Abstract
Susac syndrome is a rare autoimmune endotheliopathy characterized by encephalopathy, branch retinal artery occlusion (BRAO), and sensorineural hearing loss (SNHL). Due to its diverse presentation, it is often misdiagnosed. We report a case of a 22-year-old female with progressive neurological symptoms, hearing loss, and cognitive dysfunction. Brain MRI revealed characteristic corpus callosum “snowball” lesions, and further investigations confirmed BRAO and SNHL. CSF analysis showed elevated protein with negative oligoclonal bands. Notably, the patient tested positive for anti-GAD65 antibodies, a finding with unclear significance in Susac syndrome. The patient was treated with high-dose corticosteroids, IVIG, and Rituximab, leading to significant neurological improvement and partial recovery of auditory function. This case highlights the importance of early recognition and aggressive immunotherapy in Susac syndrome, as well as the need for further research into the role of anti-GAD65 antibodies in this disease.
Keywords
Susac Syndrome, GAD-65, Autoimmune Endotheliopathy, Case Report
Introduction
Susac syndrome is a rare autoimmune endotheliopathy that affects the microvasculature of the brain, retina, and inner ear [1,2]. It is characterized by a clinical triad of encephalopathy, branch retinal artery occlusion (BRAO), and sensorineural hearing loss (SNHL) [1,3]. First described by John Susac in 1979, the syndrome is believed to result from an autoimmune-mediated injury to the endothelium of small blood vessels, leading to microinfarctions in the central nervous system, retina, and cochlea [1–4].
The true incidence and prevalence are unknown, with a predominance observed among young female patients [2]. Due to its rarity and overlapping clinical and radiological features, Susac syndrome is frequently misdiagnosed as multiple sclerosis, central nervous system vasculitis, or other autoimmune or demyelinating disorders [1,2,4]. While the underlying pathogenesis is thought to involve immune-mediated endothelial dysfunction [1,5], the presence of anti-GAD65 antibodies in this disease has not been previously described. Early recognition is critical, as delayed diagnosis and treatment can lead to irreversible neurological, visual, and auditory damage [1,6]. Characteristic MRI findings, including corpus callosum “snowball” lesions, are key diagnostic markers that help differentiate Susac syndrome from other conditions [1,2].
The presence of anti-GAD65 antibodies in autoimmune diseases is well-documented, particularly in neurological conditions such as stiff-person syndrome, cerebellar ataxia, and limbic encephalitis [7,8]. However, its association with Susac syndrome has not been previously reported [7,8]. This case presents a young female diagnosed with Susac syndrome with a positive anti-GAD65 antibody test, raising questions about its potential role in disease pathogenesis [7–9]. Whether this antibody contributes to endothelial dysfunction in Susac syndrome or represents an incidental finding remains uncertain [7–9].
This report highlights the diagnostic challenges associated with Susac syndrome, underscores the importance of early recognition, and discusses the potential implications of anti-GAD65 antibody positivity in this condition [1,7–9]. The positive anti-GAD65 antibody finding is a novel observation in this disease and raises questions about its potential role in autoimmune-mediated microvascular injury. While anti-GAD65 antibodies are well-documented in conditions like stiff-person syndrome and autoimmune cerebellar ataxia, their significance in Susac syndrome is unknown and requires further investigation.
The patient’s response to high-dose corticosteroids, IVIG, and Rituximab supports the current evidence favoring aggressive immunosuppressive therapy in Susac syndrome. Additionally, MRI venography findings suggested cerebral venous sinus thrombosis (CVST), which led to anticoagulation therapy, further highlighting the need to consider thrombotic mechanisms in select cases of Susac syndrome.
Case Presentation
A 22-year-old female with no significant prior medical history, presented with progressive neurological symptoms. She developed cognitive dysfunction, personality changes, and disorientation, accompanied by recurrent vomiting, headaches, fatigue, and hearing loss. Her family history was significant for an older sister who had a stroke at age 36 and a history of deep vein thrombosis (DVT). She was married and had two children.
Brain MRI revealed characteristic corpus callosum “snowball” lesions, while fluorescein angiography confirmed BRAO. Audiometric testing identified sensorineural hearing loss, and cerebrospinal fluid (CSF) analysis showed elevated protein with negative oligoclonal bands. Notably, anti-GAD65 antibodies were detected, a finding with uncertain significance in Susac syndrome. Given the diagnostic complexity and the progressive nature of symptoms, the patient was started on high-dose corticosteroids, intravenous immunoglobulin (IVIG), and Rituximab. She showed significant neurological improvement and partial recovery of hearing and vision, with follow-up imaging demonstrating regression of corpus callosal lesions.
