, Bista S, Bokde R, Jasti N, Bhargav H, Bista S. Immediate Effect of Nada Yoga Meditation on Energy Levels and Alignment of Seven Chakras as Assessed by Electro-photonic Imaging: A Randomized Controlled Crossover Pilot Study. Adv Mind Body Med. 2023;37(1):11–6.
149. Robertson CE, Kravitz DJ, Freyberg J, Baron-Cohen S, Baker CI. Tunnel vision: sharper gradient of spatial attention in autism. J Neurosci. 2013;33(16):6776–81.
150. Leadbeater C. The Chakras. Second Edition ed. Judith A, editor. Theosophical Publishing House, Wheaton, IL, USA: Quest Books; 2013. pp. 63–153.