Introduction
Acute myeloid leukemia (AML) is a biologically heterogeneous hematologic malignancy characterized by uncontrolled proliferation of immature myeloid cells and disruption of normal hematopoiesis. Despite major advances in genomic classification and the development of targeted therapies, long-term survival remains limited for many patients due to relapse and treatment resistance [1–3]. Despite therapeutic advances, 5-year overall survival remains approximately 30% in adult AML, underscoring the need for strategies that effectively target disease persistence. Increasing evidence indicates that relapse is driven by a rare subpopulation of leukemia-initiating cells known as leukemic stem cells (LSCs), which possess self-renewal capacity and the ability to reconstitute leukemia following therapy [4–7].
The concept of leukemic stem cells was first demonstrated in seminal xenotransplantation experiments showing that only a small subset of AML cells could initiate leukemia when transplanted into immunodeficient mice [6,7]. These cells display several features reminiscent of normal hematopoietic stem cells, including quiescence, self-renewal, and hierarchical differentiation. Subsequent studies have confirmed that AML is organized as a cellular hierarchy in which LSCs reside at the apex and generate downstream leukemic progenitors and blasts [8–10].
The persistence of LSCs during therapy is considered one of the principal drivers of disease relapse. Unlike bulk leukemic cells, LSCs possess intrinsic mechanisms that confer resistance to chemotherapy, including enhanced DNA repair, altered metabolism, and protective interactions with the bone marrow microenvironment [11–14]. Understanding these mechanisms has therefore become central to the development of curative therapies for AML.
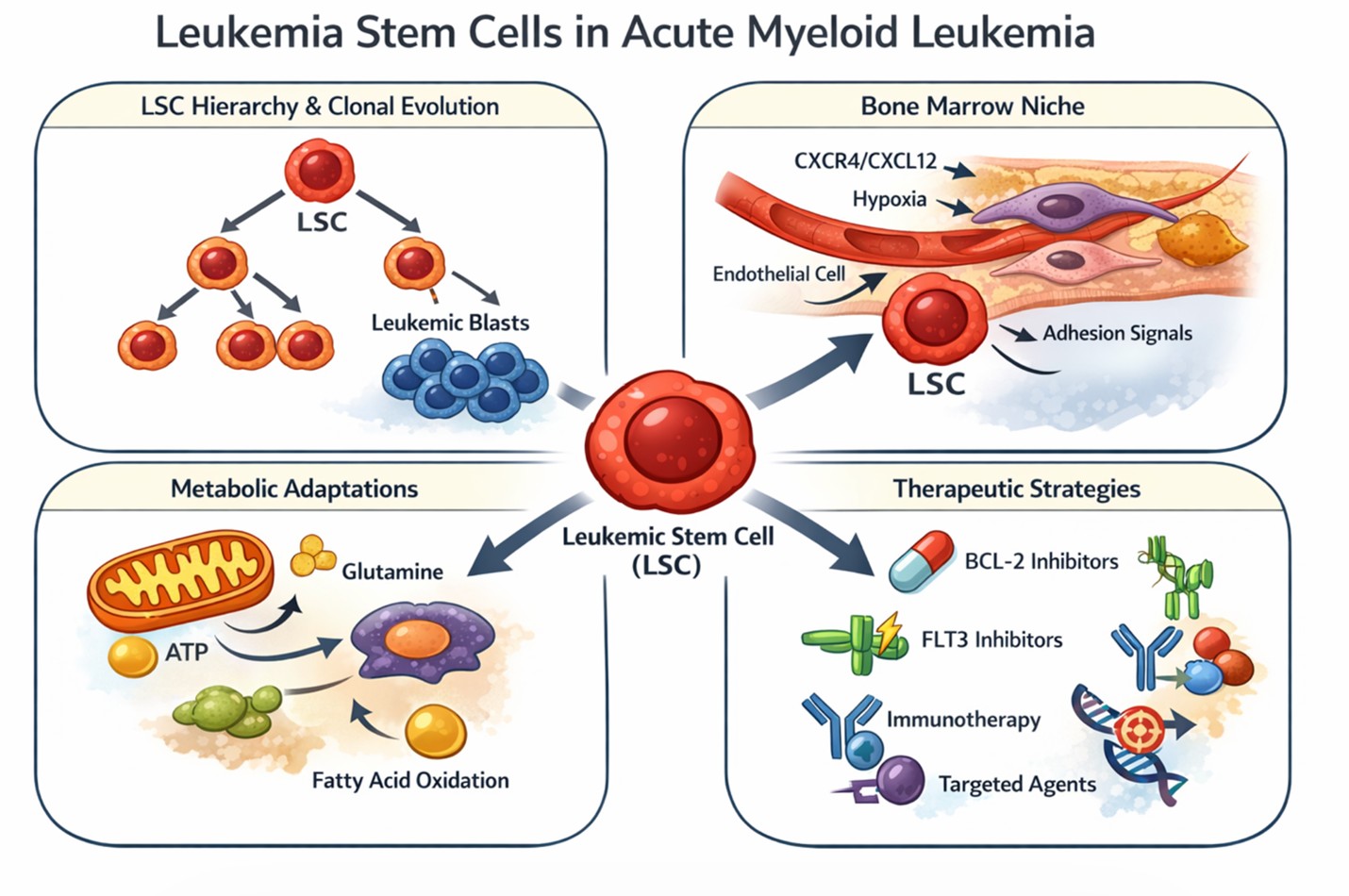
A conceptual overview of leukemic stem cell biology, including hierarchical organization, metabolic adaptations, microenvironmental interactions, and therapeutic resistance, is illustrated in Figure 1.
Figure 1. Conceptual model of leukemic stem cell biology and therapeutic resistance in AML.