Clinical Examination
On initial evaluation, the patient was alert but disoriented, with a Glasgow Coma Scale (GCS) score of 15/15. The neurological examination was limited by poor cooperation, which restricted a full assessment, particularly of motor and sensory function. Mental status examination revealed inattentiveness, low mood, impaired comprehension, and difficulty with repetition. Formal standardized cognitive testing (such as MoCA or MMSE) could not be performed due to the patient’s limited cooperation. Cranial nerve examination was unremarkable. Gait assessment demonstrated unsteadiness requiring assistance. No signs of meningeal irritation were observed.
The patient’s presentation of acute cognitive decline, disorientation, and hearing loss raised suspicion for an autoimmune or inflammatory neurological disorder. Given her history and examination findings, an extensive workup was performed to identify the underlying cause of her symptoms.
Diagnostic Workup
Neurophysiological and laboratory findings
Visual Evoked Potential (VEP): Bilateral delayed P100 waves, suggestive of optic pathway dysfunction.
Electroencephalography (EEG): Mild diffuse slowing, indicating cerebral dysfunction.
Pure Tone Audiometry (PTA): Right Ear: Mild sensorineural hearing loss (SNHL). Left Ear: Severe rising to moderate SNHL.
Cerebrospinal Fluid (CSF) Analysis: Elevated protein concentration. Negative oligoclonal bands.
Autoimmune Panel: Positive for Anti-GAD65 antibodies. Negative for Antinuclear antibodies (ANA), antiphospholipid antibodies, and vasculitis markers.
The presence of elevated CSF protein without oligoclonal bands, in combination with the delayed VEP response and EEG abnormalities, suggested an inflammatory or autoimmune-mediated neurological condition. Given these findings, further imaging studies were performed to assess structural abnormalities or microvascular pathology.
Magnetic Resonance Imaging (MRI) of the Brain shows multiple hyperintense T2 lesions in the corpus callosum with characteristic “snowball” appearance. DWI shows diffusion restriction in the centrum semiovale and parietal lobes, indicative of microinfarcts.
Magnetic Resonance Venography (MRV) shows small filling defect in the proximal straight sinus, raising suspicion of cerebral venous thrombosis (CVT).
Fluorescein Angiography shows evidence of branch retinal artery occlusion (BRAO).
Computed Tomography (CT) of the Chest, Abdomen, and Pelvis show no evidence of systemic malignancy or inflammatory pathology.
The MRI findings of corpus callosal lesions, particularly their “snowball” appearance, were highly suggestive of Susac syndrome. The concurrent presence of BRAO on fluorescein angiography further supported this diagnosis. However, the positive anti-GAD65 antibodies raised an additional question regarding their potential role in this presentation.
|
Test |
Flag |
Value |
Reference |
|
GAD65 Ab Assay, S |
High |
0.13 nmol/L |
≤ 0.02 nmol/L |
|
Volume |
- |
6.000 mL |
- |
|
CSF Appearance |
- |
Clear |
- |
|
CSF Color |
- |
Colorless |
- |
|
CSF-Cell Count WBC |
Normal |
0.0 |
0.0 - 5.0 |
|
CSF-Cell Count RBC |
High |
13.0 |
0.0 - 0.0 |
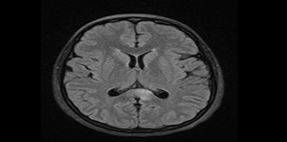
Figure 1. 06-Nov-2023 MRI: FLAIR/T2 shows ill-defined hyperintense lesions in the posterior corpus callosum.
