Abstract
Despite major advances in molecular oncology and cancer therapeutics, early detection remains a critical limitation for many malignancies. Current screening approaches rely largely on anatomical or morphological changes, highlighting a persistent gap between modern biological understanding of cancer and its clinical application. This article examines the evolution of cancer biology and explores why these advances have not yet translated into effective early detection strategies.
I propose a wearable device–based framework that integrates peripheral blood DNA methylation profiling of tumor suppressor genes and oncogenes with physical detection of early malignant cells. Technologies such as superconducting quantum interference devices (SQUID), metamaterial sensors, and microwave imaging are discussed as platforms for three-dimensional localization of nascent cancer cells. In parallel, “elimination” wearable devices capable of delivering localized energy—via radiofrequency or microwave ablation, nanoparticles, or programmable nanomachines—are proposed to eradicate malignant cells at inception.
This integrative model bridges molecular oncology, applied physics, and digital health, introducing a new paradigm for early cancer detection and prevention.
Keywords
Cancer screening, SQUID, Nano-machines, Meta material sensor, Electromagnetic field, Microwave imaging, Radio frequency ablation, Digital oncology
Introduction
In the last 83 years, since the birth of nitrogen mustard [1] which opened the new chapter of cancer therapeutics, we have also been dealing with the dilemma of early detection of cancer [2]. As of this writing, some 83 years later, we still lack a reliable and valid cancer screening test for a good number of neoplastic disorders.
Some notable examples include ovarian cancer [3], pancreatico-biliary carcinoma [4], hepatocellular carcinoma [5], brain tumors [6], lymphoma [7], and leukemias [8].
Mammogram [9], colonoscopy [10], and the controversial PSA screening for prostate cancer [11] are among the few and perhaps only success stories.
As we have advanced enormously in the development of new generation of cancer therapeutics, we have also developed a significantly deeper understanding of molecular and genetic mechanisms that underlie the neoplastic transformations [12].
This development and evolution in our understanding has not as yet been optimally employed in the development of new cancer screening and treatment in real world, while we have moved from nitrogen mustard and multi-agent chemotherapy protocols to targeted therapy [13] and new generation of immunotherapy [14], as well as tumor vaccines [15].
We still rely on calcification pattern in mammogram and visual appreciation of tumor mass or polyp, for breast and colon cancer screening, respectively.
This represents a major breakdown between our understanding of the underlying mechanisms of neoplastic transformation, and their application towards development of new cancer screening methodology.
A similar breakdown is witnessed between such understanding and development of new generations of cancer therapeutics. Even though we have jumped from nitrogen mustard to targeted therapy and new generation of immunotherapy in the last 83 years, there is still a big gap between our understanding of neoplastic transformation [16] and its application in development of new generation of cancer therapeutics. Wearable devices are a revolution in cancer diagnosis and treatment. It takes advantage of DNA methylation profiling of tumor suppressor genes and elimination of cancer at its inception.
This article proposes a novel conceptual framework for cancer detection and prevention using wearable devices that integrate blood-based DNA methylation profiling with physical detection of early malignant cells. By exploiting increased signaling network entropy and associated nano-scale electromagnetic perturbations in newly transformed cells, such devices could enable real-time localization and targeted elimination of cancer at its inception. This approach introduces a potential shift toward preemptive oncology, with implications for earlier diagnosis, reduced morbidity, and improved cancer outcomes.
Evolution in definition of cancer
For the most part, in 20th century, cancer used to be defined as proliferation of cells in a disorderly fashion [17]. As knowledge about differentiation pathways including the steroid-retinoid family of receptors evolved [18], cancer definition also evolved into uncontrolled proliferation of de-differentiated cells.
As we became more knowledgeable and aware of the associated molecular and genetic aberrancies of different cancers, we started to incorporate them in our pathology reports. We also started to teach cancer as a disorder of genome in medical schools and medical texts. Next generation sequencing [19] and comprehensive immuno-staining [20] have become an essential part of modern pathology reports.
