Abstract
Arabinoxylans (AXs) are the major structural cell wall component of various cereal brans. As an important dietary fiber source, AXs contain ferulic acids, proteins and other functional ester compounds. These functional components offer AXs well-established health benefits, such as prebiotic effect, antioxidant property, antitumor activity and immunomodulatory property. In addition, the presence of ferulic acids could trigger the covalent cross-linking of AX molecules under oxidative conditions, and therefore AX could be used as a promising functional biomaterial for fabrication of hydrogels. The formed covalent gels are resistant to harsh digestive condition, which could be developed as colon-targeted delivery system for food and biomedical application. This article aims to summarize the extraction methods of wheat arabinoxylans, discuss the covalent gelling mechanism of AX, and review the potential application of AX in food and biomedical industries.
Keywords
Wheat arabinoxylans, Extraction methods, Gelling mechanism, Colon-target delivery system
Introduction
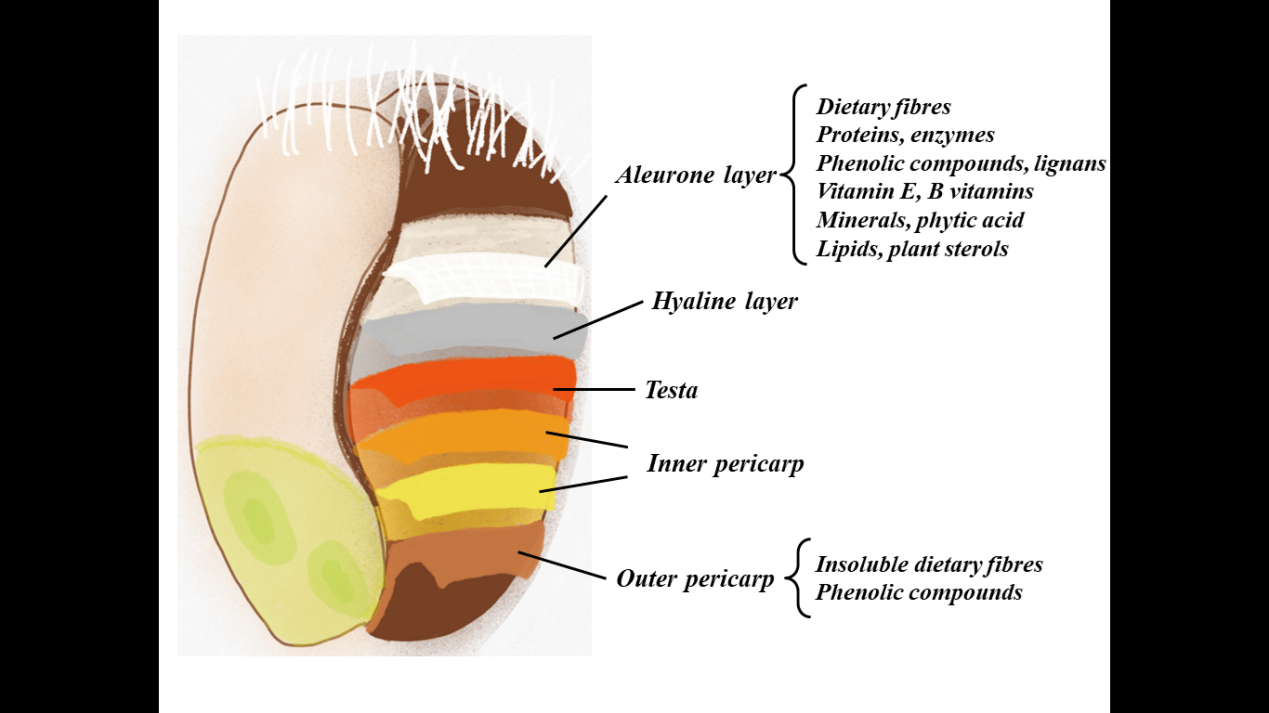
Wheat bran is an abundant industrial by-product of wheat milling operation, about 10~25% bran is inevitably produced during the processing of wheat products. It has been reported that the annual output of wheat bran could reach 3000 tons in China, and it has exceeded 200 million tons from a global perspective [1]. Most wheat brans (about 85%) are used for winemaking, animal feed and papermaking, and the utilization of deep process and high-efficiency industries are rare [2]. As shown in Figure 1, wheat bran is composed of outer and inner pericarp, testa, hyaline layer and aleurone layer. The inner and outer pericarps are composed of xylans, lignin and cellulose [3]. High contents of dietary fiber (around 50%) and varied phytochemicals (e.g. vitamins, phenolic acids, minerals) in the bran and aleurone make wheat bran have high health benefits [4]. In addition, wheat bran is abundant, low-cost natural source, and therefore wheat bran has a great potential for the development of novel healthy food products.
Figure 1: Wheat grain, showing consists of wheat bran. Adapted from Brouns et al. (2012).
