Abstract
Mastitis is an inflammatory disease of the bovine mammary gland caused by bacterial, viral, fungal, or parasitic infections, with pathogens primarily entering through the teat canal. Once established, they proliferate and evade host immunity, triggering complex immunological cascades mediated by bacterial toxins, proteins, and immunomodulators, which can result in increased somatic cell counts, vascular permeability, fibrosis, and systemic toxemia in severe cases. Mycotic mastitis is mainly caused by yeasts, although filamentous fungi, particularly Aspergillus fumigatus, have been reported, while viral infections may contribute indirectly through immunosuppression. Clinically, mastitis presents as swelling and tenderness of the udder, fever, and depression, and may occur as clinical or subclinical disease, classified by duration as acute or chronic. Early diagnosis relies on tests such as the California Mastitis Test and somatic cell count, while disease severity is influenced by host, pathogen, and environmental interactions. Histopathologically, mastitis can manifest in multiple forms: lymphoplasmacytic mastitis with chronic interstitial lymphocyte, plasma cell, and macrophage infiltration; pyogranulomatous lesions with necrosis, neutrophils, epithelioid macrophages, and peripheral fibrosis; suppurative and abscedative lesions showing squamous metaplasia and hemorrhages; necrosuppurative mastitis with multifocal coagulation necrosis; granulomatous lesions with necrotic, sometimes mineralized foci; and mixed-type lesions displaying overlapping features. Early detection, appropriate therapeutic intervention, and good management practices are critical to reduce the prevalence and severity of bovine mastitis.
Keywords
California mastitis test, Bovine, Mastitis, Mycotic and viral infections, Pathological lesions
Introduction
Mastitis has been reported in almost all domestic mammals and has a worldwide geographic distribution [1]. It is known not only for affecting a wide range of animal species but also for its multifactorial nature. Mastitis is defined as inflammation of the mammary gland caused by the invasion of microorganisms that enter the gland mainly through the teat canal or following traumatic injury to the udder [2]. The disease is characterized by physical, chemical, and usually bacteriological changes in milk, along with pathological alterations in the glandular tissue of the udder. The major clinical signs include swelling of the udder, tenderness to touch, fever, and depression [3].
Based on cost analyses, mastitis has been ranked as the most economically important disease of dairy cattle [4]. Mammary tissue damage leading to reduced milk production accounts for nearly 70% of the total economic losses associated with mastitis, making it the leading cause of financial loss in the dairy industry. Additional losses arise from decreased milk yield, increased veterinary care costs, premature culling, and deterioration of milk quality. Globally, the annual economic loss due to mastitis has been estimated at approximately 35 billion US dollars, with losses in the United States alone ranging from 1.5 to 2.0 billion US dollars per year [5,6].
Subclinical mastitis is considered more economically significant than clinical mastitis because it remains undetected by gross examination while markedly compromising milk quality and productivity [7]. In Ethiopia, the overall prevalence of bovine mastitis has been reported to be 40.3%, of which 12% were clinical cases and 28.3% were subclinical cases [8]. Mastitis occurs when bacterial, viral, or fungal pathogens gain access to the mammary gland through the teat canal and multiply within the udder tissue. In addition to infectious agents, environmental conditions, milking practices such as udder washing and drying, post-milking teat dipping, and host-related factors play crucial roles in the occurrence and severity of the disease [9]. The most important milk changes associated with mastitis include discoloration, clot formation, and an increased number of leukocytes [10].
Effective mastitis control measures include routine screening using the California Mastitis Test (CMT), wearing gloves during milking, changing gloves between cows—particularly after milking infected animals—and testing newly introduced cows before herd integration [11]. The primary strategy for mastitis treatment relies on antibiotic therapy, including drugs such as penicillin, ampicillin, tetracycline, and gentamicin, administered via intramammary infusion or systemic routes such as intramuscular or intravenous injection [6]. However, prolonged or high-dose antibiotic use may reduce vitamin A levels, damage the udder epithelium, and disrupt the normal mammary microflora, which serves as a natural defense mechanism. This disruption may facilitate secondary infections, particularly fungal and yeast mastitis [12]. Although several studies in Ethiopia have documented the prevalence and incidence of bovine mastitis across different regions and production systems, comprehensive investigations integrating the causative factors, diagnostic approaches, treatment strategies, and pathological mechanisms remain limited. Therefore, the objective of this review is to provide an updated summary of bovine mastitis, emphasizing its causopathology, diagnostic methods, therapeutic approaches, and patho-pathogenesis, with particular focus on correlating pathological lesions with the etiological agents involved.
Causopathology
Etiology
Mastitis is characterized by an inflammatory response in the udder tissue of the mammary gland induced by microbial infections or physical damage. Mastitis-causing pathogens include bacteria, fungi, yeasts, and virus. Among them, bacterial intra-mammary infection is the most common one. Bovine mastitis can be caused by a minimum of 137 microorganisms from a broad phylogenetic spectrum without including viral causes [13].
Bacterial infection
Bacteria causing mastitis can be either contagious or environmental. Contagious bacteria spread from cow to cow at the time of milking through the hands, towels, and/or the milking machine. Environmental mastitis comes from the cow’s environment such as bedding material, soil, manure, feces, and stagnant water. Staphylococcus aureus, Streptococcus agalactiae, Mycoplasma spp. are among contagious bacteria. Escherichia coli, Enterococcus spp., coagulase-negative Staphylococcus, and Streptococcus uberis are among environmental mastitis-causing pathogens which are highly influenced by management practices [14].