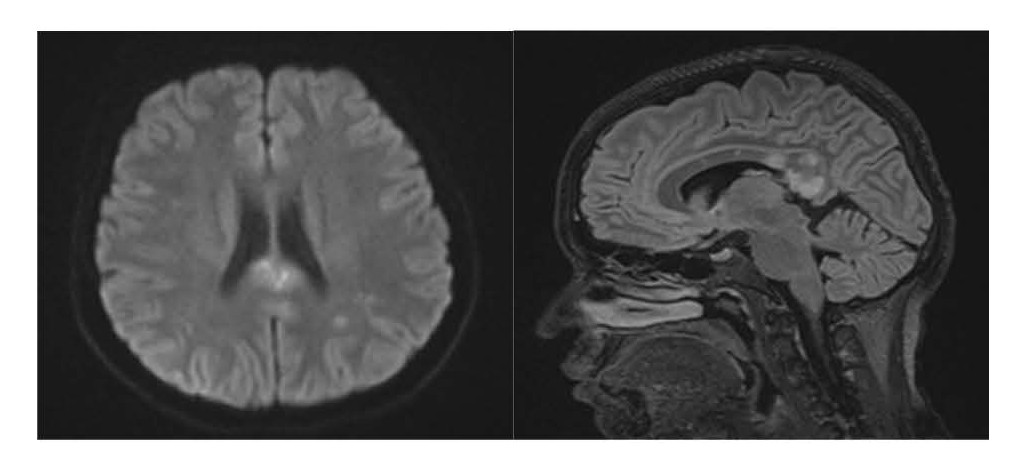
Figure 2. 07-Nov-2023 MRI: (1) DWI shows another diffusion restriction in centrum semiovale and parietal lobes suggested microinfarcts in white matter. (2) Sagittal FLAIR/T2 hyperintense lesions in the corpus callosum characterized by snowball-like lesion.
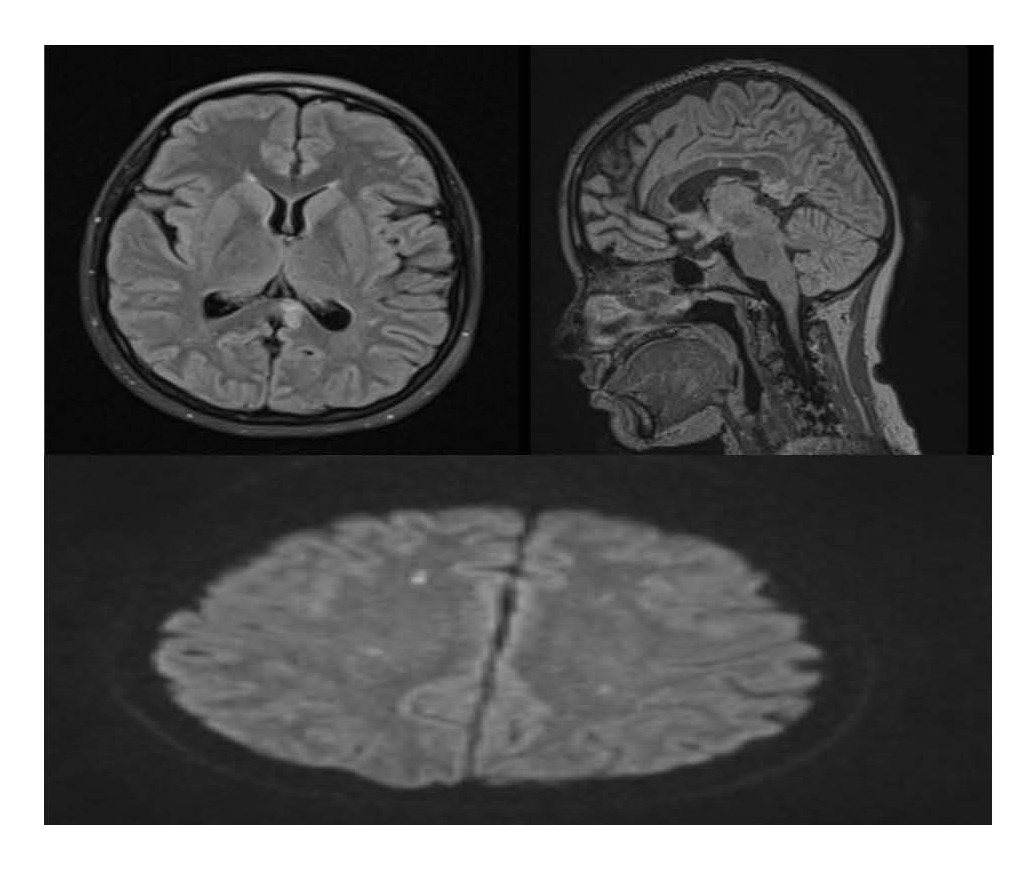
Figure 3. 21-Nov-2023 MRI: (1,2) Axial/Sagittal FLAIR/T2 shows regression in lesion size (indicating partial recovery), (3) also on DWI there are multiple tiny diffusion restrictions (microinfarcts).