AML is hierarchically organized, with leukemic stem cells (LSCs) at the apex generating progenitor populations and bulk leukemic blasts. LSCs reside within the bone marrow microenvironment, where interactions with stromal and endothelial components, along with adhesion signals and chemokine axes, promote retention and survival. Within this niche, hypoxic conditions and microenvironmental cues drive metabolic reprogramming, characterized by increased reliance on mitochondrial oxidative phosphorylation (OXPHOS), glutamine metabolism, and lipid utilization. These adaptations support LSC quiescence and enhance resistance to cytotoxic and targeted therapies. LSCs further evade therapy through intrinsic mechanisms, including altered apoptotic priming and activation of pro-survival signaling pathways. As a result, conventional and targeted treatments often eliminate bulk leukemic cells but fail to eradicate the LSC compartment, leading to disease persistence and relapse. Therapeutic strategies aimed at durable remission must therefore integrate approaches targeting metabolic dependencies, niche interactions, and survival pathways within LSCs.
Origin and Hierarchical Organization of Leukemic Stem Cells
AML arises through a multistep process involving the accumulation of genetic and epigenetic alterations in hematopoietic stem or progenitor cells. Early mutations may occur in long-lived hematopoietic stem cells, creating pre-leukemic clones that persist over time and provide a reservoir for further leukemogenic events [15–18]. Additional mutations affecting signaling pathways, transcription factors, and epigenetic regulators ultimately give rise to fully transformed leukemic stem cells capable of sustaining malignant hematopoiesis.
Functional studies have demonstrated that LSCs maintain the hierarchical organization of AML. These cells generate more differentiated leukemic progenitors, which in turn produce the bulk population of leukemic blasts observed in patients [19–22]. Importantly, only the LSC population retains long-term self-renewal capacity, making it the critical cellular target for effective therapy.
Clonal evolution further contributes to the complexity of AML stem cell biology. Distinct LSC subclones may coexist within a single patient, each characterized by unique mutational profiles and therapeutic sensitivities [23]. This heterogeneity contributes to treatment resistance and underscores the need for therapeutic strategies capable of targeting multiple LSC populations simultaneously.