The primary bacteria isolated from suppurative, mixed, and lymphoplasmacytic mastitis were Streptococcus spp., CNS, S. aureus, S. agalactiae, S. uberis, and C. bovis [15]. For Listeria monocytogenes, all internal organs showed no abnormalities, and the organism could only be isolated from the affected mammary parenchyma and mammary lymph node [16].
Mycoplasma species evolved from gram-positive bacteria in a degenerative evolution where these simple organisms lost the ability to produce a cell wall. Unresponsiveness to antibiotics and recurrent occurrence of mastitis is the best indication of mycoplasmal infection. Mycoplasma bovis is the most prevalent during outbreaks in cattle; however, several other species have been isolated from bovine mastitis including Mycoplasma arginini, M. bovirhinis, M. californicum, M. canadense, and M. dispar [17]. In outbreaks with mycoplasma mastitis, Mycoplasma-associated arthritis and bovine respiratory cases are found due to spread via lymphatic and peripheral blood systems. The organism was found colonizing the nares of the animal. Most cases of mycoplasmal mastitis are subclinical, and the greatest loss to a dairy is a result of the subclinical nature of the disease [18].
Mycotic mastitis
Most studies of mycotic mastitis in ruminants have focused on bovine infections, and only a few reports have dealt with infection of sheep and goats. Mastitis due to filamentous fungi is less common both in cows and small ruminants, although Aspergillus spp. have been incriminated. Mycotic mastitis in cows is predominantly caused by yeasts, especially those of the genera Candida, Cryptococcus, and Trichosporon [19]. Bovine mycotic mastitis is usually caused by yeasts, but mastitis due to filamentous fungi, mostly Aspergillus fumigatus, has also been reported. Mycotic mastitis is on the increase, and Candida krusei is frequently isolated from affected quarters [20].
The most frequently isolated organisms in mycotic mastitis are Candida species, which are unicellular opportunistic organisms ever present in the natural surroundings of dairy cattle (milkers’ hands, milking machines, treatment instruments, floors, straw, feed, dust, soil, drug mixtures, and sanitizing solutions) and are normal inhabitants of the skin of the udder and teats, where they exist in low numbers. Some intramammary fungal infections such as A. fumigatus and Candida spp. may result in death of affected animals. Outbreaks of mycotic mastitis are generally believed to result from ascending infection following incorrect administration of antibiotic preparations during the drying-off period [20].
Viral mastitis
Bovine herpesvirus 1, bovine herpesvirus 4, foot-and-mouth disease virus, and parainfluenza 3 virus have been isolated from milk from cows with clinical mastitis. Bovine herpesvirus 2, vaccinia, cowpox, pseudocowpox, vesicular stomatitis, foot-and-mouth disease viruses, and bovine papillomaviruses can play an indirect role in the etiology of bovine mastitis. Especially bovine viral diarrhea virus, bovine immunodeficiency virus, and bovine leukemia virus infections may play an indirect role in bovine mastitis due to their immunosuppressive properties [21].
Classification of mastitis
Mastitis can be presented as clinical or exist in the absence of visible signs of infection, in which case it is referred to as subclinical mastitis. In mild cases of clinical mastitis, flakes or clots are observed in the milk, whereas severe cases are associated with heat, swelling, and discoloration of the udder, as well as abnormal secretion. Subclinical mastitis produces no visible signs such as fever or loss of appetite, and changes in the udder and milk can only be detected using diagnostic tools. Subclinical mastitis occurs 15 to 40 times more frequently than the clinical form and has a longer duration. Consequently, it is more difficult to detect and serves as a reservoir of pathogens that spread udder infection among animals within the herd. Based on the duration of infection, mastitis is further classified into acute and chronic forms, where acute mastitis is characterized by sudden onset, while chronic mastitis involves prolonged inflammation lasting for months and resulting in progressive fibrous tissue development [13,14,22].
Transmission
Depending on the mode of transmission, mastitis etiological agents are classified as contagious or environmental. Contagious mastitis is caused by bacteria spreading from infected quarters to other quarters or from infected cows to healthy cows. The most common contagious mastitis pathogens are Staphylococcus aureus and Streptococcus agalactiae. Contagious mastitis is transmitted primarily during milking, with the udder serving as the main reservoir, and it tends to be subclinical in nature. Dry cow therapy, post-milking teat disinfection, and effective pre-milking hygiene are effective control measures for most contagious mastitis pathogens Mycoplasma bovis is a less common but important cause of contagious mastitis [23,24].
Environmental mastitis is caused by coliform bacteria such as E. coli, Klebsiella spp., and Enterobacter spp., which originate from contaminated environments including manure. Dairy cows often lie in areas heavily contaminated with manure, allowing easy access of bacteria to the udder and teat canal. Environmental bacteria such as Streptococcus uberis and Streptococcus dysgalactiae are common in the cow’s surroundings, and most infections caused by these pathogens are clinical and of short duration [13]. Pathogens may be transmitted through multiple routes, including contaminated milk, poor milking hygiene, bedding, feces, urine, and other environmental contaminants. Environmental mastitis occurs when microorganisms present in the animal’s surroundings gain access to the udder through the teat canal, with feces being the primary source of environmental pathogens [14]. Approximately 87% of mastitis cases are associated with udder or teat injuries, indicating poor udder health management. Barbed wire fencing and ticks around the udder have been identified as major causes of udder and teat injuries on farms [25]. Sand piles may also serve as reservoirs for Mycoplasma mastitis [17].