Wheat bran arabinoxylan is an important dietary fiber, accounting for more than 80% of wheat non-starch polysaccharides. It has been reported that AX can be selectively degraded in the intestinal tract to produce arabinoxylan oligosaccharides (AXOS), short-chain fatty acids (SCFA), and other health-promoting bioactive factors, which is beneficial for promoting the growth of intestinal microorganisms [4,5]. The presence of functional groups makes AXs possess health-promoting benefits, such as enhancing body’s immune system [6], regulating the lipid and glucose metabolism and antitumor effects [7]. The viscosity of AX can affect their fat-binding capacity and easily improve the postprandial blood glucose response [8-9]. The presence of ferulic acid on the side chain of AXs make them a natural antioxidant and excellent immune system enhancer [10]. Overall, there is a huge application potential of AXs in food, cosmetic, and medical industries.
In addition to bioactive functions, arabinoxylans have some unique physicochemical properties, such as rheological properties, water holding capacity and oxidative cross-linking (gelling) properties. It is worth noting that the extraction methods of AXs have a significant effect on their fine structure and subsequent functional properties, and suitable extraction methods are necessary for obtaining arabinoxylan with desired properties. This article aims to summarize the extraction methods of wheat bran arabinoxylan and compare their structural difference. More importantly, the gelling property of AXs has been discussed to provide the theoretic basis for broadening their application in varied fields.
Isolation of Wheat Arabinoxylans
Arabinoxylans have a complex structure, consisting of a linear skeleton of β-(1→4)-linked D-xylopyranosyl (Xylp) units, through the O-3 of xylose residues and/or O-2,3 position is connected with α-L-arabinofuranosyl substituent (Araf). Due to the diverse forms of xylose residues at O-2 and O-3 positions, they may be unsubstituted (uX), monosubstituted (mX) or disubstituted (dX) [11], with a small amount of α-D-glucuronic acid or 4-O-methyl-glucuronic acid, galactose pyranose, and xylose pyranose present on the side chain of AXs. Generally, arabinoxylans are divided into water-extractable arabinoxylan (WEAX) and water unextractable arabinoxylan (WUAX). WEAX accounts for only a minority in wheat bran, which is about 6% of the total AX content [12]. WUAX could interact with protein, lignin and other components via hydrogen bonds and covalent ether bonds, making it insoluble in water and more difficult to extract from wheat bran [2,13].
The extraction method could directly affect the monosaccharide composition, molecular weight and substitution degree of AXs. As shown in Table 1, the molecular weight of wheat bran arabinoxylans ranges from 10 kDa to 10,000 kDa by using different extraction methods under different conditions [2,14-18]. The substitution degree of AX is expressed by the ratio of arabinose to xylose (Ara/Xyl). The A/X ratios of wheat bran arabinoxylan are approximately in the range of 0.4-0.9 [2,14-18]. In addition, the ethanol concentration also affects the monosaccharide composition of AX. According to previous study, wheat brans underwent 20%-80% ethanol fractionation treatment could obtain AX fractions with Ara/Xyl ratios ranging from 0.31 to 0.85 [19].
|
Source |
Pretreatment |
Extraction condition |
Separation and purification condition |
Yield (%) |
Total AX (%) |
Mw (kDa) |
A/X ratio |