Recent single-cell sequencing approaches have further highlighted the functional and phenotypic heterogeneity of LSC populations, including both CD34+ and CD34- subsets, with distinct transcriptional and therapeutic profiles.
Metabolic Adaptations of Leukemic Stem Cells (Table 1)
A defining feature of leukemic stem cells is their unique metabolic profile. While rapidly proliferating leukemic blasts often rely on glycolysis, LSCs depend predominantly on mitochondrial oxidative phosphorylation for energy production [24–26]. This metabolic state has been associated with increased sensitivity to therapies targeting apoptotic priming, including BCL-2 inhibition. This metabolic program supports the quiescent state of LSCs and enhances their resistance to conventional chemotherapy.
Glutamine metabolism plays a particularly important role in maintaining mitochondrial function and redox homeostasis in AML cells. Recent studies have demonstrated that targeting glutamine metabolism can selectively impair leukemic stem cell survival while sparing normal hematopoietic stem cells [27–29]. In addition, alterations in lipid metabolism and fatty acid oxidation contribute to metabolic flexibility and allow LSCs to adapt to changing microenvironmental conditions within the bone marrow niche [30–32].
Importantly, these metabolic dependencies distinguish LSCs from normal hematopoietic stem cells and provide a therapeutic window for selective targeting.
These metabolic adaptations are closely interconnected with signals derived from the bone marrow microenvironment, highlighting the dynamic interplay between intrinsic cellular programs and extrinsic niche factors in sustaining leukemic stem cell survival.
|
Metabolic pathway |
Functional role in LSCs |
References |
|
Oxidative phosphorylation |
Primary energy source for quiescent LSCs |
[24–26] |
|
Glutamine metabolism |
Supports mitochondrial metabolism and redox balance |
[27,28] |
|
Lipid metabolism |
Provides metabolic plasticity and survival advantage |
[29,30] |
|
Fatty acid oxidation |
Adaptation to metabolic stress in the bone marrow niche |
[31,32] |
Leukemic Stem Cells and the Bone Marrow Microenvironment (Table 2)
The bone marrow microenvironment provides a specialized niche that supports leukemic stem cell survival and protects them from therapeutic stress. Interactions between LSCs and stromal cells, endothelial cells, and extracellular matrix components create a protective environment that promotes leukemia progression [33–36].
Additional mechanisms, including CXCR4–CXCL12 signaling, adhesion pathways such as VLA-4/VCAM-1, and stromal-derived soluble factors, further reinforce LSC retention and survival within the niche.
These niche interactions regulate key signaling pathways involved in stem cell maintenance, including Hedgehog, Wnt, and Notch signaling pathways. Activation of these pathways contributes to LSC self-renewal and therapeutic resistance [37–38]. Moreover, hypoxic conditions within the bone marrow niche further promote leukemic stem cell survival by inducing metabolic adaptations and altering transcriptional programs [39–40].
These interactions not only support LSC maintenance but also contribute to metabolic reprogramming and resistance to therapy.
|
Niche component |
Function in AML stem cell biology |
References |
|
Mesenchymal stromal cells |
Provide survival and anti-apoptotic signals |
[33,34] |
|
Endothelial cells |
Maintain vascular niche supporting LSCs |
[35] |
|
Osteoblastic niche |
Regulates stem cell quiescence |
[36] |
|
Hypoxic microenvironment |
Promotes metabolic adaptation and therapy resistance |
[39,40] |
Immunophenotypic Identification of Leukemic Stem Cells (Table 3)
Leukemic stem cells are commonly enriched within the CD34+CD38- compartment, although significant phenotypic heterogeneity exists among patients. Additional markers such as CD123, TIM-3, and CLL-1 have been identified that help distinguish LSCs from normal hematopoietic stem cells [11–13].