Diagnosis
Clinical mastitis can be easily diagnosed based on changes in milk color and texture or the presence of blood, pus, flakes, and clots. In contrast, subclinical mastitis is difficult to diagnose due to the absence of visible clinical signs and requires laboratory-based investigations. Milk samples may be cultured, and organisms isolated, stained, and identified using molecular techniques such as PCR for confirmatory diagnosis [26].
Correct identification of infected cows is fundamental for mastitis control and treatment. Therefore, simple, low-cost, and accurate sampling and diagnostic procedures tailored to specific epidemiological scenarios are required, as these may vary between regions and farms [27]. Early disease detection minimizes tissue damage, prompting the development of reliable on-farm diagnostic tools. Various diagnostic methods include visual examination, direct and indirect tests, California Mastitis Test (CMT), somatic cell count (SCC), resazurin tests, stir cup test, bromothymol blue test, modified Whiteside test, Wisconsin mastitis test, electrical conductivity test, and culture methods [28].
Somatic cell count remains a key indicator of intramammary infection. The California Mastitis Test is a simple, rapid, and inexpensive cow-side test for estimating SCC, based on gel formation from DNA released by disrupted somatic cells. However, CMT results from freshly calved cows are difficult to interpret, and the test should not be used before four days post-calving [29]. Failure to isolate bacteria from CMT-positive samples may result from spontaneous elimination of infection, low pathogen concentration, intermittent shedding, intracellular pathogen localization, or inhibitory substances in milk. In some cases, bacteria may disappear while leukocyte infiltration persists during healing [25].
Advanced diagnostic techniques such as PCR, nucleotide sequencing, and ELISA-based biomarker detection are increasingly used due to their high specificity and rapid turnaround, although they are limited by cost, infrastructure requirements, and technical expertise [30]. PCR and microarray platforms enable rapid and simultaneous detection of viral, yeast, and fungal pathogens involved in bovine mastitis [20].
Treatment
The success of mastitis treatment depends on the causative microorganism, udder environment, and milking system (hand or machine). In addition to parenteral antibiotic therapy, the use of non-steroidal anti-inflammatory drugs has been shown to reduce somatic cell counts, milk yield losses, clinical severity, and culling rates. NSAIDs are particularly effective as supportive therapy in E. coli mastitis [31]. Among mastitis pathogens, Staphylococcus aureus poses a major challenge due to its unique pathogenesis, contagious nature, environmental persistence, ability to colonize skin and mucosa, and poor response to antimicrobial therapy [32].
Infections during the dry period adversely affect subsequent lactation, emphasizing the importance of udder health management before drying off. Cows should be screened for mastitis using the California Mastitis Test, and intramammary antibiotic therapy combined with teat sealants should be applied immediately after the last milking to provide a physical barrier against bacterial invasion. Although vaccination against bovine mastitis remains challenging due to the diversity of causative agents, S. aureus, S. uberis, and E. coli are considered major targets for vaccine development [33–35].
Patho-Pathogenesis
Pathogenesis
After milking, the teat canal may remain open for one to two hours, leaving the teat end exposed to environmental pathogens. Infection generally occurs through the teat canal, where microbes find favorable conditions to multiply, damaging the lining of the milk ducts, cistern, and alveoli [36]. Systemic and local inflammatory responses alter metabolism, reduce milk production, and increase culling rates in lactating dairy cows, with more aggressive responses observed during the peripartum period compared with late lactation [37].
The teat canal’s keratin layer acts as the first line of defense against pathogen entry. Microbes can invade via the teat canal or, less commonly, through the bloodstream, multiply within the udder, infect glandular tissue, and trigger inflammation, leading to tissue injury, reduced cell numbers, and secondary necrosis [38]. Mammary infection may release harmful toxins, causing lesions that range from increased somatic cell counts without gross changes to increased vascular permeability or progressive fibrosis. Milk somatic cells—neutrophils, macrophages, lymphocytes, and epithelial cells—play a key role in early infection control. However, excessive leukocyte infiltration can damage mammary parenchyma and reduce milk yield [39].
Although apoptosis is a normal process in the mammary gland, infection significantly increases cellular loss. In E. coli infections, proapoptotic factors are upregulated and antiapoptotic factors downregulated; similar effects are observed with Staphylococcus aureus in vitro. Apoptotic cells are cleared by macrophages. Following bacterial entry, white blood cells and polymorphonuclear (PMN) leukocytes migrate to the infection site, killing microbes through oxygen-dependent and independent mechanisms. Activated PMNs, interleukins, and necrotic factors may contribute to mammary tissue damage. Plasminogen activators from PMNs, along with bacterial plasminogen receptors, convert plasminogen to plasmin, causing epithelial membrane degradation, sodium leakage, and tissue injury. Virulence often involves multiple toxins, proteins, and modulators leading to cell death [40,41].