Reference |
|
|
Water unextractable solids of wheat bran |
Destarch |
1 M NaOH (1:10, w/v) |
3 volumes of ethanol precipitation |
54.0 |
≈69 |
76 |
0.69 |
Li et al., 2020 [2] |
|
|
Wheat bran |
Destarch |
0.5 M NaOH, 150 rpm, 4 h (1:6, w/v) |
3 volumes of ethanol precipitation |
3.7- 5.6 |
68-72 |
1250-1490/3.57-3.83 |
0.46- 0.54 |
Wang et al., 2019 [14] |
|
|
Water unextractable solids of wheat bran |
Destarch |
0.5 M Ba(OH)2, 16 h (1:10, w/v) |
3 volumes of ethanol precipitation, |
48.0 |
0.36 |
70 |
0.78 |
Li et al., 2020 [2] |
|
|
Wheat bran |
Destarch |
Endoxylanase, 55°/24h hydrothermal treatment
|
4 volumes of ethanol precipitation |
19.5 |
62.5 |
12/160 |
0.52 |
Aguedo et al., 2014 [15] |
|
|
Wheat bran |
Destarch |
RuCl3/Al-MCM-48/180° C |
—— |
—— |
78.0 |
9 |
0.53 |
Sánchez-Bastardo et al., 2017 [16] |
|
|
Wheat bran |
Destarch |
Endoxylanase, 55 ° C/24 h vacuum filtration |
membranes ultrafiltration |
4.2- 4.3 |
77.8- 78.3 |
5/12.5 |
0.68- 0.73 |
Aguedo et al., 2014 [15] |
|
|
Wheat bran |
Destarch, deprotein |
Sunzymes, 180 W ultrasound bath |
—— |
14.3 |
0.62 |
—— |
—— |
Wang et al., 2014 [17] |
|
|
Wheat bran |
—— |
Electrostatic separation |
50μm/4000Pa air jet sieving |
8.0 |
0.43 |
—— |
—— |
Wang et al.2015 [18] |
|
|
Water unextractable solids of wheat bran |
Destarch |
160° C, Subcritical water extraction |
3 volumes of ethanol precipitation |
—— |
≈66.0 |
39 |
0.52 |
Li et al., 2020 [2] |
Extraction process of AXs includes raw material pretreatment, extraction, separation, and purification and final freeze-drying treatment. After milling and sieving process, wheat brans are treated with heat-stable α-amylase to remove the starch. The current extraction methods are roughly divided into chemical treatment, enzyme treatment and physical treatment. The most common extraction method is the alkaline method, which has better selectivity and higher extraction yield. Zhou et al. found that the peroxide-assisted alkaline extraction was 50% more effective than enzymatic treatment for AX extraction [20]. Alkaline treatment could improve the solubilization of hemicellulose, but a stronger alkaline solution treatment would destroy some functional groups of AX. For example, hydroxycinnamic acid and acetic acid could partially or completely disappear after alkaline treatment, and the esterification degree of the fraction is lower [21,22]. The physical extraction technologies, including extrusion, ball milling, steam, ultrasonic treatment, subcritical water extraction, microwave treatment [19,23-25], could be used for AX extraction. Mechanical processes are efficient, while the energy input required for the equipment is high and the reaction conditions are strict. Hell et al. have compared the difference between several extraction methods including the alkaline extraction method, ball milling method and extrusion method [23]. It has been reported that hydrogen peroxide has the highest solubilization effect on AX, increasing by 105%. Although the ball milling method results in low yield, it has high specificity to AX and the extraction process is green. As for the extrusion method, it has high selectivity but low extraction rate. Overall, the ball milling tends to be the best method. Recently, subcritical water extraction (SWE) had drawn researchers’ much attention, due to its green process and high extraction efficiency [24]. It has been stated that AX extracted by SWE method owns higher molecular weight and more ferulic acid content, which is beneficial for enhancing