Advances in multiparametric flow cytometry and molecular diagnostics have enabled more precise detection and quantification of LSC populations in patient samples. These approaches are increasingly used for monitoring measurable residual disease and predicting relapse risk [37–40].
These markers are increasingly being explored as therapeutic targets and for measurable residual disease monitoring, with potential implications for clinical decision-making.
|
Marker |
Biological significance |
References |
|
CD34?CD38? |
Primitive leukemia-initiating phenotype |
[11,12] |
|
CD123 |
IL-3 receptor α chain enriched in AML LSCs |
[13] |
|
TIM-3 |
Immune checkpoint receptor associated with LSC function |
[14] |
|
CLL-1 |
AML-specific antigen under investigation for immunotherapy |
[15] |
Therapeutic Strategies Targeting Leukemic Stem Cells
Targeting leukemic stem cells is considered essential for achieving durable remission in AML. Several therapeutic approaches are currently under investigation, including metabolic inhibitors, epigenetic therapies, and immunotherapeutic strategies [16–18].
Recent advances include therapies targeting apoptotic pathways (e.g., BCL-2 inhibitors such as venetoclax), kinase-driven signaling (e.g., FLT3 inhibitors), metabolic vulnerabilities, and immunotherapeutic approaches targeting LSC-associated antigens.
Metabolic targeting has emerged as a promising strategy due to the dependence of LSCs on oxidative phosphorylation. Agents that disrupt mitochondrial metabolism have demonstrated the ability to selectively impair LSC survival in preclinical models [24–26].
In addition, immunotherapies targeting LSC-associated antigens are being actively explored. Monoclonal antibodies and cellular immunotherapies directed against CD123 and other LSC markers have shown encouraging activity in early clinical studies [37,38].
However, the emergence of resistance and the persistence of functionally diverse LSC populations remain major obstacles to durable disease control.
Conclusion
Leukemic stem cells represent a central barrier to durable remission in AML due to their capacity for self-renewal, metabolic adaptation, and protection within specialized bone marrow niches.
Emerging therapeutic strategies targeting metabolic dependencies, niche interactions, and survival pathways offer promising avenues for improving outcomes. However, LSC heterogeneity and adaptive resistance remain significant challenges.
Future efforts integrating multi-target approaches and insights from single-cell and multi-omics technologies will be critical to achieving durable disease eradication.
Acknowledgments
The author acknowledges the contributions of investigators whose foundational and translational work continue to advance understanding of acute myeloid leukemia stem cell biology.
Funding
This work was supported by the University of Maryland Marlene and Stewart Greenebaum Comprehensive Cancer Center American Cancer Society Institutional Research Grant (IRG-24-1290479-19, G.S.), the National Cancer Institute (NCI) Cancer Center Support Grant (CCSG) P30CA134274, and the State of Maryland Department of Health’s Cigarette Restitution Fund Program.
Conflicts of Interest
The author declares no conflicts of interest related to this work.
References
2. Döhner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022 Sep 22;140(12):1345–77.
3. Kantarjian HM, DiNardo CD, Kadia TM, Daver NG, Altman JK, Stein EM, et al. Acute myeloid leukemia management and research in 2025. CA Cancer J Clin. 2025 Jan-Feb;75(1):46–67.
4. Chen Y, Li J, Xu L, Găman MA, Zou Z. The genesis and evolution of acute myeloid leukemia stem cells in the microenvironment: From biology to therapeutic targeting. Cell Death Discov. 2022 Sep 26;8(1):397.
5. Urs AP, Goda C, Kulkarni R. Remodeling of the bone marrow microenvironment during acute myeloid leukemia progression. Ann Transl Med. 2024 Aug 1;12(4):63.
6. Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994 Feb 17;367(6464):645–8.
7. Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997 Jul;3(7):730–7.