Meanwhile, we have gone through whole genome profiling [21], exomics [22], epigenomics [23], microRNA network development [24], proteomics [25], polyomics [26], and spatial polyomics [27].
This rapidly expanding body of molecular insight into tumor biology has reshaped how cancer is classified, studied, and conceptualized.
Discussion
The big question is, why these massive advancements in our understanding of neoplastic transformation have not yet been translated into, not only screening tests, but also identification of cancer cells at much earlier stages. Recently, methylation profiling of tumor suppressor genes and oncogenes in peripheral blood [28] has opened the way for achievement of this goal. Through this methodology we become able to recognize preneoplastic changes and potential future neoplastic transformation of different organs. One major barrier so far has been that such advances in understanding of neoplastic transformation have not lent themselves to physical measurement of the earliest events [29].
To overcome this barrier, I have proposed measurement of perturbations in master regulator complex network entropy of normal cells [30] in different stages down the path of neoplastic transformation. It has been well shown, described and published that master regulator complex network entropy of cancer cells are significantly higher as compared to their normal counterparts [31]. Introduction of methylation profiling of tumor suppressor genes and oncogenes in peripheral blood, discussed in AACR 2025, would enable us to identify earlier neoplastic transformation events of different organs. This, in and of itself, would not enable us to pinpoint and identify a small fraction of cells undergoing neoplastic transformation in each organ. We could, however, take advantage of the increase in master regulator complex network entropy of that fraction of cells in organ of interest towards their identification in 3D space [32].
Squid [33] and microwave [34] imaging are two of practical applications of physical parameter, such as magnetic field of cancer cells at a nano-scale [35].
Nano-scale variation of magnetic field of cancer cells with elevated master regulator complex network entropy [36], might be a better and more distinctive measurement in this regard.
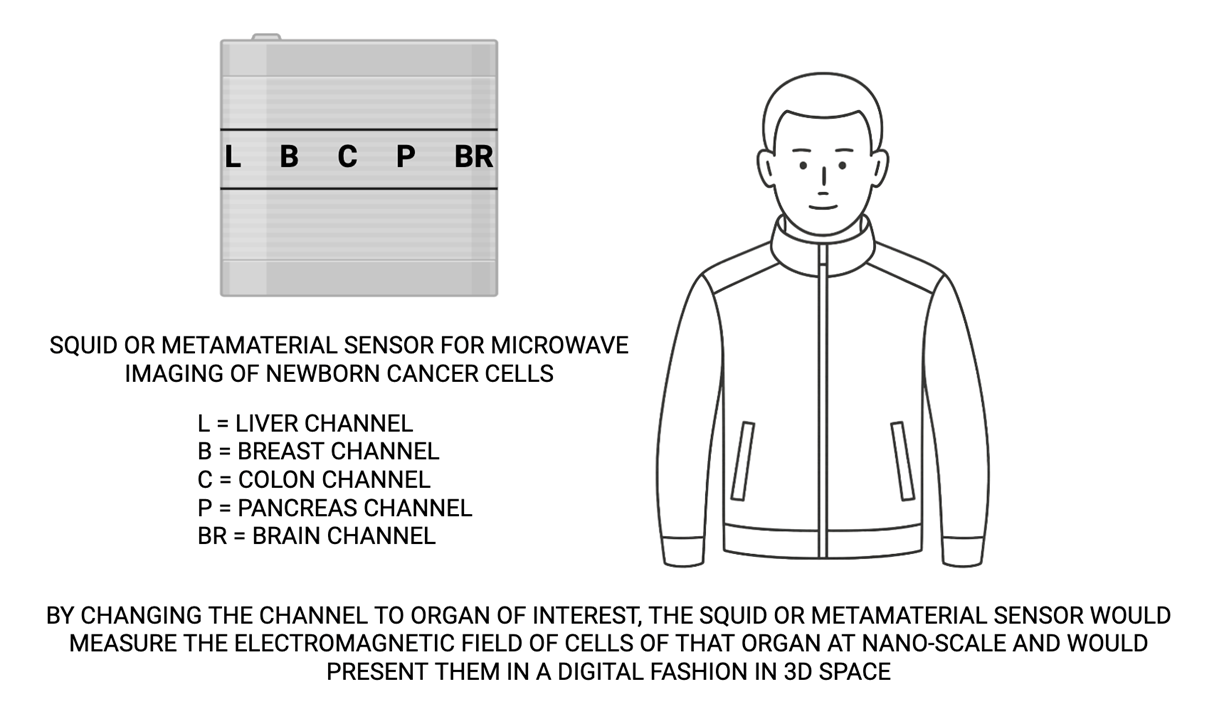
As such, my proposed wearable device should be loaded either with squid [37] or metamaterial sensor for microwave imaging of recently born cancer cells.