Each mammary gland has a self-contained ductal network; quarters are isolated from each other by the medial suspensory ligament. In lipopolysaccharide-induced mastitis, systemic acute-phase responses include elevated body temperature, heart rate, and respiratory rate [37]. Environmental fungi, such as Candida spp., can infect the mammary gland when hygiene is poor or equipment is inadequately cleaned. Overuse of antibiotics may reduce vitamin A levels, injuring epithelial tissue and disrupting protective microflora, facilitating fungal invasion. Fungal infections, including Aspergillus fumigatus and Candida spp., may cause severe disease or death. These infections often arise from ascending infection following improper antibiotic administration during the drying-off period [42].
Viruses, including bovine leukemia virus, bovine herpesvirus types 1 and 4, foot-and-mouth disease virus, and parainfluenza virus 3, have been detected in mammary tissue or milk. Viral replication in mammary tissue can lead to clinical or subclinical mastitis, indirectly promoting secondary bacterial infections, such as Arcanobacter pyogenes. Transmission occurs via direct contact and aerosols [37,41,43].
Pathological lesions
Mastitis-affected mammary tissue exhibits marked inflammation, reduction of alveolar epithelium and lumen, increased stromal connective tissue, and leukocytosis. Lesion patterns vary due to the multifactorial etiology of mastitis. Histopathologically, mastitis can be classified into seven categories: mixed, lymphoplasmacytic, suppurative, pyogranulomatous, abscedative, necrosuppurative, and granulomatous mastitis [37,41,44,45].
Mixed mastitis
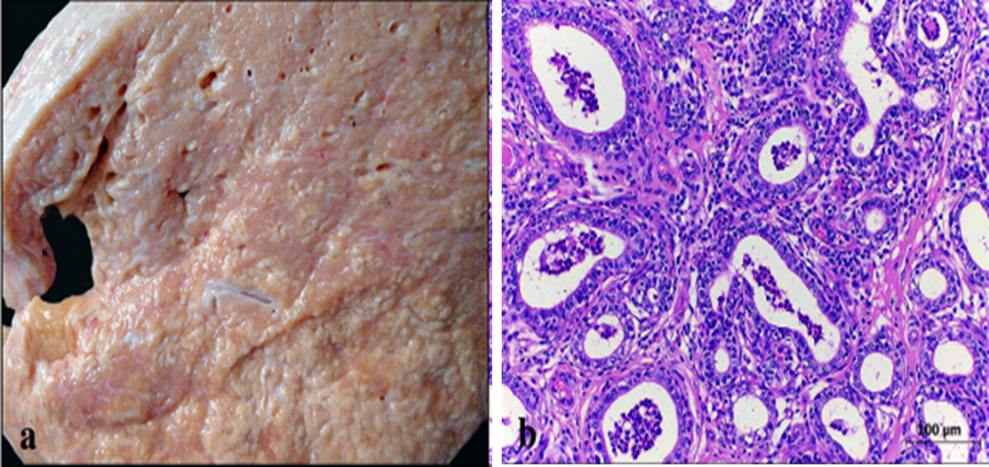
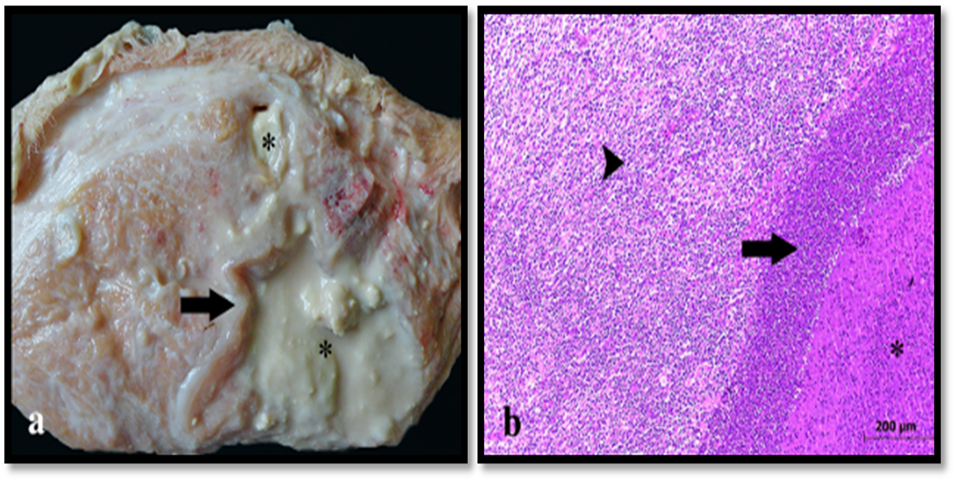
Grossly, mixed mastitis is characterized by a pronounced lobular pattern with small yellowish nodules (0.2–0.5 cm) projecting toward the ducts and cistern, interspersed with thin white septa. Histologically, there is a discrete to moderate inflammatory infiltrate of neutrophils in alveoli and ducts, with multifocal infiltration of neutrophils, lymphocytes, plasma cells, and macrophages in the interstitium (Figure 1) [37,46,47].
Figure 1. Gross (a) and histopathological (b) lesion of mixed mastitis (H&E) [37].