the cross-linking ability of AX molecules [2]. The enzymatic method is more environmentally friendly than the chemical method, while the enzymatic reaction requires a specific and strict environment. In addition, the high price of enzymes requires a constant search for alternatives [23]. Since each method has its pros and cons, in future, more efforts should be focused on to explore novel extraction technique, in order to enhance the extraction efficiency, improve the quality of extracted arabinoxylan, reduce the cost and guarantee the green extraction process.
Gelation Mechanism of Wheat Arabinoxylans
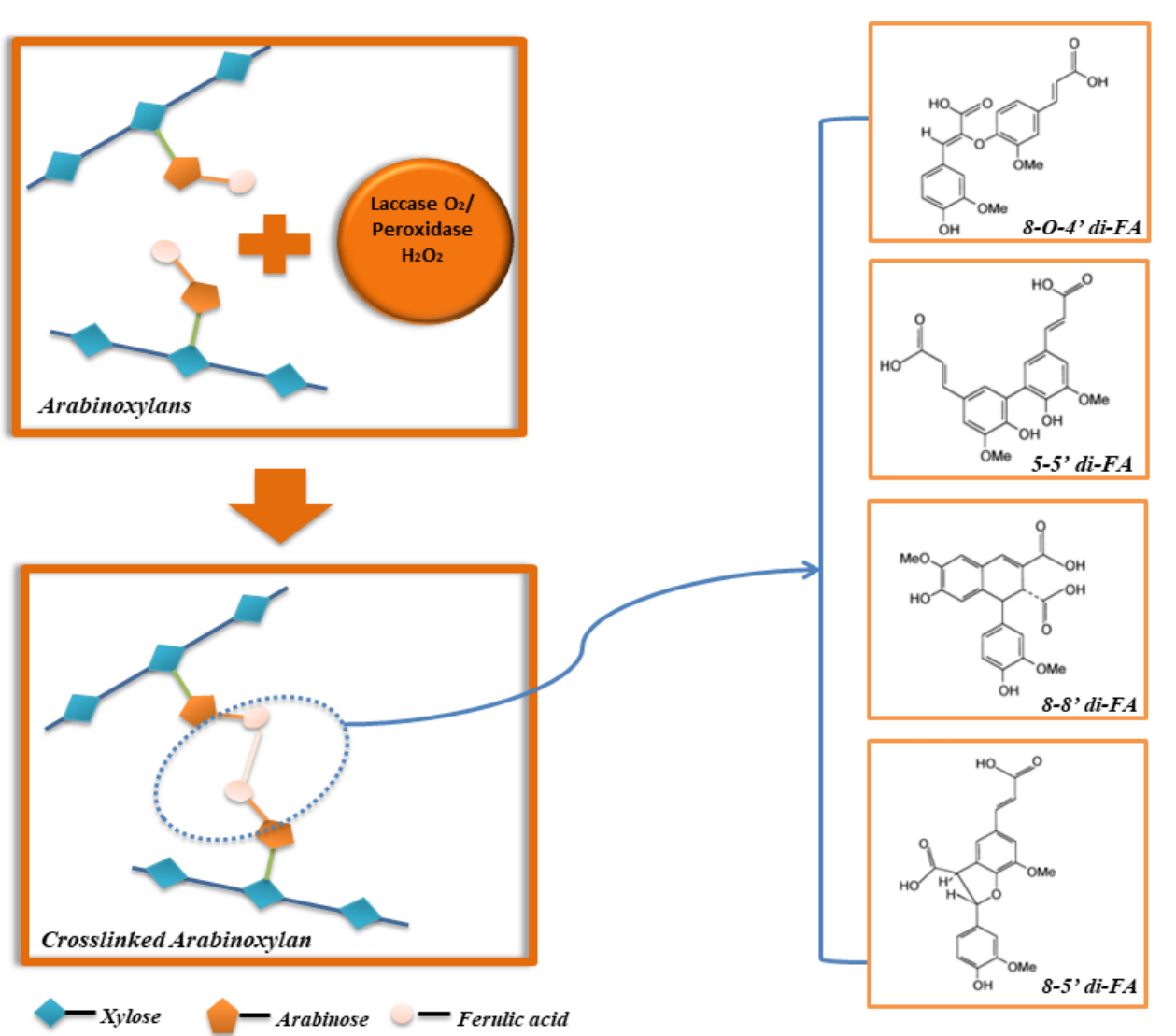
AX with ferulic acid can form gel under the action of oxidizing agents such as laccase, peroxidases, hydrogen peroxide. In the early stage of AX gelation, ferulic acids are oxidized and gradually disappeared, transforming into ferulic acid dimers (di-FA), and ferulic acid trimers (tri-FA). The oxidant could attack the hydrogen atoms of the hydroxyl group on the ferulic acid ring to obtain a resonance-stable (C-4, C-5, C-8) phenoxy group, and then the unpaired electron coupling of free radicals (phenoxy/alkoxy) forms the covalent bond to connect two adjacent arabinoxylan chains, contributing to the gelation of AX molecules [26,27]. As shown in Figure 2, five forms of ferulic acid dimers including 5-5', 8-5' benzo, 8-8', 8-O-4', 8-5' could be generated during AX gelation [28]. In addition, during laccase coupling reaction, the conformation of AX converts into a more ordered and closer-packing structure with higher degree of crystallinity as well as an increased molecular weight. During the process of O2/laccase-induced cross-linking of wheat AX gelation, 8-5' accounts for 80%, and tri-FA in the form of 4-O-8' and 5'-5''-dehydroferric acids are also present [26]. Han et al. reported that after 2 h of gelation, around 70%-96% esterified FA were oxidized [29]. The eFAs were transformed into 8-5' Benzofuran dimers, 8-O-4' and 8-5' diFA. It was worth noting that the covalent cross-linking process of AX mainly depended on the AX structure, especially the position and contents of FA [30]. A high proportion of 5-5’ di-FA in the AX gel could lower the effective elasticity and further affected the gel performance [31]. Zhang et al. found that the increase in contents of decarboxylated 8-5' di-FA favored the formation of AX gels [32]. Overall, there is insufficient research investigating the influence of formed di-FA and tri-FA on the gel performance of AX gel, which will be an important research subject on elucidating the gelling mechanism of AX in future studies.
Figure 2: The transformation of FA into various di-FA during covalent crosslinking of AX.
In fact, the gelling process of AX is a complex system, and it depends on several factors including the molecular weight, concentration, ferulic acids, A/X ratios and so on [2,14,27,33-35]. As shown in Table 2, AX with different structural properties exhibit varied gelling properties. Generally, arabinoxylans with higher molecular weight (Mw), ferulic acids and purity are easier to form hydrogels. Berlanga-Reyes et al. found that prolonged alkaline extraction time could reduce the FA contents and A/X ratios, and thereby hampering the cross-linking ability of extracted AX and reducing the prepared gels’ hardness [32]. More recent studies by Wang et al. [14] and Han et al. [29] also confirmed that AX with higher FA contents and Mw showed a higher oxidative cross-linking property under the action of laccase, and the obtained gels had denser network structure and higher viscoelastic modulus.
