Abstract
Tumor-derived exosomes (TDES) play a vital role in facilitating cancer development and dissemination through the trafficking of oncogenic nucleic acids and proteins, such as cytokines and chemokines, which remodel the tumor microenvironment (TME) to facilitate malignant traits. This process ultimately contributes to tumorigenesis, invasion, metastasis, and treatment resistance. Therefore, restricting cancer-induced communication with healthy cells by eliminating TDEs may serve as a promising approach to curb cancer progression and dissemination. In this commentary, we discuss the pivotal role of tumor-derived exosomes (TDEs) in cancer progression and metastasis, and propose a novel therapeutic strategy: an anti-TDE vaccine. Building on our preliminary work, which showed that pantoprazole can inhibit tumor cell proliferation by disrupting TDE secretion and altering the surface charge of secreted TDEs, we elaborate on a duplex approach to engineer tumors to produce 'tagged' TDEs that are both poorly absorbed and targeted for immune clearance. This novel approach can overcome most of the challenges of pharmacological inhibition, including off-target effects, wide diversity of biogenesis pathways, and drug delivery and specificity.
Keywords
Tumor-derived exosomes (TDEs), Oncogenic communication, Targeting TDEs, Anti-TDE vaccine
Introduction
Exosomes are lipid-bilayered extracellular vesicles (30–150 nm) secreted by all cells—from prokaryotes to highly specialized eukaryotes serving as critical mediators of both local and systemic intercellular communication [1–4]. They transport bioactive cargo; proteins, nucleic acids, and lipids—from donor cells to recipient cells, thereby reprogramming the phenotype and function of target cells [5–7]. Under physiological conditions, exosomes released by healthy cells deliver regulatory signals that maintain homeostasis and coordinate essential biological processes [8]. In contrast, malignant cells secrete aberrant exosomes known as tumor-derived exosomes (TDEs), which shuttle oncoproteins, non-coding RNAs, and metabolites that drive cancer progression [9–11], as exemplified by pancreatic-cancer exosomes that drive neoplastic transformation of normal recipient cells [12].
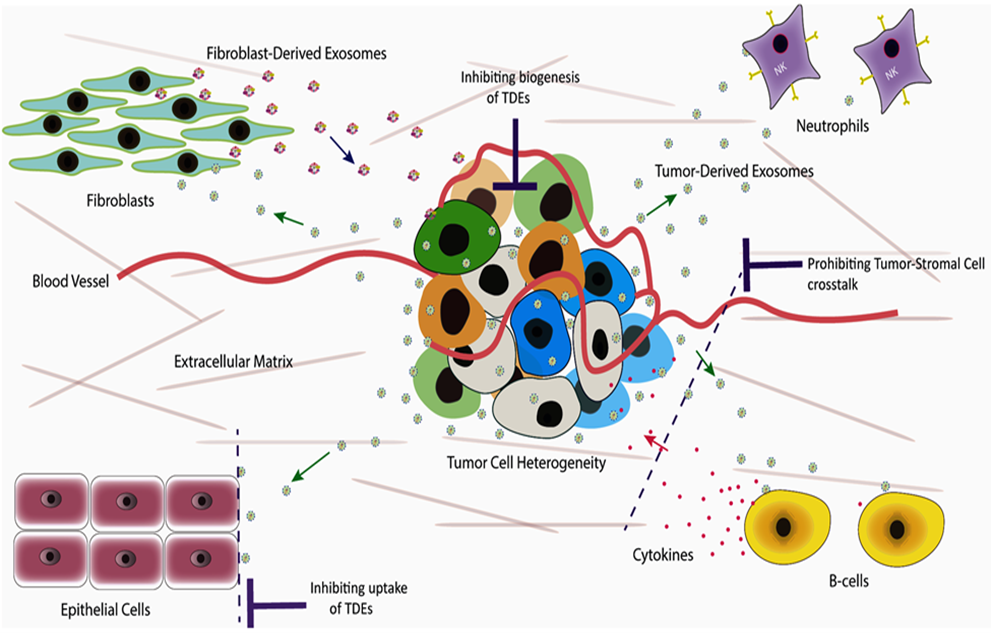
Cancer remains a leading cause of global mortality, with metastasis accounting for the vast majority of cancer-related deaths [13–16]. This complex process persists as a major therapeutic challenge, driven significantly by intricate interactions between cancerous and non-cancerous cells within the tumor microenvironment (TME) [17,18]. Recent evidence underscores the pivotal role of TDEs in mediating communication within this cancer ecosystem. TDEs profoundly influence tumor invasion, progression, survival, metastasis, and resistance to therapy [18,19]. Consequently, disrupting this oncogenic signaling network represents a critical therapeutic imperative. A novel strategy targeting TDEs—by limiting their release from cancer cells and inhibiting their uptake by recipient cells—holds significant promise. Such intervention is expected to substantially suppress tumor growth and dissemination while simultaneously enhancing cancer cell sensitivity to therapeutic agents.