Differential diagnosis
Susac syndrome was considered the most probable diagnosis based on the combination of clinical and radiological findings. Other differential diagnoses included central nervous system (CNS) vasculitis, acute disseminated encephalomyelitis (ADEM), multiple sclerosis (MS), and cerebral venous sinus thrombosis (CVST).
Susac syndrome is frequently misdiagnosed due to its variable clinical presentation and overlapping features with other autoimmune and demyelinating diseases. This diagnostic difficulty is not unique to Susac syndrome but is a common theme in managing rare neurological conditions. For instance, similarly complex and delayed diagnoses have been reported in cases of cerebellar hemangioblastoma, where non-specific symptoms like chronic headaches can lead to a prolonged diagnostic odyssey with potentially severe consequences, underscoring the universal challenge of diagnosing rare CNS pathologies [10]. The patient’s presentation of neurological dysfunction, hearing loss, and retinal artery involvement, in conjunction with snowball lesions in the corpus callosum, strongly favored Susac syndrome over other differential diagnoses. The absence of oligoclonal bands made multiple sclerosis less likely, while the negative vasculitis panel and the lack of systemic inflammatory signs reduced the likelihood of CNS vasculitis. ADEM was considered but was less consistent with the chronic nature of the symptoms. CVST was also considered but was not supported by imaging findings.
Therapeutic Interventions
Given the clinical and radiological evidence of Susac syndrome, the patient was started on an aggressive immunosuppressive regimen with Rituximab 1000 mg IV, administered in two doses 14 days apart, followed by maintenance every 6 months, it was chosen due to its efficacy in autoimmune diseases and its role in cases refractory to steroids and IVIG. This was combined with intravenous methylprednisolone (1 g/day for 5 days), followed by oral prednisone taper over six weeks. In addition, she was also given Intravenous Immunoglobulin 22 g/day IV for 5 days.
Anticoagulation Therapy, Warfarin was initiated due to the MRI venography findings suggesting venous sinus thrombosis. It has been titrated based on international normalized ratio (INR) levels (target level 2-3), Enoxaparin: 60 mg subcutaneous injection twice daily for 30 days as bridging therapy.
Audiological management includes intratympanic steroid injections by three dexamethasone injections in the left ear over multiple weeks, leading to partial improvement in hearing.
Supportive therapy with paracetamol for pain and metoclopramide for nausea control.
The patient tolerated the treatment well, with no reported complications. Throughout hospitalization, she showed progressive neurological improvement, which was further confirmed on follow-up imaging and clinical assessment
Outcome and Follow-up
Neurological Status shows marked improvement in cognitive function and resolution of disorientation. Her follow-up MRI showed regression of corpus callosal lesions. Audiological reassessment shows residual mild sensorineural hearing loss persisted despite treatment. Ophthalmological reassessment shows persistent BRAO with no further deterioration. Coagulation Monitoring and Warfarin therapy continued, with future reassessment planned based on disease activity.
Rheumatology follow up for further evaluation to determine ongoing autoimmune activity before discontinuation of anticoagulation therapy.
The patient was discharged with close neurology, ophthalmology, and audiology follow-up. At her three-month follow-up, she remained neurologically stable, with no evidence of disease progression. Her hearing function showed partial recovery, and she was scheduled for repeat another audiometric assessment.
Discussion
Pathophysiology and clinical considerations
Susac syndrome is a rare autoimmune microangiopathy affecting the brain, retina, and cochlea, leading to the classic clinical triad of encephalopathy, branch retinal artery occlusion (BRAO), and sensorineural hearing loss (SNHL) [1–3]. The underlying pathophysiology is thought to involve an immune-mediated injury to the endothelial cells of small blood vessels, causing microvascular occlusion and subsequent ischemic damage to affected organs [1,2]. Our case aligns with previous reports describing the characteristic MRI findings of corpus callosum “snowball” lesions, which are considered highly specific for Susac syndrome [1,5]. Additionally, the presence of BRAO and progressive sensorineural hearing loss further solidified the diagnosis [1,2]. However, this case introduced a unique finding: the presence of anti-GAD65 antibodies, which has not been previously associated with Susac syndrome [1,2].