|
Source |
Phenolic acid profile |
AX content (%) |
Oxidation system |
G’/G’’ (Pa) /tanδ |
Mw (kDa) |
A/X ratio |
Reference |
|
|
FA content (μg/mg ) |
Diferulic acids (di-FA) |
|||||||
|
Wheat bran AX |
2.17 |
8-O-4'/8-5' |
6 |
Laccase |
9800-10000/2200-5000/≤0.8 |
—— |
—— |
Khalighi et al., 2019 [26] |
|
Wheat bran AX |
6.22-7.39 |
—— |
5 |
Laccase |
77-184/64-96/0.52-0.83 |
1250-1490 /3.57-3.83 |
0.46-0.54 |
Wang et al., 2019 [14] |
|
Wheat bran AX |
14.2 |
8-O-4'/5-5', 8-5' |
2 |
Laccase |
100-200//0.01-0.02 |
39 |
0.52 |
Li et al., 2020 [2] |
|
Wheat bran AX |
0.006-0.009 |
—— |
5-6 |
Laccase |
—— |
66-77 |
0.76-0.83 |
Berlanga-Reyes et al., 2011 [32] |
|
Wheat bran AX |
0.008 |
—— |
5 |
Laccase |
177/20/0.11 |
74 |
0.8 |
Berlanga-Reyes et al., 2014 [33] |
|
Wheat endosperm AX |
0.526 |
—— |
2 |
Laccase |
31//0.160 |
504 |
0.66 |
Morales-Ortega et al., 2014 [34] |
Application of Wheat Arabinoxylans Gels
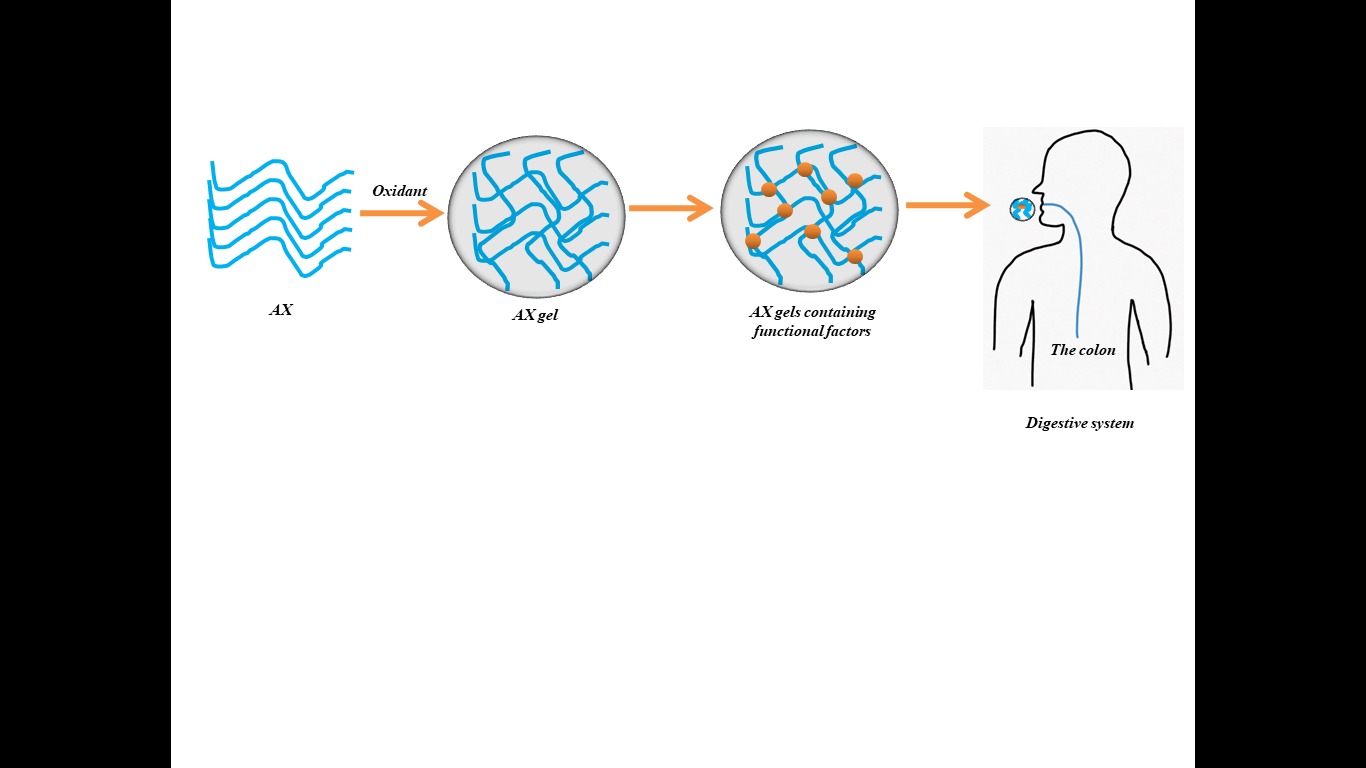
AX, as a dietary fiber, are resistant to digestive enzyme and can be degraded and fermented by microorganism in the colon [36]. Therefore, covalent AX hydrogels could be developed for novel colon-specific delivery system for hydrophilic drugs and bioactive compounds (Figure 3). It has been reported that the structure feature of AX gels could significantly affect their degradation property in the colon. AX gel with a higher cross-linking density and more compact structure could slow down the degradation of three-dimensional gel structure when they are fermented in the intestine by B. longum and B. adolescentis [37]. In comparison to AX-free culture substrate, the AX gel samples produce significantly higher short chain fatty acids (SCFAs) during fermentation. It has been stated that during the degradation of AX gels, partial AX chains could be converted to SCFAs such as acetic acids, propionic and butyric acids by the Bifidobacterium mix. Therefore, AX gels can be well developed for designing controlled delivery system activated by the colon microbiota. Carvajal-Millan et al. have fabricated AX gels as oral delivery system for insulin, to develop a painless therapy for diabetics [38]. It has been reported that the proteolysis of insulin encapsulated in AX gels is reduced to 17% in comparison to pure insulin solution, and the AX gels allow the release of insulin in the colon. The water-extractable arabinoxylan (WEAX) hydrogels are prepared via laccase treatment to encapsulate the protein drug model bovine serum albumin (BSA) [30]. The diffusion coefficient of BSA from AX gels tends to decrease with the increase in cross-linking degree and AX concentrations, and therefore the release property of protein drug could be controlled by regulating the structure of AX hydrogels. In a more recent study, arabinoxylan was used as biomaterial to prepare pH-responsive microgel for BSA delivery [39]. The obtained results showed that only 14.53% BSA was released in simulated gastric fluids, while 66.64% BSA was released in intestinal solutions at pH7.4, indicating that AX microgels had superior controlled release properties of BSA. In addition, small molecules caffeine could be encapsulated in AX gels, and the gels were stable in the stomach acid condition [40]. Overall, these studies have showed the great potential for development of AX gel as delivery system for hydrophilic drugs, which would broaden its practical application in biomedical field.