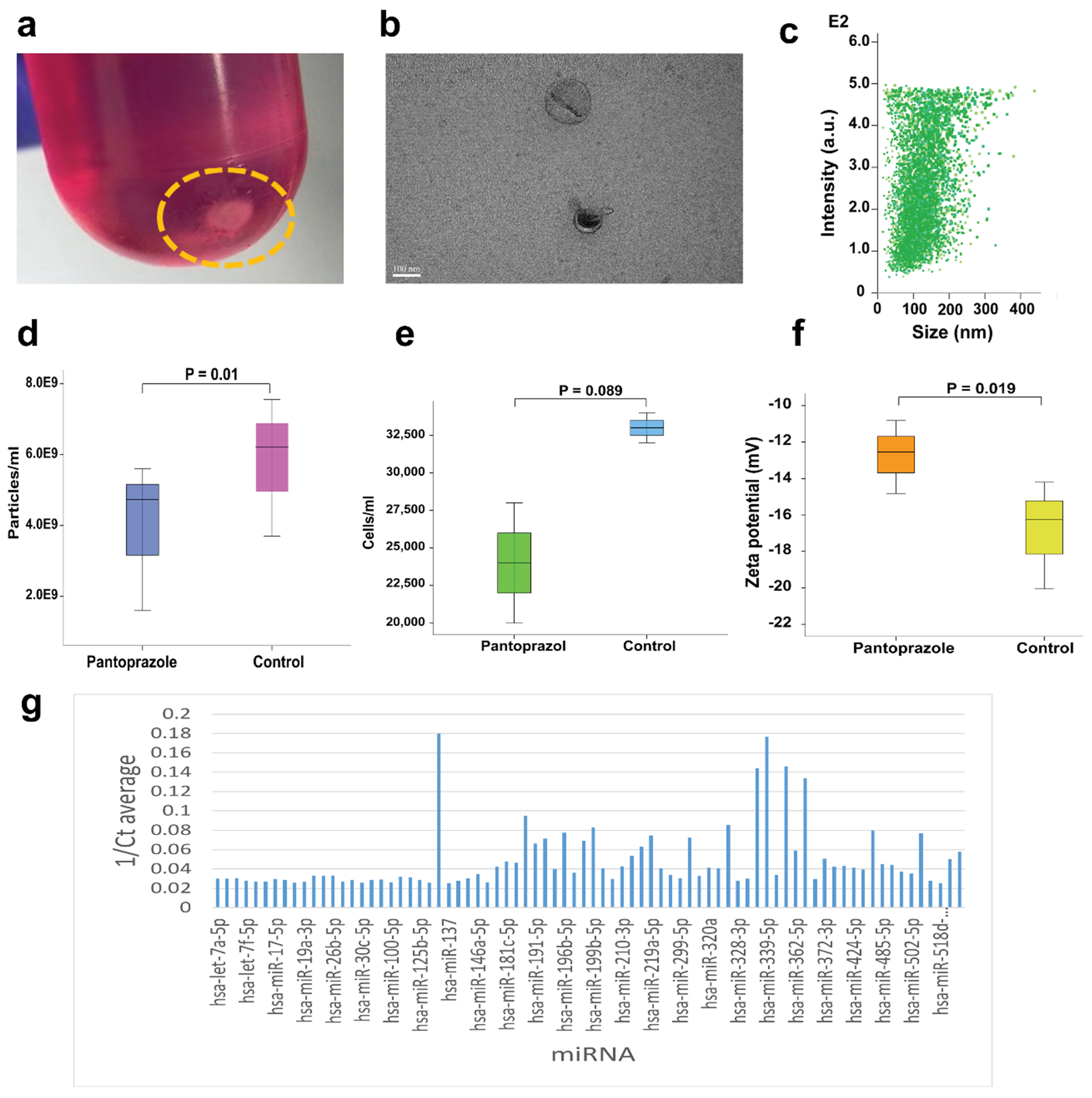
In our previous research investigation [20], we developed a laboratory-based study primarily focused on evaluating the impact of pantoprazole, a proton pump inhibitor (PPI), on the suppression of exosomes derived from triple negative breast cancer cells (MDA –MB-231), consequently limiting tumor growth. The impact of PPI on the suppression of breast cancer-derived exosomes (TDEs) secretion and the growth of MDA MB-231 cells was assessed. Therefore, MDA MB-231 cells were cultivated in a conditioned culture medium (CCM) with or without pantoprazole (100 μg/ml) for a duration of 24 hours. In the experimental group, 2 x 10^3 MDA MB-231 cells were treated with 100 µg/ml of pantoprazole. Conversely, the control group consisted of the same number of cells that were maintained without pantoprazole treatment. Each group underwent three repetitions of the experiments. For the analysis of TDEs, the CCM from both groups was collected 24 hours after the treatment of cells with pantoprazole in the experimental group, while the control group remained untreated with pantoprazole. TDEs were extracted from the supernatant of cell cultures using an ultracentrifugation method, and the resulting pellets (Figure 1a) were assessed. Following the isolation of TDEs, various characterization techniques were employed, such as visualizing TDE morphology through transmission electron microscopy (TEM) (Figure 1b) and assessing particle size distribution via nanoparticle tracking analysis (NTA) (Figure 1c). The preliminary results of our study suggest that pantoprazole significantly decreased the secretion of TDEs (Figure 1d) and suppressed the growth of MDA MB-231 cells (Figure 1e). Furthermore, pantoprazole modified the surface charge of TDEs (Figure 1f), making them less negatively charged. This alteration subsequently disrupted the electrostatic interaction between TDEs and target cells, thus impeding their selective uptake. This is because the presence of a negative surface charge on exosomes influences binding by enhancing interactions with positively charged components on the membranes of target cells [21,22]. Conversely, the analysis of the molecular content carried by TDEs indicated a high presence of miRNAs linked to cancer (Figure 1f).