Lymphoplasmacytic mastitis
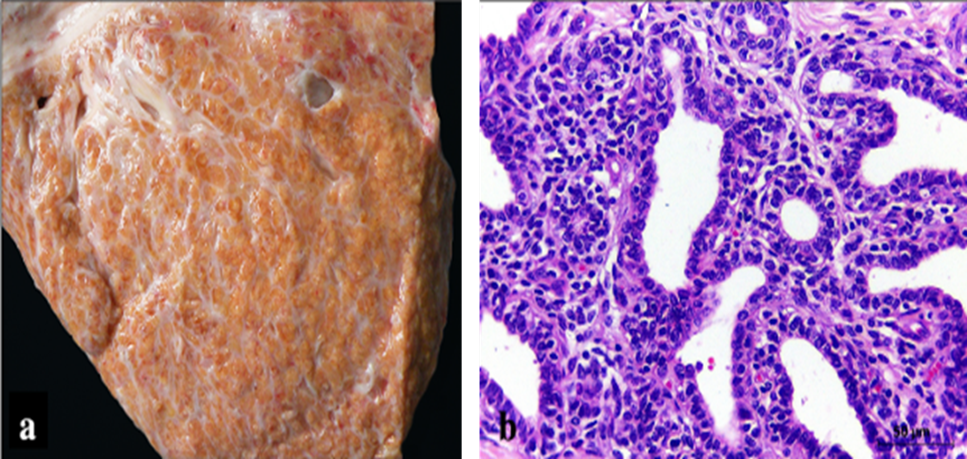
Grossly, firm mammary quarters with decreased lobulation and thick white septa dissecting the parenchyma are observed. Histologically, chronic interstitial mastitis shows diffuse infiltration of lymphocytes, plasma cells, and macrophages with moderate fibrosis. Nodular, white, polypoid structures may correspond to dilated alveoli with hyperplastic epithelium (Figure 2) [37,38,44,48,49].
Figure 2. Gross (a) and histopathological (b) lesion of lymphoplasmacytic (H&E) [38].
Pyogranulomatous mastitis
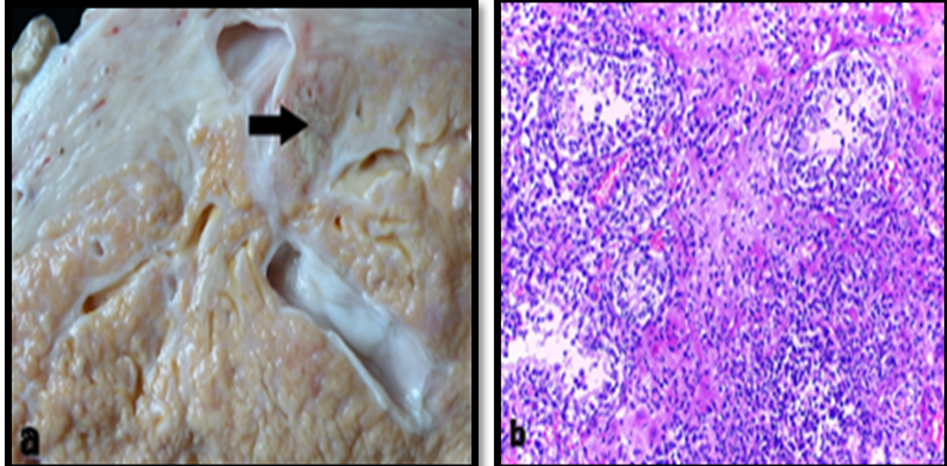
Bacteria and filamentous fungi may be detected. Nodular, yellowish, firm structures (0.5–1.5 cm) contain purulent material at the center and may be associated with Nocardia spp. or fungi. Histology shows multifocal necrosis, infiltration of intact and degenerate neutrophils, epithelioid macrophages, multinucleated giant cells, lymphocytes, and plasma cells with peripheral fibrosis (Figure 3) [37,38,42,50].
Figure 3. Gross (a) and histopathological (b) lesion of pyogranulomatous mastitis (H &E) [42].
Suppurative mastitis
Gross lesions are similar to mixed mastitis. Histologically, mild to moderate intact and degenerate neutrophils are observed in alveoli, ducts, and interstitium, with epithelial hyperplasia, degeneration, alveolar dilatation, fibrosis, and occasional bacteria (Figure 4) [37,38].
Figure 4. Gross (a) and histopathological (b) lesion of suppurative mastitis (H&E) [38].
Abscedative mastitis
Gross lesions show single or multiple abscesses in the mammary parenchyma. Histologically, areas of necrosis with bacteria and marked inflammatory infiltrate of neutrophils, macrophages, and lymphocytes are observed, often with thick fibrous capsules and squamous metaplasia (Figure 5) [37,38].
Figure 5. Gross (a) and histopathological (b) lesion of abscedative mastitis (H&E) [37].