8. Barreto IV, Pessoa FMCP, Machado CB, Pantoja LDC, Ribeiro RM, Lopes GS, et al. Leukemic Stem Cell: A Mini-Review on Clinical Perspectives. Front Oncol. 2022 Jun 24;12:931050.
9. Shin DY. Human acute myeloid leukemia stem cells: evolution of concept. Blood Res. 2022 Apr 30;57(S1):67–74.
10. Hansen Q, Bachas C, Smit L, Cloos J. Characteristics of leukemic stem cells in acute leukemia and potential targeted therapies for their specific eradication. Cancer Drug Resist. 2022 May 5;5(2):344–67.
11. Cozzio A, Passegué E, Ayton PM, Karsunky H, Cleary ML, Weissman IL. Similar MLL-associated leukemias arising from self-renewing stem cells and short-lived myeloid progenitors. Genes Dev. 2003 Dec 15;17(24):3029–35.
12. van der Werf I, Mondala PK, Steel SK, Balaian L, Ladel L, Mason CN, et al. Detection and targeting of splicing deregulation in pediatric acute myeloid leukemia stem cells. Cell Rep Med. 2023 Mar 21;4(3):100962.
13. Patel SB, Nemkov T, D'Alessandro A, Welner RS. Deciphering Metabolic Adaptability of Leukemic Stem Cells. Front Oncol. 2022 Jun 8;12:846149.
14. Rattigan KM, Zarou MM, Helgason GV. Metabolism in stem cell-driven leukemia: parallels between hematopoiesis and immunity. Blood. 2023 May 25;141(21):2553–65.
15. Jones CL, Inguva A, Jordan CT. Targeting Energy Metabolism in Cancer Stem Cells: Progress and Challenges in Leukemia and Solid Tumors. Cell Stem Cell. 2021 Mar 4;28(3):378–93.
16. Mishra SK, Millman SE, Zhang L. Metabolism in acute myeloid leukemia: mechanistic insights and therapeutic targets. Blood. 2023 Mar 9;141(10):1119–35.
17. Dembitz V, James SC, Gallipoli P. Targeting lipid metabolism in acute myeloid leukemia: biological insights and therapeutic opportunities. Leukemia. 2025 Aug;39(8):1814–23.
18. Nisticò C, Chiarella E. An Overview on Lipid Droplets Accumulation as Novel Target for Acute Myeloid Leukemia Therapy. Biomedicines. 2023 Nov 30;11(12):3186.
19. Bhattacharjee R, Ghosh S, Nath A, Basu A, Biswas O, Patil CR, et al. Theragnostic strategies harnessing the self-renewal pathways of stem-like cells in the acute myeloid leukemia. Crit Rev Oncol Hematol. 2022 Sep;177:103753.
20. Wang X, Huang S, Chen JL. Understanding of leukemic stem cells and their clinical implications. Mol Cancer. 2017 Jan 30;16(1):2.
21. Kollmann S, Grausenburger R, Klampfl T, Prchal-Murphy M, Bastl K, Pisa H, et al. A STAT5B-CD9 axis determines self-renewal in hematopoietic and leukemic stem cells. Blood. 2021 Dec 9;138(23):2347–59.
22. Joshi K, Zhang L, Breslin S J P, Zhang J. Leukemia Stem Cells in the Pathogenesis, Progression, and Treatment of Acute Myeloid Leukemia. Adv Exp Med Biol. 2019;1143:95–128.
23. Azizidoost S, Nasrolahi A, Sheykhi-Sabzehpoush M, Anbiyaiee A, Khoshnam SE, Farzaneh M, et al. Signaling pathways governing the behaviors of leukemia stem cells. Genes Dis. 2023 Mar 23;11(2):830–46.
24. Queiroz KC, Ruela-de-Sousa RR, Fuhler GM, Aberson HL, Ferreira CV, Peppelenbosch MP, et al. Hedgehog signaling maintains chemoresistance in myeloid leukemic cells. Oncogene. 2010 Dec 2;29(48):6314–22.