These findings add pantoprazole to a growing list of pharmacological agents that have shown the ability to inhibit the secretion of TDEs. Several challenges are hindering the clinical efficacy of pharmacological inhibition of TDE secretion, including limited target specificity, wide diversity of biogenesis pathways, pharmacokinetics and drug delivery challenges, off-target effects and toxicity, and drug resistance [23–25]. Therefore, innovative strategies that can inhibit TDE secretion or uptake while overcoming the limitations of pharmacological inhibition are needed to harness the potential of these pathways to control and eradicate tumors.
Figure 1. Characterization of TDEs. a. Pellets of TDES obtained from the breast cancer cell line (MDA MB-231) through ultracentrifugation. b. TEM image verifying that the particles we isolated were indeed TDES. c. NTA image illustrating the size distribution of TDEs. d. Comparison of TDEs concentration between the experimental group and the control group. e. Number of MDA MB-231 cells cultured in both the experimental and control groups. f. Surface charge of TDEs isolated from the cell culture supernatant, treated with PPI and without PPI. g. List of miRNAs extracted from TDES.
Novel Approaches of Targeting TDEs to Restrict Cancer Progression and Metastasis
In our recently published paper [26], we introduced an innovative approach for creating anti-TDE vaccines to manage cancer progression and metastasis. This approach can overcome most hurdles to pharmacological inhibition of TDE biogenesis and secretion. The anti-TDE vaccine functions by disrupting the TDE-mediated transfer of oncogenic signaling molecules to normal cells, thereby curtailing their pro-tumorigenic effects. The primary concept behind the development of an anti-TDE vaccine is to engineer solid tumors to generate modified TDEs that express a foreign antigen (antigen-tagged TDEs) on their surface, triggering an immune response. The activation of the immune response by the foreign antigen will prompt immune cells to eliminate all antigen-tagged TDEs [26]. By intercepting TDE-mediated communication, this approach aims to halt the tumor-promoting alterations in the microenvironment and sensitize cancers to therapy.