Necrosuppurative mastitis
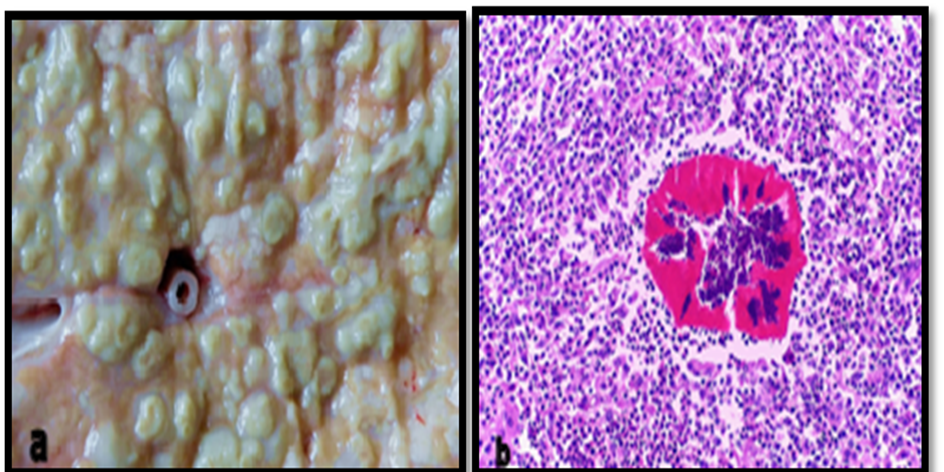
Gross lesions include yellowish areas filled with purulent material, sometimes with subcutaneous edema. Histology shows multifocal coagulation necrosis with marked neutrophil infiltration (Figure 6) [37,43,51].
Figure 6. Gross (a) and histopathological (b) lesion of necrosuppurative mastitis (H&E) [43].
Granulomatous mastitis
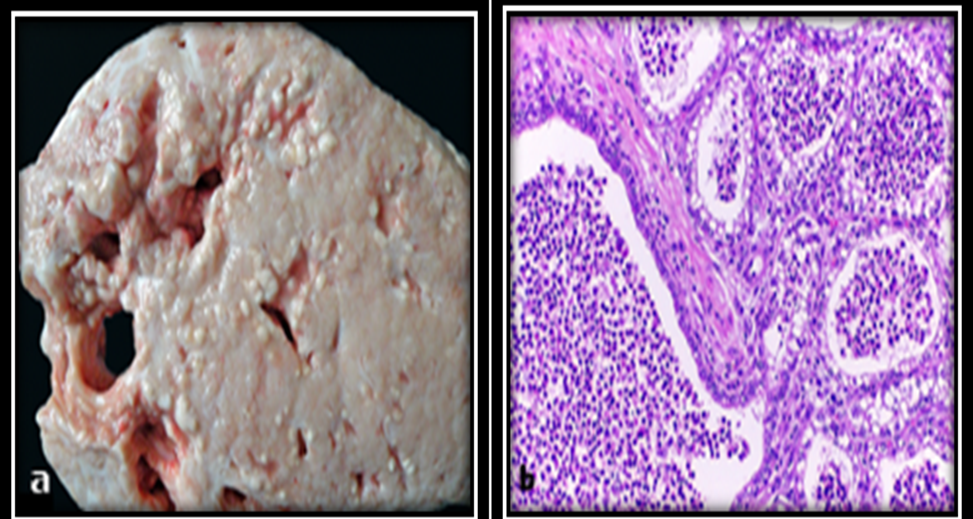
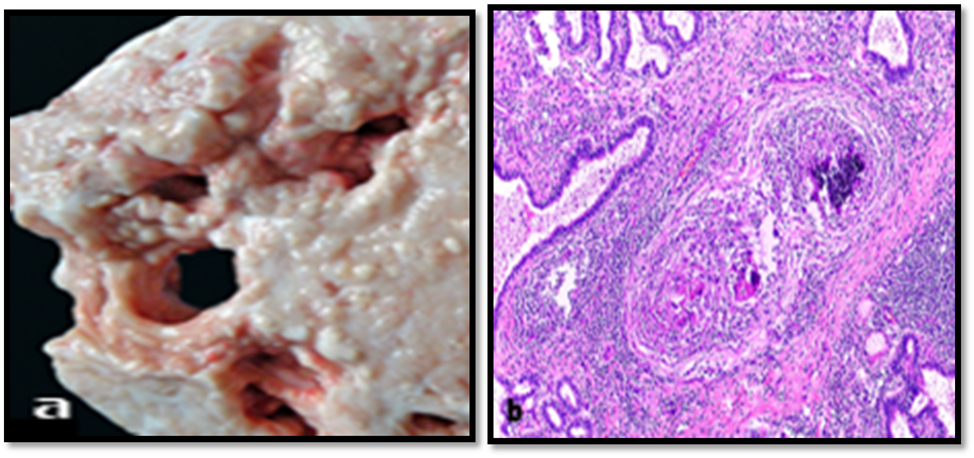
Multiple granulomas in the mammary parenchyma with small necrotic areas are observed. Acid-fast bacilli may be detected via ZN stain, morphologically compatible with Mycobacterium spp (Figure 7) [37,38,45,52].
Figure 7. Gross (a) and histopathological (b) lesion of granulomatous mastitis, H&E [45].
Conclusion
Bovine mastitis is a complex, multifactorial disease driven by a diverse spectrum of microorganisms, including over 137 bacterial species, alongside viral and fungal agents. It may present as acute or chronic, with most cases manifesting as mild clinical or subclinical inflammation, while severe infections can result in systemic complications or mortality. Histopathologically, mastitis is classified into seven categories—mixed, lymphoplasmacytic, suppurative, pyogranulomatous, abscedative, necrosuppurative, and granulomatous—reflecting the stage of infection and the causative pathogen. Comprehensive understanding of these pathological alterations is essential for accurate diagnosis, staging, and prognosis. Effective control relies on precise pathogen identification, as fungal or viral involvement may compromise antibiotic efficacy. Early recognition of gross and histopathological changes, combined with timely therapeutic intervention and sound management practices, is pivotal to mitigating the prevalence, severity, and economic burden of bovine mastitis.