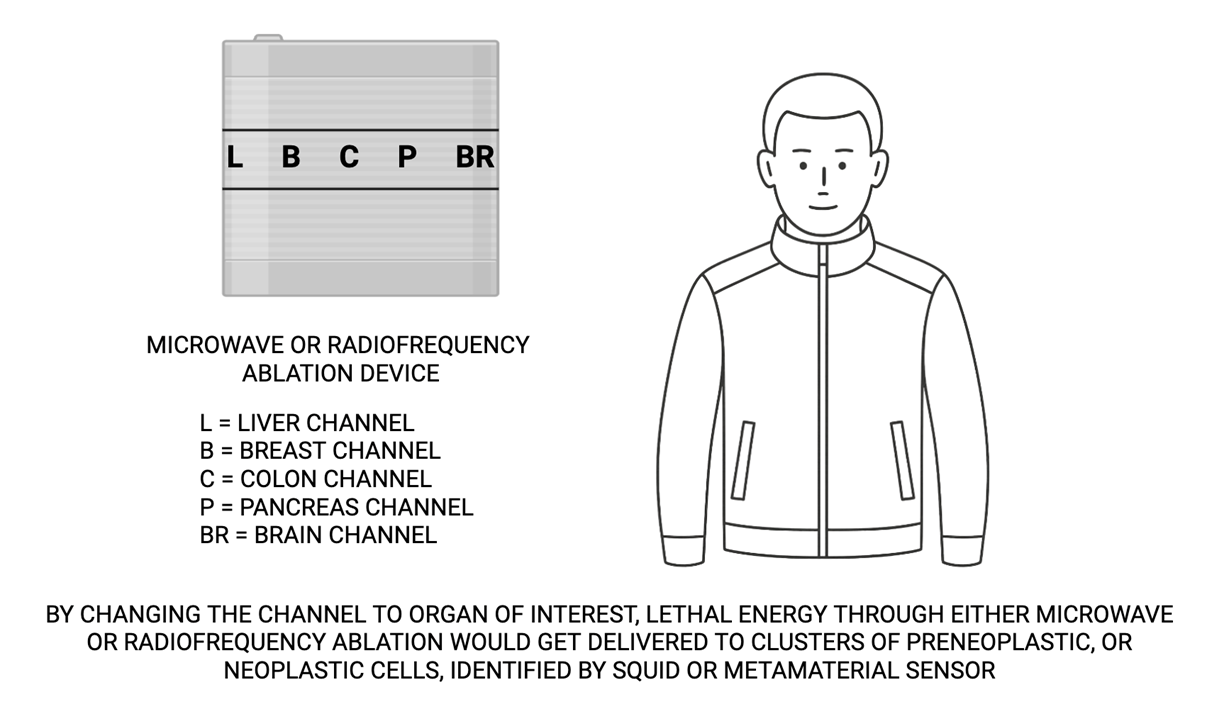
Such wearable device should also be designed with multiple channels: each channel focusing on one organ of interest, based on information given to us by methylation profiling of specific tumor suppressor genes and oncogenes related to the organ of interest. Hereby, I would coin “D” for detection wearable devices and “E” for cancer cell elimination wearable device. The “E” wearable device should be able to deliver lethal high energy particles at nano-scale level to the newborn malignant cells [38].