Figure 3: AX gel as a nutrient delivery vehicle.
In recent years, arabinoxylan based gels have been widely investigated and developed to encapsulate the functional nutrients which are unstable and easily degraded during storage, processing and human digestion process. Morales-Ortega et al. have successfully developed AX gels as delivery system for the probiotics B. longum, and it has been reported that the presence of probiotics could affect the crosslinking of AX and results in the a slight reduction of AX gel elasticity [34]. Carvajal-Millan’s research team has fabricated a series of AX based gel particles for probiotic entrapment using electrospray technique [41-42]. The simulated gastrointestinal digestion results show that the prepared AX gel particles are not degraded in the stomach and small intestinal, which is beneficial for colon targeted delivery of probiotics. Previous study by Hernández-Espinoza et al. has reported that arabinoxylan gel could encapsulate lycopene, and the AX concentrations are the dominant factor modulating lycopene release. The increase in AX concentration could decrease the diffusion coefficient of lycopene [43].
Except for development of delivery system, the utilization of AX-based gel system as biomaterial have drawn researchers’ much attention more recently. Proteins and AX are used as substrates to fabricate interpenetrating polymer network (IPN) gel, due to more compact gel structure and strengthened mechanical properties [44,45]. Khan et al. have employed the hydrothermal method to fabricate AX-graphene oxide based functional composite hydrogels for bone tissue engineering [46]. Graphene oxide could increase AX gels’ mechanical property. In addition, the fabricated composite gel has antibacterial activity and favored for cell viability and proliferation, which could be used for wound healing.
Conclusion and Future Trend
Wheat bran arabinoxylan and its gel formulations are of high nutritive values, which is beneficial for development of both functional foods and biomedical products. The extraction method and the subsequent structural composition of obtained AX significantly affect their gel forming capacity, and therefore the structure-function relationship should be investigated more clearly to illustrate the potential gelling mechanism. More importantly, more eco-friendly extraction technique should be explored and established to improve the extraction efficiency, and to avoid the loss of functional components (e.g. ferulic acids, proteins, etc) from the commonly-used alkaline extraction method. Due to the fact that AX could be degraded by the colonic bacterial enzymes, AX based gels have huge potential to be designed as an ideal carrier for colon-specific delivery of protein/peptide drugs and bioactive compounds. However, pure AX gels have poor mechanical property and uncontrolled porous gel structure, which may significantly affect their delivery performance. In addition, the poor mechanical property of AX gel also could limit its application in tissue engineering. In future, fabrication of AX based interpenetrating polymer network gel system or addition of micro/nano-polymer as reinforcing materials can provide a simple and valid strategy to strengthen the composite gel structure.
Author Contribution Statement
QZ and MZ conceived, proofread and wrote the manuscript. PW reviewed and revised the manuscript.
Acknowledgments
This work was financially supported by Natural Science Foundation of Tianjin (20JCQNJC00080), Tianjin “131” Innovative Talent Team Project (201926).
References
2. Li C, Wang L, Chen Z, Li Y, Li J. Facile and green preparation of diverse arabinoxylan hydrogels from wheat bran by combining subcritical water and enzymatic crosslinking. Carbohydrate Polymers. 2020 Aug 1;241:116317.
3. Benamrouche S, Crônier D, Debeire P, Chabbert B. A chemical and histological study on the effect of (1→ 4)-β-endo-xylanase treatment on wheat bran. Journal of Cereal Science. 2002 Sep 1;36(2):253-60.
4. Nystrom L, Lampi AM, Andersson AA, Kamal-Eldin A, Gebruers K, Courtin CM, et al. Phytochemicals and dietary fiber components in rye varieties in the HEALTHGRAIN diversity screen. Journal of Agricultural and Food Chemistry. 2008 Nov 12;56(21):9758-66.
5. Vardakou M, Palop CN, Christakopoulos P, Faulds CB, Gasson MA, Narbad A. Evaluation of the prebiotic properties of wheat arabinoxylan fractions and induction of hydrolase activity in gut microflora. International Journal of Food Microbiology. 2008 Mar 31;123(1-2):166-70.
6. Kouzounis D, Jonathan MC, Soares N, Kabel MA, Schols HA. In vivo formation of arabinoxylo-oligosaccharides by dietary endo-xylanase alters arabinoxylan utilization in broilers. Carbohydrate Polymers. 2022 Sep 1;291:119527.
7. Mendis M, Simsek S. Arabinoxylans and human health. Food Hydrocolloids. 2014 Dec 15;42:239-43.
8. Cao L, Liu X, Qian T, Sun G, Guo Y, Chang F, et al. Antitumor and immunomodulatory activity of arabinoxylans: A major constituent of wheat bran. International Journal of Biological Macromolecules. 2011 Jan 1;48(1):160-4.