Acknowledgments
Not applicable.
Author Contributions
All authors contributed equally and approved it.
Conflicts of Interest
The author declares no conflicts of interest.
References
2. Ballou MA. Growth and Development Symposium: Inflammation: Role in the etiology and pathophysiology of clinical mastitis in dairy cows. J Anim Sci. 2012 May;90(5):1466–78.
3. Wellenberg GJ, van der Poel WH, Van Oirschot JT. Viral infections and bovine mastitis: a review. Vet Microbiol. 2002 Aug 2;88(1):27–45.
4. Rinaldi M, Li RW, Capuco AV. Mastitis associated transcriptomic disruptions in cattle. Vet Immunol Immunopathol. 2010 Dec 15;138(4):267–79.
5. Ashraf A, Imran M. Diagnosis of bovine mastitis: from laboratory to farm. Trop Anim Health Prod. 2018 Aug;50(6):1193–202.
6. Cheng WN, Han SG. Bovine mastitis: risk factors, therapeutic strategies, and alternative treatments - A review. Asian-Australas J Anim Sci. 2020 Nov;33(11):1699–713.
7. Sharun K, Dhama K, Tiwari R, Gugjoo MB, Iqbal Yatoo M, Patel SK, et al. Advances in therapeutic and managemental approaches of bovine mastitis: a comprehensive review. Vet Q. 2021 Dec;41(1):107–36.
8. Rudenko P, Sachivkina N, Vatnikov Y, Shabunin S, Engashev S, Kontsevaya S, et al. Role of microorganisms isolated from cows with mastitis in Moscow region in biofilm formation. Vet World. 2021 Jan;14(1):40–8.
9. Hota A, Jambagi K, Pritam A, Mishra UN, Rout S, Barik N. MASTITIS IN CATTLE-ITS PATHOPHYSIOLOGY AND MECHANISM OF INFECTION. Journal of the Maharaja Sayajirao University of Baroda ISSN.;25:0422.
10. Sharma N, Singh NK, Bhadwal MS. Relationship of somatic cell count and mastitis: An overview. Asian-Australasian Journal of Animal Sciences. 2011 Feb 22;24(3):429–38.
11. Birhanu M, Leta S, Mamo G, Tesfaye S. Prevalence of bovine subclinical mastitis and isolation of its major causes in Bishoftu Town, Ethiopia. BMC Res Notes. 2017 Dec 21;10(1):767.
12. Abd El-Razik KA, Abdelrahman KA, Abd El-Moez SI, Danial EN. New approach in diagnosis and treatment of bovine mycotic mastitis in Egypt. African Journal of Microbiology Research. 2011 Dec 23;5(31):5725–32.
13. Goulart DB, Mellata M. Escherichia coli Mastitis in Dairy Cattle: Etiology, Diagnosis, and Treatment Challenges. Front Microbiol. 2022 Jul 7;13:928346.
14. Andreeva AA, Evgrafova VA, Voronina MS, Pruntova OV, Shadrova NB. Etiology and epizootology of bovine mastitis (analytical review). Veterinary Science Today. 2024 Mar 15;13(1):27–35.
15. Yu W, Zhang Z, Wang Z, Lin X, Dong X, Hou Q. Comprehensive Prevention and Control of Mastitis in Dairy Cows: From Etiology to Prevention. Vet Sci. 2025 Aug 23;12(9):800.
16. Contreras GA, Rodríguez JM. Mastitis: comparative etiology and epidemiology. J Mammary Gland Biol Neoplasia. 2011 Dec;16(4):339–56.
17. Fox L. Mycoplasma Mastitis in Dairy Cattle: Pathogenesis and Control. 1st ed. New York: Springer; 2012.
18. Cobirka M, Tancin V, Slama P. Epidemiology and Classification of Mastitis. Animals (Basel). 2020 Nov 26;10(12):2212.
19. Khan MZ, Khan A. Basic facts of mastitis in dairy animals: A review. Pakistan Veterinary Journal. 2006 Oct 1;26(4):204.
20. Hill AW, Shears AL, Hibbitt KG. The pathogenesis of experimental Escherichia coli mastitis in newly calved dairy cows. Res Vet Sci. 1979 Jan;26(1):97–101.
21. Ashraf A, Imran M. Causes, types, etiological agents, prevalence, diagnosis, treatment, prevention, effects on human health and future aspects of bovine mastitis. Anim Health Res Rev. 2020 Jun;21(1):36–49.
22. Meaza B. Laboratory and field diagnostic methods for bovine mastitis. Int J Vet Sci. 2016;5(2):45–55.
23. Chakraborty S, Dhama K, Tiwari R, Iqbal Yatoo M, Khurana SK, Khandia R, et al. Technological interventions and advances in the diagnosis of intramammary infections in animals with emphasis on bovine population-a review. Vet Q. 2019 Dec;39(1):76–94.
24. Bogni C, Odierno L, Raspanti C, Giraudo J, Larriestra A, Reinoso E, et al. War against mastitis: Current concepts on controlling bovine mastitis pathogens. Science against microbial pathogens: Communicafing Current Research and Technological Advances. 2011:483–94.