The role of anti-GAD65 antibodies
Anti-GAD65 antibodies have been implicated in a range of autoimmune neurological disorders, including Stiff-person syndrome, Autoimmune cerebellar ataxia, Limbic encephalitis, type 1 diabetes mellitus [7,8].
To date, no direct link between Susac syndrome and anti-GAD65 antibodies has been established. The detection of these antibodies in our patient raises two possible hypotheses. 1) An incidental finding unrelated to the disease process, reflecting an underlying predisposition to autoimmunity. 2) A potential pathogenic role, contributing to endothelial dysfunction or inflammatory microangiopathy in Susac syndrome.
Further research is needed to determine whether anti-GAD65 antibodies play a role in the immune dysregulation seen in Susac syndrome.
Assay methods for detecting anti-GAD65 antibodies
The detection of anti-GAD65 antibodies is a crucial diagnostic marker for autoimmune neurological syndromes. Various immunoassay techniques are utilized for their detection, differing in sensitivity, specificity, and quantitative measurement capabilities. Given the unexpected presence of anti-GAD65 antibodies in this case of Susac syndrome, it is essential to evaluate both the methodology of detection and the clinical significance of the result [8].
Anti-GAD65 antibodies were detected using a radioimmunoassay at a low titer; therefore, their clinical relevance in this case remains uncertain. Further evaluation, including cerebrospinal fluid analysis and confirmatory testing such as immunoblot assays, may help clarify whether this finding represents a coincidental marker of autoimmune susceptibility rather than a pathogenic contributor to Susac syndrome [8].
Diagnostic challenges and differential diagnosis
Susac syndrome is frequently misdiagnosed due to its variable clinical presentation and overlapping features with other autoimmune and demyelinating diseases. In this case, multiple sclerosis (MS), CNS vasculitis, acute disseminated encephalomyelitis (ADEM), and cerebral venous sinus thrombosis (CVST) were all considered in the differential.
Multiple Sclerosis (MS): often presents with demyelinating lesions in periventricular white matter, while Susac syndrome primarily affects the corpus callosum. Oligoclonal bands are typically positive in MS but were absent in our patient. BRAO is not a feature of MS, making this diagnosis less likely.
CNS Vasculitis: While CNS vasculitis can present with encephalopathy and multifocal ischemic lesions, MRI features of Susac syndrome (snowball lesions) are distinct. The negative vasculitis panel and lack of systemic inflammatory markers further ruled out CNS vasculitis.
ADEM (Acute Disseminated Encephalomyelitis): ADEM is an immune-mediated demyelinating disease that typically follows an infection or vaccination. Unlike Susac syndrome, ADEM does not involve BRAO or progressive SNHL.
Cerebral Venous Sinus Thrombosis (CVST): The MRI venography findings of straight sinus thrombosis initially raised concerns for CVST.
Given the co-occurrence of BRAO and SNHL, Susac syndrome remained the most likely diagnosis, and anticoagulation was initiated alongside immunosuppressive therapy.
Therapeutic approach and management considerations
Early aggressive immunosuppressive therapy is essential in Susac syndrome to prevent irreversible neurological and sensory deficits. Our patient was treated with high-dose corticosteroids, intravenous immunoglobulin (IVIG), and Rituximab, which led to significant clinical improvement.
Corticosteroids remain the first-line therapy, reducing inflammation and endothelial injury.
IVIG is used as an adjunct in severe cases to modulate immune responses and prevent disease progression.
Rituximab, a B-cell–depleting agent, was chosen due to its efficacy in autoimmune diseases and its role in cases refractory to steroids and IVIG.
The decision to initiate anticoagulation therapy was made due to the MRI venography findings suggesting venous sinus thrombosis. While thrombosis is not a classic feature of Susac syndrome, some reports suggest a hypercoagulable state in autoimmune vasculopathies. Future studies are needed to assess whether routine anticoagulation should be considered in selected cases.
Long-term prognosis and follow-up
Susac syndrome typically follows either a monophasic or relapsing-remitting course. Potential long-term complications include: Persistent cognitive impairment due to recurrent microvascular insults. Permanent hearing loss if inner ear ischemia is severe. Residual visual deficits from BRAO leading to scotomas.
In this patient, neurological symptoms improved significantly, with partial recovery of hearing and stable ophthalmological findings. Given the positive response to Rituximab, maintenance therapy every six months is planned to reduce the risk of relapse.