Radiofrequency [39] and microwave ablation [40] are among the two major modalities that could be employed for annihilation of new population of cancer cells. Of course, we could employ other methodologies for annihilation of the newborn cancer cells, such as nano-particles [41] and nano-machines [42] that could get programmed to eliminate cancer cells of interest by delivering lethal energy of different types, including vibration [43], heat or cold [44].
Conclusion
The design and manufacture of above-mentioned wearable devices and the application of nano-particles, and nano-machines would take us into the new era of cancer cell detection at the time of inception and its elimination, or abortion, before it could acquire the capability of causing the demise of the host. As such, we would take a quantum leap into the new field of cancer detection and cure.
References
2. Schwartzberg L, Broder MS, Ailawadhi S, Beltran H, Blakely LJ, Budd GT, et al. Impact of early detection on cancer curability: A modified Delphi panel study. PLoS One. 2022 Dec 21;17(12):e0279227.
3. Bast RC Jr, Lu Z, Han CY, Lu KH, Anderson KS, Drescher CW, et al. Biomarkers and Strategies for Early Detection of Ovarian Cancer. Cancer Epidemiol Biomarkers Prev. 2020 Dec;29(12):2504–12.
4. Luo W, Wang J, Chen H, Cao Z, Qiu J, Liu Y, et al. Advances in Screening and Early Diagnosis of Pancreatic Cancer. Cancer Screen Prev 2024;3(3):154–62.
5. Singal AG, Mittal S, Yerokun OA, Ahn C, Marrero JA, Yopp AC, et al. Hepatocellular Carcinoma Screening Associated with Early Tumor Detection and Improved Survival Among Patients with Cirrhosis in the US. Am J Med. 2017 Sep;130(9):1099–106.e1.
6. Elias MG, Hadjiyiannis H, Vafaee F, Scott KF, de Souza P, Becker TM, et al. The Quest for Non-Invasive Diagnosis: A Review of Liquid Biopsy in Glioblastoma. Cancers (Basel). 2025 Aug 19;17(16):2700.
7. Witzig TE, Taylor WR, Mahoney DW, Bamlet WR, Foote PH, Burger KN, et al. Blood Plasma Methylated DNA Markers in the Detection of Lymphoma: Discovery, Validation, and Clinical Pilot. Am J Hematol. 2025 Feb;100(2):218–28.
8. Qin Y, Pu X, Hu D, Yang M. Machine learning-based biomarker screening for acute myeloid leukemia prognosis and therapy from diverse cell-death patterns. Sci Rep. 2024 Aug 2;14(1):17874.
9. Moss SM, Cuckle H, Evans A, Johns L, Waller M, Bobrow L. Effect of mammographic screening from age 40 years on breast cancer mortality at 10 years' follow-up: a randomised controlled trial. Lancet. 2006 Dec 9;368(9552):2053–60.
10. Segnan N, Armaroli P, Bonelli L, Risio M, Sciallero S, Zappa M, et al. Onceonly sigmoidoscopy in colorectal cancer screening: follow-up findings of the Italian Randomized Controlled Trial--SCORE. J Natl Cancer Inst. 2011;103(17):1310–22.
11. Martin RM, Turner EL, Young GJ, Metcalfe C, Walsh EI, Lane JA, et al. Prostate-Specific Antigen Screening and 15-Year Prostate Cancer Mortality: A Secondary Analysis of the CAP Randomized Clinical Trial. JAMA. 2024 May 7;331(17):1460–70.