25. Dego OK, Tareke F. Bovine mastitis in selected areas of southern Ethiopia. Trop Anim Health Prod. 2003 Jun;35(3):197–205.
26. Haider A, Ikram M, Shahzadi I, Raza MA. Polymeric Nanoparticles for Bovine Mastitis Treatment. Springer; 2023 Sep 22.
27. Kumar P, Ojasvita AD, Sharma H, Sharma S, Mittal D, Bhanot V, et al. Bovine mastitis: a review. Middle East J Sci Res. 2020;28(6):497–507.
28. Heikkilä AM, Liski E, Pyörälä S, Taponen S. Pathogen-specific production losses in bovine mastitis. J Dairy Sci. 2018 Oct;101(10):9493–504.
29. El-Sayed A, Kamel M. Bovine mastitis prevention and control in the post-antibiotic era. Trop Anim Health Prod. 2021 Mar 31;53(2):236.
30. Kitchen BJ. Review of the progress of dairy science: bovine mastitis: milk compositional changes and related diagnostic tests. J Dairy Res. 1981 Feb;48(1):167–88.
31. Holmes MA, Zadoks RN. Methicillin resistant S. aureus in human and bovine mastitis. J Mammary Gland Biol Neoplasia. 2011 Dec;16(4):373–82.
32. Tomanić D, Samardžija M, Kovačević Z. Alternatives to Antimicrobial Treatment in Bovine Mastitis Therapy: A Review. Antibiotics (Basel). 2023 Mar 30;12(4):683.
33. Mimoune N, Saidi R, Benadjel O, Khelef D, Kaidi R. Alternative treatment of bovine mastitis. Veterinarska Stanica. 2021 May 2;52(6):639–49.
34. Contreras A, Sierra D, Sánchez A, Corrales JC, Marco JC, Paape MJ, et al. Mastitis in small ruminants. Small Ruminant Research. 2007 Mar 1;68(1-2):145–53.
35. Hogan J, Larry Smith K. Coliform mastitis. Vet Res. 2003 Sep-Oct;34(5):507–19.
36. Zadoks R, Fitzpatrick J. Changing trends in mastitis. Ir Vet J. 2009 Apr 1;62 Suppl 4(Suppl 4):S59–70.
37. Rainard P, Riollet C. Innate immunity of the bovine mammary gland. Vet Res. 2006 May-Jun;37(3):369–400.
38. Oliver SP, Murinda SE. Antimicrobial resistance of mastitis pathogens. Vet Clin North Am Food Anim Pract. 2012 Jul;28(2):165–85.
39. Ruegg PL. A 100-Year Review: Mastitis detection, management, and prevention. J Dairy Sci. 2017 Dec;100(12):10381–97.
40. Tommasoni C, Fiore E, Lisuzzo A, Gianesella M. Mastitis in Dairy Cattle: On-Farm Diagnostics and Future Perspectives. Animals (Basel). 2023 Aug 6;13(15):2538.
41. Sordillo LM, Streicher KL. Mammary gland immunity and mastitis susceptibility. J Mammary Gland Biol Neoplasia. 2002 Apr;7(2):135–46.
42. Smith KL, Todhunter DA, Schoenberger PS. Environmental mastitis: cause, prevalence, prevention. J Dairy Sci. 1985 Jun;68(6):1531–53.
43. Pyörälä S. Indicators of inflammation in the diagnosis of mastitis. Vet Res. 2003 Sep-Oct;34(5):565–78.
44. Huszenicza G, Jánosi S, Gáspárdy A, Kulcsár M. Endocrine aspects in pathogenesis of mastitis in postpartum dairy cows. Anim Reprod Sci. 2004 Jul;82-83:389–400.
45. Phiri AM, Phiri IK, Monrad J. Prevalence of amphistomiasis and its association with Fasciola gigantica infections in Zambian cattle from communal grazing areas. J Helminthol. 2006 Mar;80(1):65–8.
46. Kerro Dego O, Vidlund J. Staphylococcal mastitis in dairy cows. Front Vet Sci. 2024 May 28;11:1356259.
47. Hota A, Jambagi K, Swain S. Bovine mastitis: pathogenesis and susceptibility. Agro Economist-An International Journal. 2020 Nov 24;7:107–10.
48. Boireau C, Cazeau G, Jarrige N, Calavas D, Madec JY, Leblond A, et al. Antimicrobial resistance in bacteria isolated from mastitis in dairy cattle in France, 2006-2016. J Dairy Sci. 2018 Oct;101(10):9451–62.
49. Kim H, Min Y, Choi B. Real-time temperature monitoring for the early detection of mastitis in dairy cattle: Methods and case researches. Computers and Electronics in Agriculture. 2019 Jul 1;162:119–25.
50. Temesgen AB, Shiferaw SA. Antimicrobial Multidrug Resistance and Mechanisms of Action: An Overview. Biomed Res Int. 2025 Nov 25;2025:8847267.
51. Koba IS, Lysenko AA, Koshchaev AG, Shantyz AK, Donnik IM, Dorozhkin VI, et al. Prevention of mastitis in dairy cows on industrial farms. Journal of Pharmaceutical Sciences and Research. 2018 Oct 1;10(10):2582–5.
52. Bramley AJ, Neave FK. Studies on the control of coliform mastitis in dairy cows. Br Vet J. 1975 Mar-Apr;131(2):160–9.