25. Kellaway SG, Potluri S, Keane P, Blair HJ, Ames L, Worker A, et al. Leukemic stem cells activate lineage inappropriate signalling pathways to promote their growth. Nat Commun. 2024 Feb 14;15(1):1359.
26. Bouligny IM, Maher KR, Grant S. Augmenting Venetoclax Activity Through Signal Transduction in AML. J Cell Signal. 2023;4(1):1–12.
27. Valent P, Sadovnik I, Eisenwort G, Bauer K, Herrmann H, Gleixner KV, et al. Immunotherapy-Based Targeting and Elimination of Leukemic Stem Cells in AML and CML. Int J Mol Sci. 2019 Aug 29;20(17):4233.
28. Dunn WG, McLoughlin MA, Vassiliou GS. Clonal hematopoiesis and hematological malignancy. J Clin Invest. 2024 Oct 1;134(19):e180065.
29. Choi HS, Kim BS, Yoon S, Oh SO, Lee D. Leukemic Stem Cells and Hematological Malignancies. Int J Mol Sci. 2024 Jun 17;25(12):6639.
30. Marchand T, Pinho S. Leukemic Stem Cells: From Leukemic Niche Biology to Treatment Opportunities. Front Immunol. 2021 Oct 15;12:775128.
31. Tasian SK, Bornhäuser M, Rutella S. Targeting Leukemia Stem Cells in the Bone Marrow Niche. Biomedicines. 2018 Feb 21;6(1):22.
32. Karantanou C, Godavarthy PS, Krause DS. Targeting the bone marrow microenvironment in acute leukemia. Leuk Lymphoma. 2018 Nov;59(11):2535–45.
33. Semedo C, Caroço R, Almeida A, Cardoso BA. Targeting the bone marrow niche, moving towards leukemia eradication. Frontiers in Hematology. 2024 Jul 22;3:1429916.
34. Soto CA, Lo Celso C, Purton LE, Frisch BJ. From the niche to malignant hematopoiesis and back: reciprocal interactions between leukemia and the bone marrow microenvironment. JBMR Plus. 2021 Jun 3;5(10):e10516.
35. Yao Y, Li F, Huang J, Jin J, Wang H. Leukemia stem cell-bone marrow microenvironment interplay in acute myeloid leukemia development. Exp Hematol Oncol. 2021 Jul 10;10(1):39.
36. Zeijlemaker W, Kelder A, Oussoren-Brockhoff YJ, Scholten WJ, Snel AN, Veldhuizen D, et al. A simple one-tube assay for immunophenotypical quantification of leukemic stem cells in acute myeloid leukemia. Leukemia. 2016 Feb;30(2):439–46.
37. Zeijlemaker W, Grob T, Meijer R, Hanekamp D, Kelder A, Carbaat-Ham JC, et al. CD34+CD38- leukemic stem cell frequency to predict outcome in acute myeloid leukemia. Leukemia. 2019 May;33(5):1102–12.
38. Das N, Panda D, Gajendra S, Gupta R, Thakral D, Kaur G, et al. Immunophenotypic characterization of leukemic stem cells in acute myeloid leukemia using single tube 10-colour panel by multiparametric flow cytometry: Deciphering the spectrum, complexity and immunophenotypic heterogeneity. Int J Lab Hematol. 2024 Aug;46(4):646–56.
39. Reuvekamp T, Janssen LLG, Ngai LL, Carbaat-Ham J, den Hartog D, Scholten WJ, et al. The role of the primitive marker CD133 in CD34-negative acute myeloid leukemia for the detection of leukemia stem cells. Cytometry B Clin Cytom. 2025 Jan;108(1):23–34.
40. Petersen MA, Bill M, Rosenberg CA. OMIP 072: A 15-color panel for immunophenotypic identification, quantification, and characterization of leukemic stem cells in children with acute myeloid leukemia. Cytometry A. 2021 Apr;99(4):382–7.