9. Hartvigsen ML, Gregersen S, Laerke HN, Holst JJ, Bach Knudsen KE, Hermansen K. Effects of concentrated arabinoxylan and β-glucan compared with refined wheat and whole grain rye on glucose and appetite in subjects with the metabolic syndrome: a randomized study. European Journal of Clinical Nutrition. 2014 Jan;68(1):84-90.
10. Dhital S, Dolan G, Stokes JR, Gidley MJ. Enzymatic hydrolysis of starch in the presence of cereal soluble fibre polysaccharides. Food & Function. 2014;5(3):579-86.
11. Xiao X, Qiao J, Wang J, Kang J, He L, Li J, et al. Grafted ferulic acid dose-dependently enhanced the apparent viscosity and antioxidant activities of arabinoxylan. Food Hydrocolloids. 2022 Jul 1;128:107557.
12. Nishitsuji Y, Whitney K, Nakamura K, Hayakawa K, Simsek S. Changes in structure and solubility of wheat arabinoxylan during the breadmaking process. Food Hydrocolloids. 2020 Dec 1;109:106129.
13. Escarnot E, Aguedo M, Agneessens R, Wathelet B, Paquot M. Extraction and characterization of water-extractable and water-unextractable arabinoxylans from spelt bran: Study of the hydrolysis conditions for monosaccharides analysis. Journal of Cereal Science. 2011 Jan 1;53(1):45-52.
14. Wang L, Zhang L, Qiu S, Liu C, Zhang P, Yin L, et al. Rheological properties and structural characteristics of arabinoxylan hydrogels prepared from three wheat bran sources. Journal of Cereal Science. 2019 Jul 1;88:79-86.
15. Aguedo M, Fougnies C, Dermience M, Richel A. Extraction by three processes of arabinoxylans from wheat bran and characterization of the fractions obtained. Carbohydrate Polymers. 2014 May 25;105:317-24.
16. Sánchez-Bastardo N, Romero A, Alonso E. Extraction of arabinoxylans from wheat bran using hydrothermal processes assisted by heterogeneous catalysts. Carbohydrate Polymers. 2017 Mar 15;160:143-52.
17. Wang J, Sun B, Liu Y, Zhang H. Optimisation of ultrasound-assisted enzymatic extraction of arabinoxylan from wheat bran. Food Chemistry. 2014 May 1;150:482-8.
18. Wang J, Smits E, Boom RM, Schutyser MA. Arabinoxylans concentrates from wheat bran by electrostatic separation. Journal of Food Engineering. 2015 Jun 1;155:29-36.
19. Zhang Z, Smith C, Li W. Extraction and modification technology of arabinoxylans from cereal by-products: A critical review. Food Research International. 2014 Nov 1;65:423-36.
20. Maes C, Delcour JA. Structural characterisation of water-extractable and water-unextractable arabinoxylans in wheat bran. Journal of Cereal Science. 2002 Mar 1;35(3):315-26.
21. Zhou S, Liu X, Guo Y, Wang Q, Peng D, Cao L. Comparison of the immunological activities of arabinoxylans from wheat bran with alkali and xylanase-aided extraction. Carbohydrate Polymers. 2010 Jul 23;81(4):784-9.
22. García JC, Díaz MJ, Garcia MT, Feria MJ, Gómez DM, López F. Search for optimum conditions of wheat straw hemicelluloses cold alkaline extraction process. Biochemical Engineering Journal. 2013 Feb 15;71:127-33.
23. Hell J, Donaldson L, Michlmayr H, Kraler M, Kneifel W, Potthast A, et al. Effect of pretreatment on arabinoxylan distribution in wheat bran. Carbohydrate Polymers. 2015 May 5;121:18-26.
24. Yilmaz-Turan S, Jiménez-Quero A, Menzel C, de Carvalho DM, Lindström ME, Sevastyanova O, et al. Bio-based films from wheat bran feruloylated arabinoxylan: Effect of extraction technique, acetylation and feruloylation. Carbohydrate Polymers. 2020 Dec 15;250:116916.
25. Jiang Y, Bai X, Lang S, Zhao Y, Liu C, Yu L. Optimization of ultrasonic-microwave assisted alkali extraction of arabinoxylan from the corn bran using response surface methodology. International Journal of Biological Macromolecules. 2019 May 1;128:452-8.
26. Carvajal-Millan E, Guigliarelli B, Belle V, Rouau X, Micard V. Storage stability of laccase induced arabinoxylan gels. Carbohydrate Polymers. 2005 Jan 10;59(2):181-8.
27. Khalighi S, Berger RG, Ersoy F. Cross-linking of wheat bran arabinoxylan by fungal laccases yields firm gels. Processes. 2019 Dec 30;8(1):36.
28. Yilmaz-Turan S, Lopez-Sanchez P, Jiménez-Quero A, Plivelic TS, Vilaplana F. Revealing the mechanisms of hydrogel formation by laccase crosslinking and regeneration of feruloylated arabinoxylan from wheat bran. Food Hydrocolloids. 2022 Jul 1;128:107575.
29. Han K, Gao J, Wei W, Zhu Q, Fersht V, Zhang M. Laccase‐induced wheat bran arabinoxylan hydrogels from different wheat cultivars: Structural, physicochemical, and rheological characteristics. Journal of Food Processing and Preservation. 2022 Mar;46(3):e16394.