Limitation
This case report only discusses a single patient; the findings of this case cannot be generalized. The patient had low levels of anti-GAD65 antibodies, at this stage it’s unclear whether this is clinically significant or simply an incidental sign of underlying autoimmunity.
Conclusion
This case highlights the diagnostic and therapeutic complexities of Susac syndrome and introduces the novel finding of anti-GAD65 antibody positivity. The key takeaways from this case include: 1) Early recognition of Susac syndrome is critical to prevent irreversible neurological and sensory deficits. 2) Anti-GAD65 antibodies may have an unexplored role in autoimmune microangiopathies, warranting further research. 3) MRI “snowball lesions” in the corpus callosum remain a hallmark feature, aiding differentiation from multiple sclerosis and CNS vasculitis. 4) Immunosuppressive therapy, particularly Rituximab and IVIG, plays a crucial role in disease control and long-term stability. 5) The presence of cerebral venous thrombosis (CVT) on imaging raises questions about the role of anticoagulation therapy in Susac syndrome, which needs further exploration.
This case contributes to the growing understanding of atypical presentations of Susac syndrome and underscores the importance of multidisciplinary management and individualized treatment strategies.
Acknowledgements
The authors would like to thank the Neurology and Internal Medicine Departments at Security Forces Hospital, Riyadh, for their support in the evaluation and management of this case. Special thanks to the Radiology and Ophthalmology teams for their contribution to the diagnostic process.
Conflict of Interest Statement
None declared.
Funding
No funding was obtained for this study.
Consent for Publication
Written consent for publication was obtained from the patient.
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Author Contributions
Anas Mohammed Alamri – Primary Consultant, Supervision, Validation, Project Administration.
Omar Sanad Aldhafeeri – Visualization, Writing, Review & Editing, Software.
Abdullah Helal Almotairi – Conceptualization, Methodology.
Jehad Ahmed Alabdulminaim – Resources, Writing Original Draft, Formal Analysis.
Abdulrahman Abullah Aljebreen – Investigation, Data Curation.
Farah Fayez Alfayez – Investigation, Data Curation, Corresponding author.
References
2. Dörr J, Krautwald S, Wildemann B, Jarius S, Ringelstein M, Duning T, et al. Characteristics of Susac syndrome: a review of all reported cases. Nat Rev Neurol. 2013 Jun;9(6):307–16.
3. Susac JO, Hardman JM, Selhorst JB. Microangiopathy of the brain and retina. Neurology. 1979 Mar;29(3):313–6.
4. Rennebohm RM, Lubow M, Rusin J, Martin L, Grzybowski DM, Susac JO. Aggressive immunosuppressive treatment of Susac's syndrome in an adolescent: using treatment of dermatomyositis as a model. Pediatr Rheumatol Online J. 2008 Jan 29;6:3.
5. Hardy TA, Garsia RJ, Halmagyi GM, Lewis SJ, Harrisberg B, Fulham MJ, et al. Tumour necrosis factor (TNF) inhibitor therapy in Susac's syndrome. J Neurol Sci. 2011 Mar 15;302(1-2):126–8.
6. Mateen FJ, Zubkov AY, Muralidharan R, Fugate JE, Rodriguez FJ, Winters JL, et al. Susac syndrome: clinical characteristics and treatment in 29 new cases. Eur J Neurol. 2012 Jun;19(6):800–11.
7. Saiz A, Blanco Y, Sabater L, González F, Bataller L, Casamitjana R, et al. Graus F. Spectrum of neurological syndromes associated with glutamic acid decarboxylase antibodies: diagnostic clues for this association. Brain. 2008 Oct;131(Pt 10):2553–63.
8. Höftberger R, Bien CG, Schwarz O, et al. Glutamic acid decarboxylase antibodies in neurological disorders. Neurology. 2012 Sep 4;79(10):1070–5.
9. Greco A, De Virgilio A, Gallo A, Fusconi M, Turchetta R, Tombolini M, et al. Susac's syndrome--pathogenesis, clinical variants and treatment approaches. Autoimmun Rev. 2014 Aug;13(8):814–21.
10. Kanani J. Diagnostic challenges in hemangioblastoma: lessons from a rare case presentation. Egypt J Neurosurg. 2025 Aug 22;40(1):115.