12. Kamp EJ, Dinjens WN, Doukas M, van Marion R, Verheij J, Ponsioen CY, et al. Genetic alterations during the neoplastic cascade towards cholangiocarcinoma in primary sclerosing cholangitis. J Pathol. 2022 Nov;258(3):227–35.
13. Zhu K, Yang X, Tai H, Zhong X, Luo T, Zheng H. HER2-targeted therapies in cancer: a systematic review. Biomark Res. 2024 Feb 2;12(1):16.
14. Ling SP, Ming LC, Dhaliwal JS, Gupta M, Ardianto C, Goh KW, et al. Role of Immunotherapy in the Treatment of Cancer: A Systematic Review. Cancers (Basel). 2022 Oct 24;14(21):5205.
15. Zhou Y, Wei Y, Tian X, Wei X. Cancer vaccines: current status and future directions. J Hematol Oncol. 2025 Feb 17;18(1):18.
16. Neal JT, Kuo CJ. Organoids as Models for Neoplastic Transformation. Annu Rev Pathol. 2016 May 23; 11:199–220.
17. Brown JS, Amend SR, Austin RH, Gatenby RA, Hammarlund EU, Pienta KJ. Updating the Definition of Cancer. Mol Cancer Res. 2023 Nov 1;21(11):1142-7.
18. Esposito M, Amory JK, Kang Y. The pathogenic role of retinoid nuclear receptor signaling in cancer and metabolic syndromes. J Exp Med. 2024 Sep 2;221(9):e20240519.
19. Cummings CA, Peters E, Lacroix L, Andre F, Lackner MR. The Role of Next-Generation Sequencing in Enabling Personalized Oncology Therapy. Clin Transl Sci. 2016 Dec;9(6):283–92.
20. Kim SW, Roh J, Park CS. Immunohistochemistry for Pathologists: Protocols, Pitfalls, and Tips. J Pathol Transl Med. 2016 Nov;50(6):411–8.
21. Fujimoto A, Wong JH, Yoshii Y, Akiyama S, Tanaka A, Yagi H, et al. Whole-genome sequencing with long reads reveals complex structure and origin of structural variation in human genetic variations and somatic mutations in cancer. Genome Med. 2021 Apr 29;13(1):65.
22. Champiat S, Ferté C, Lebel-Binay S, Eggermont A, Soria JC. Exomics and immunogenics: Bridging mutational load and immune checkpoints efficacy. Oncoimmunology. 2014 Jan 1;3(1):e27817.
23. Sandoval J, Esteller M. Cancer epigenomics: beyond genomics. Curr Opin Genet Dev. 2012 Feb;22(1):50-5.
24. Bandyopadhyay S, Mitra R, Maulik U, Zhang MQ. Development of the human cancer microRNA network. Silence. 2010 Feb 2;1(1):6.
25. Haga Y, Minegishi Y, Ueda K. Frontiers in mass spectrometry-based clinical proteomics for cancer diagnosis and treatment. Cancer Sci. 2023 May;114(5):1783–91.
26. Menyhárt O, Győrffy B. Multi-omics approaches in cancer research with applications in tumor subtyping, prognosis, and diagnosis. Comput Struct Biotechnol J. 2021 Jan 22;19:949–60.
27. Ravi VM, Will P, Kueckelhaus J, Sun N, Joseph K, Salié H, et al. Spatially resolved multi-omics deciphers bidirectional tumor-host interdependence in glioblastoma. Cancer Cell. 2022 Jun 13;40(6):639–55.e13.
28. Radpour R, Barekati Z, Kohler C, Lv Q, Bürki N, Diesch C, et al. (2011) Hypermethylation of Tumor Suppressor Genes Involved in Critical Regulatory Pathways for Developing a Blood-Based Test in Breast Cancer. PLoS ONE 6(1): e16080.
29. Schiffman JD, Fisher PG, Gibbs P. Early detection of cancer: past, present, and future. Am Soc Clin Oncol Educ Book. 2015:57–65.