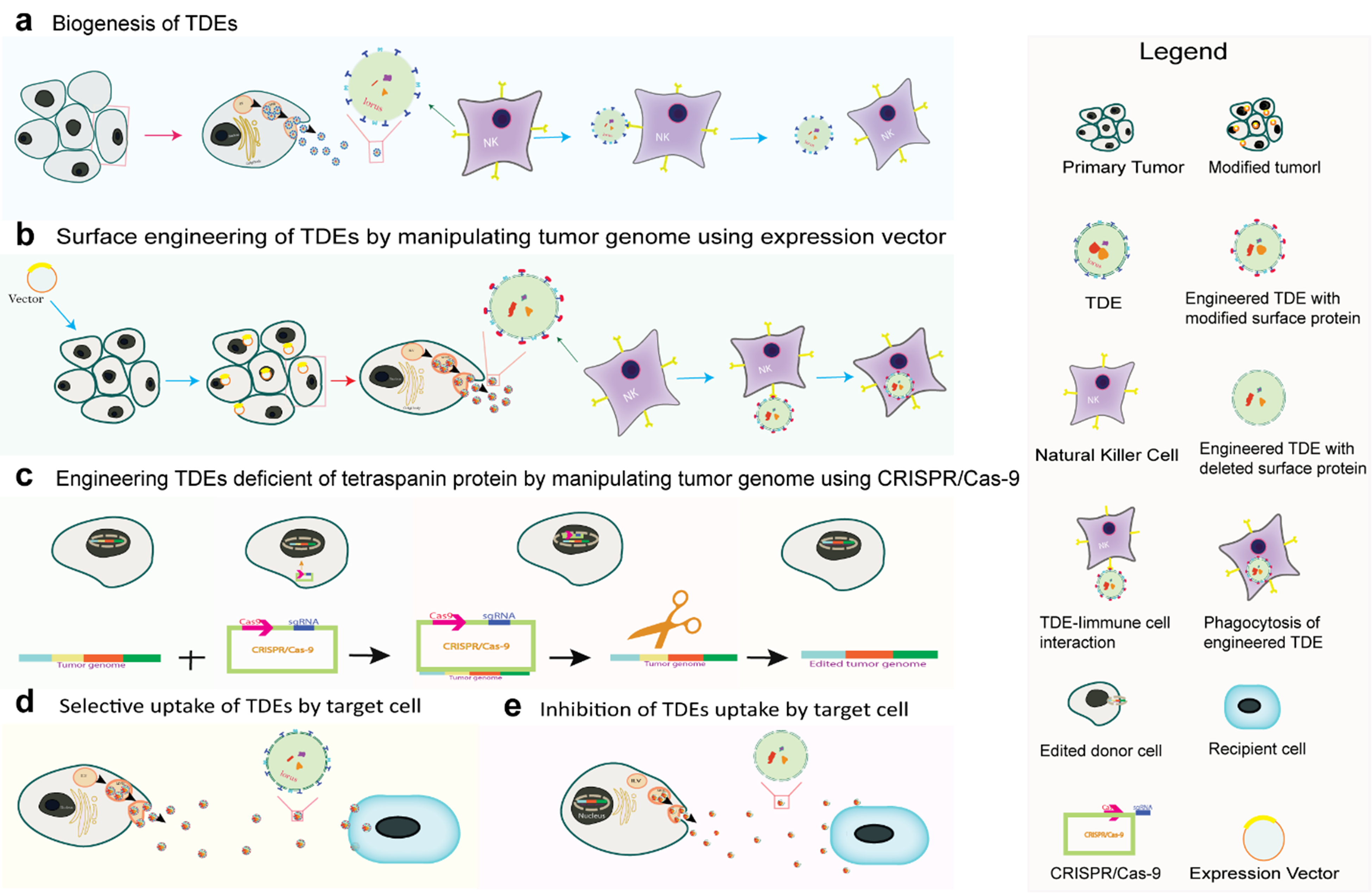
In addition to tagging TDEs with foreign antigens to elicit immune clearance, engineering strategies can be utilized to inhibit the uptake of TDEs by target cells by the knockout of specific genes that encode distinct transmembrane proteins predominantly expressed by TDEs to enhance their selective uptake by targeted recipient cells to potentiate the efficacy of the anti-TDE vaccine. This duplex strategy of modifying tumor cells to secrete TDEs lacking ligands that mediate their selective uptake by target cells, along with surface engineering of TDEs to display foreign antigens, will effectively obstruct TDE-mediated oncogenic communication (Figure 2).