30. Carvajal-Millan E, Guilbert S, Morel MH, Micard V. Impact of the structure of arabinoxylan gels on their rheological and protein transport properties. Carbohydrate Polymers. 2005 Jun 20;60(4):431-8.
31. Martínez-López AL, Carvajal-Millan E, Marquez-Escalante J, Campa-Mada AC, Rascón-Chu A, López-Franco YL, et al. Enzymatic cross-linking of ferulated arabinoxylan: Effect of laccase or peroxidase catalysis on the gel characteristics. Food Science and Biotechnology. 2019 Apr;28(2):311-8.
32. Zhang X, Chen T, Lim J, Gu F, Fang F, Cheng L, et al. Acid gelation of soluble laccase-crosslinked corn bran arabinoxylan and possible gel formation mechanism. Food Hydrocolloids. 2019 Jul 1;92:1-9.
33. Berlanga-Reyes CM, Carvajal-Millan E, Lizardi-Mendoza J, Islas-Rubio AR, Rascón-Chu A. Enzymatic cross-linking of alkali extracted arabinoxylans: Gel rheological and structural characteristics. International Journal of Molecular Sciences. 2011 Sep 9;12(9):5853-61.
34. Berlanga-Reyes CM, Carvajal-Millan E, Hicks KB, Yadav MP, Rascón-Chu A, Lizardi-Mendoza J, et al. Protein/arabinoxylans gels: effect of mass ratio on the rheological, microstructural and diffusional characteristics. International Journal of Molecular Sciences. 2014 Oct 21;15(10):19106-18.
35. Morales-Ortega A, Carvajal-Millan E, Brown-Bojorquez F, Rascón-Chu A, Torres-Chavez P, López-Franco YL, et al. Entrapment of probiotics in water extractable arabinoxylan gels: Rheological and Microstructural Characterization. Molecules. 2014 Mar 24;19(3):3628-37.
36. Rascón-Chu A, Martínez-López AL, Berlanga-Reyes C, Carvajal-Millan E, Campa-Mada AC, Gardea AA, et al. Arabinoxylans gels as lycopene carriers: In vitro degradation by colonic bacteria. MRS Online Proceedings Library (OPL). 2012;1487.
37. Martínez-López AL, Carvajal-Millan E, Micard V, Rascón-Chu A, Brown-Bojorquez F, Sotelo-Cruz N, et al. In vitro degradation of covalently cross-linked arabinoxylan hydrogels by bifidobacteria. Carbohydrate Polymers. 2016 Jun 25;144:76-82.
38. Carvajal-Millan E, Berlanga-Reyes C, Rascón-Chu A, Martínez-López AL, Márquez-Escalante JA, Campa-Mada AC, et al. In vitro evaluation of arabinoxylan gels as an oral delivery system for insulin. MRS Online Proceedings Library (OPL). 2012;1487.
39. Li J, Lu Z, Chen Z, Li C, Du Y, Chen C, et al. Preparation and characterization of pH-responsive microgel using arabinoxylan from wheat bran for BSA delivery. Food Chemistry. 2021 Apr 16;342:128220.
40. Iravani S, Fitchett CS, Georget DM. Physical characterization of arabinoxylan powder and its hydrogel containing a methyl xanthine. Carbohydrate Polymers. 2011 Apr 22;85(1):201-7.
41. Paz‐Samaniego R, Rascón‐Chu A, Brown‐Bojorquez F, Carvajal‐Millan E, Pedroza‐Montero M, Silva‐Campa E, et al. Electrospray‐assisted fabrication of core‐shell arabinoxylan gel particles for insulin and probiotics entrapment. Journal of Applied Polymer Science. 2018 Jul 10;135(26):46411.
42. Ohlmaier-Delgadillo F, Carvajal-Millan E, López-Franco YL, Islas-Osuna MA, Micard V, Antoine-Assor C, et al. Ferulated Pectins and Ferulated Arabinoxylans Mixed Gel for Saccharomyces boulardii Entrapment in Electrosprayed Microbeads. Molecules. 2021 Jan;26(9):2478.
43. Hernández-Espinoza AB, Piñón-Muñiz MI, Rascón-Chu A, Santana-Rodríguez VM, Carvajal-Millan E. Lycopene/arabinoxylan gels: Rheological and controlled release characteristics. Molecules. 2012 Feb 28;17(3):2428-36.
44. Zhu Q, Han K, Wang S, Muhindo EM, Wei W, Li J, et al. Design and structural characterization of edible double network gels based on wheat bran arabinoxylan and pea protein isolate. International Journal of Biological Macromolecules. 2022 Jun 9.
45. Yan J, Yin L, Qu Y, Yan W, Zhang M, Su J, et al. Effect of calcium ions concentration on the properties and microstructures of doubly induced sorghum arabinoxylan/soy protein isolate mixed gels. Food Hydrocolloids. 2022 Jul 22:107997.
46. Aslam Khan MU, Haider A, Abd Razak SI, Abdul Kadir MR, Haider S, Shah SA, et al. Arabinoxylan/graphene‐oxide/nHAp‐NPs/PVA bionano composite scaffolds for fractured bone healing. Journal of Tissue Engineering and Regenerative Medicine. 2021 Apr;15(4):322-35.