30. Teschendorff AE, Banerji CR, Severini S, Kuehn R, Sollich P. Increased signaling entropy in cancer requires the scale-free property of protein interaction networks. Sci Rep. 2015 Apr 28;5:9646.
31. Feng L, Sun YD, Li C, Li YX, Chen LN, Zeng R. Pan-cancer network disorders revealed by overall and local signaling entropy. J Mol Cell Biol. 2021 Dec 6;13(9):622–35.
32. Li A, Yin X, Pan Y. Three-Dimensional Gene Map of Cancer Cell Types: Structural Entropy Minimisation Principle for Defining Tumour Subtypes. Sci Rep. 2016 Feb 4;6:20412.
33. Johnson C, Adolphi NL, Butler KL, Debbie M L, Larson R, Schwindt PD, et al. Magnetic Relaxometry with an Atomic Magnetometer and SQUID Sensors on Targeted Cancer Cells. J Magn Magn Mater. 2012 Aug 1;324(17):2613–9.
34. Khalid N, Zubair M, Mehmood MQ, Massoud Y. Emerging paradigms in microwave imaging technology for biomedical applications: unleashing the power of artificial intelligence. NPJ Imaging. 2024 Jun 3;2(1):13.
35. Lopez S, Hallali N, Lalatonne Y, Hillion A, Antunes JC, Serhan N, et al. Magneto-mechanical destruction of cancer-associated fibroblasts using ultra-small iron oxide nanoparticles and low frequency rotating magnetic fields. Nanoscale Adv. 2021 Nov 18;4(2):421–36.
36. Bergandi L, Lucia U, Grisolia G, Salaroglio IC, Gesmundo I, Granata R, et al. Thermomagnetic Resonance Effect of the Extremely Low Frequency Electromagnetic Field on Three-Dimensional Cancer Models. Int J Mol Sci. 2022 Jul 19;23(14):7955.
37. G. G. Kenning, R. Rodriguez, V. S. Zotev, A. Moslemi, S. Wilson, L. Hawel, et al. Detection of magnetically enhanced cancer tumors using SQUID magnetometry: A feasibility study. Rev Sci Instrum. 2005 Jan 1;76(1):014303.
38. Ghosh S, Lee SJ, Hsu JC, Chakraborty S, Chakravarty R, Cai W. Cancer Brachytherapy at the Nanoscale: An Emerging Paradigm. Chem Biomed Imaging. 2023 Nov 21;2(1):4–26.
39. Friedman M, Mikityansky I, Kam A, Libutti SK, Walther MM, Neeman Z, et al. Radiofrequency ablation of cancer. Cardiovasc Intervent Radiol. 2004 Sep-Oct;27(5):427–34.
40. Lubner MG, Brace CL, Hinshaw JL, Lee FT Jr. Microwave tumor ablation: mechanism of action, clinical results, and devices. J Vasc Interv Radiol. 2010 Aug;21(8 Suppl):S192–203.
41. Sun L, Liu H, Ye Y, Lei Y, Islam R, Tan S, et al. Smart nanoparticles for cancer therapy. Signal Transduct Target Ther. 2023 Nov 3;8(1):418.
42. Wang Y, Baars I, Berzina I, Rocamonde-Lago I, Shen B, Yang Y, et al. A DNA robotic switch with regulated autonomous display of cytotoxic ligand nanopatterns Nature Nanotechnology. 2024;19:1366–74.
43. Ayala-Orozco C, Galvez-Aranda D, Corona A, Seminario JM, Rangel R, Myers JN, et al. Molecular jackhammers eradicate cancer cells by vibronic-driven action. Nat Chem. 2024 Mar;16(3):456–65.
44. Liebl CM, Kutschan S, Dörfler J, Käsmann L, Hübner J. Systematic review about complementary medical hyperthermia in oncology. Clin Exp Med. 2022 Nov;22(4):519–65.