Figure 2. Engineering of primary tumor cells to secrete immunogenic TDEs and TDEs deficient in a specific tetraspanin of interest. a. Formation and release of TDEs (non-immunogenic) by a tumor cell. b. Selective uptake of non-immunogenic TDEs secreted by a cancer cell. c. Vector-based surface engineering of TDEs expressing foreign antigen to induce an immune reaction. d. Engineering of TDEs deficient in the tetraspanin protein of interest using the CRISPR/Cas-9 gene editing system. e. Inhibition of the selective uptake of TDES by target cells due to the lack of transmembrane tetraspanin protein of interest that facilitates specific binding and internalization.
The inability of target cells to uptake antigen-tagged TDEs will lead to their accumulation in the extracellular space, making them available for immune clearance. Overall, the intended goal of the anti-TDE vaccine is to significantly inhibit the TDE-mediated transfer of oncogenic signals, thereby suppressing tumor growth, metastasis, and enhancing the sensitivity of cancer cells to oncotherapy.
Conclusion and Perspectives
Our research study has demonstrated that the suppression of TDEs secretion correlates with the inhibition of cancer cell growth. This
phenomenon is believed to be attributed to the disruption of the exchange of oncogenic signaling molecules. Therefore, the proposed intervention of developing anti-TDE vaccines is crucial for controlling tumor growth, preventing metastasis, and improving the responsiveness of cancer cells to conventional therapies. This strategy encompasses comprehensive measures aimed at targeting TDEs secretion, inhibiting the specific uptake of TDEs by recipient cells, and facilitating their elimination through immune surveillance/life-long immunity.
Several challenges need to be addressed before the clinical translation of the proposed strategies can be achieved. First, this strategy requires highly efficient tools to transduce tumor cells with genes encoding foreign proteins or to knock down genes encoding vital proteins required for TDE uptake. While several tools are available for cellular transduction, such as CRISPR/Cas9 systems and viral vectors, achieving such high transduction efficiencies remains challenging. Furthermore, several biological challenges remain, such as the possibility of autoimmune reactions triggered by tagged proteins and tumor cells acquiring the ability to stop producing the tag or secreting other types of vesicles, such as microvesicles that can have similar pro-tumorigenic effects as TDEs.
Future work should focus on testing the proposed strategy in relevant animal models before moving to translational research. For instance, tumors can be engineered in mouse models to evaluate the transduction efficiencies, characterize the incorporation of foreign protein tags in secreted TDEs, measure immunological markers of anti-TDE immunity and TDE clearance, as well as evaluate the effects on tumor growth and metastasis.
References
2. Turturici G, Tinnirello R, Sconzo G, Geraci F. Extracellular membrane vesicles as a mechanism of cell-to-cell communication: advantages and disadvantages. Am J Physiol Cell Physiol. 2014 Apr 1;306(7):C621–33.
3. Zaborowski MP, Balaj L, Breakefield XO, Lai CP. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience. 2015 Aug 1;65(8):783–97.
4. Isola AL, Chen S. Exosomes: The Messengers of Health and Disease. Curr Neuropharmacol. 2017;15(1):157–65.
5. Dioufa N, Clark AM, Ma B, Beckwitt CH, Wells A. Bi-directional exosome-driven intercommunication between the hepatic niche and cancer cells. Mol Cancer. 2017 Nov 14;16(1):172.
6. Mittelbrunn M, Sánchez-Madrid F. Intercellular communication: diverse structures for exchange of genetic information. Nat Rev Mol Cell Biol. 2012 Apr 18;13(5):328–35.
7. Fitzgerald W, Freeman ML, Lederman MM, Vasilieva E, Romero R, Margolis L. A System of Cytokines Encapsulated in ExtraCellular Vesicles. Sci Rep. 2018 Jun 12;8(1):8973.
8. Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020 Feb 7;367(6478):eaau6977.
9. Wee I, Syn N, Sethi G, Goh BC, Wang L. Role of tumor-derived exosomes in cancer metastasis. Biochim Biophys Acta Rev Cancer. 2019 Jan;1871(1):12–9.
10. Bai S, Wang Z, Wang M, Li J, Wei Y, Xu R, et al. Tumor-Derived Exosomes Modulate Primary Site Tumor Metastasis. Front Cell Dev Biol. 2022 Mar 2;10:752818.
11. Mashouri L, Yousefi H, Aref AR, Ahadi AM, Molaei F, Alahari SK. Exosomes: composition, biogenesis, and mechanisms in cancer metastasis and drug resistance. Mol Cancer. 2019 Apr 2;18(1):75.
12. Stefanius K, Servage K, de Souza Santos M, Gray HF, Toombs JE, Chimalapati S, et al. Human pancreatic cancer cell exosomes, but not human normal cell exosomes, act as an initiator in cell transformation. Elife. 2019 May 28;8:e40226.
13. Ma X, Yu H. Global burden of cancer. Yale J Biol Med. 2006 Dec;79(3-4):85–94.
14. Wu Z, Xia F, Lin R. Global burden of cancer and associated risk factors in 204 countries and territories, 1980-2021: a systematic analysis for the GBD 2021. J Hematol Oncol. 2024 Nov 29;17(1):119.
15. Mani K, Deng D, Lin C, Wang M, Hsu ML, Zaorsky NG. Causes of death among people living with metastatic cancer. Nat Commun. 2024 Feb 19;15(1):1519.
16. Dillekås H, Rogers MS, Straume O. Are 90% of deaths from cancer caused by metastases? Cancer Med. 2019 Sep;8(12):5574–6.
17. Biray Avci C, Goker Bagca B, Nikanfar M, Takanlou LS, Takanlou MS, Nourazarian A. Tumor microenvironment and cancer metastasis: molecular mechanisms and therapeutic implications. Front Pharmacol. 2024 Nov 12;15:1442888.
18. Baghban R, Roshangar L, Jahanban-Esfahlan R, Seidi K, Ebrahimi-Kalan A, Jaymand M, et al. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun Signal. 2020 Apr 7;18(1):59.
19. Ten A, Kumeiko V, Farniev V, Gao H, Shevtsov M. Tumor Microenvironment Modulation by Cancer-Derived Extracellular Vesicles. Cells. 2024 Apr 15;13(8):682.
20. Meena SS, Mrema A, Ndumbalo J, Chuwa HR, Soko G, Chambuso R, et al. Tumor-derived exosomes (TDEs) as communicators for cancer development and metastasis. J Clin Oncol. 2025 Jun, 43(16_suppl):e13031.
21. Buzás EI, Tóth EÁ, Sódar BW, Szabó-Taylor KÉ. Molecular interactions at the surface of extracellular vesicles. Semin Immunopathol. 2018 Sep;40(5):453–64.
22. Midekessa G, Godakumara K, Ord J, Viil J, Lättekivi F, Dissanayake K, et al. Zeta Potential of Extracellular Vesicles: Toward Understanding the Attributes that Determine Colloidal Stability. ACS Omega. 2020 Jun 30;5(27):16701–10.
23. Li Y, Chen ZK, Duan X, Zhang HJ, Xiao BL, Wang KM, et al. Targeted inhibition of tumor-derived exosomes as a novel therapeutic option for cancer. Exp Mol Med. 2022 Sep;54(9):1379–89.
24. Gabaran SG, Nejati V, Dilsiz N, Rezaie J. An updated review on the inhibition of exosome biogenesis, release, and uptake: a potential anticancer approach. Biochem Pharmacol. 2025 Sep;239:117019.
25. Kim JH, Lee CH, Baek MC. Dissecting exosome inhibitors: therapeutic insights into small-molecule chemicals against cancer. Exp Mol Med. 2022 Nov;54(11):1833–43.
26. Meena SS, Kosgei BK, Soko GF, Tingjun C, Chambuso R, Mwaiselage J, et al. Developing anti-TDE vaccine for sensitizing cancer cells to treatment and metastasis control. NPJ Vaccines. 2025 Jan 27;10(1):18.