Abstract
Breast cancer remains a heterogeneous disease in which prognostic stratification, particularly within estrogen receptor (ER)-negative subtypes, remains clinically challenging. While genomic and transcriptomic profiling have advanced risk classification, protein-level regulation and subcellular localization are rarely incorporated into prognostic frameworks. NEDD8 ultimate buster 1 (NUB1) is a proteostasis-associated protein involved in degradation of ubiquitin-like modifiers and regulation of cell-cycle progression. Emerging evidence suggests that altered subcellular distribution of NUB1, rather than expression alone, associates with aggressive tumor behavior in ER-negative disease. In this editorial, we argue that NUB1 serves as a demonstrator protein illustrating how localization-dependent biology can refine prognostic stratification. Drawing on tissue-based outcome associations and paired primary–metastatic observations, we propose that loss of cytoplasmic NUB1 highlights a biologically distinct poor-outcome subset within ER-negative breast cancer. We further discuss validation priorities and practical considerations for incorporating localization-aware biomarkers into pathology workflows.
Keywords
Breast cancer, NUB1, Tumor, Triple-negative, Biomarker
Introduction: Prognostic Gaps in ER-Negative Breast Cancer
Despite advances in molecular oncology, ER-negative breast cancer continues to pose substantial prognostic and therapeutic challenges. Unlike ER-positive disease, which benefits from endocrine therapy and established biomarkers, ER-negative and triple-negative tumors remain biologically heterogeneous and clinically unpredictable. High-throughput genomic and transcriptomic approaches have yielded important insights; however, they incompletely capture post-transcriptional regulation, protein stability, and intracellular compartmentalization, all of which critically influence tumor behavior.
Proteostasis networks enable cells to adapt to stress and maintain protein homeostasis [1,2] and are frequently repurposed by tumors to support survival and therapy resistance [3,4]. Notably, not all proteostasis regulators function uniformly as tumor enablers. In selected contexts, loss or mislocalization of these proteins may instead mark biological vulnerability or aggressive disease states.
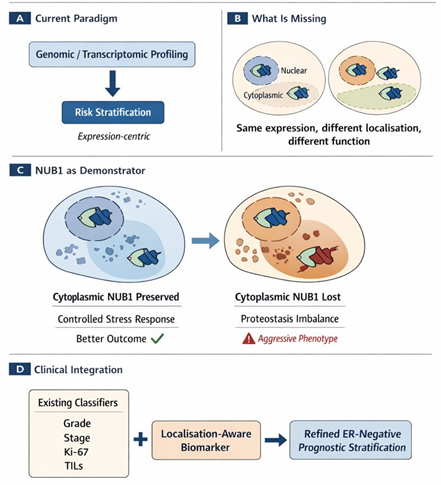
This editorial advance the broader proposition that subcellular localization represents an underutilized dimension of prognostic biomarker biology in ER-negative breast cancer. NUB1 is discussed not as an isolated candidate, but as a concrete example through which this principle can be examined.
Why Localization Matters More Than Expression
NUB1 is an interferon-inducible protein expressed as multiple splice variants, including NUB1L, which contains an additional ubiquitin-associated domain [5,6]. Functionally, NUB1 facilitates proteasomal degradation of NEDD8- and FAT10-conjugated proteins, thereby influencing proteostasis, stress responses, and cell-cycle control [7].
AIPL1, originally characterized in retinal biology, has been shown to modulate NUB1 stability and intracellular trafficking [8]. Subcellular localization determines substrate accessibility, interaction partners, and biological output. For proteins involved in degradation and stress signaling, compartmentalization may be particularly informative.
In oncology, reliance on transcript abundance as a surrogate for protein function is increasingly recognized as insufficient, as mRNA levels often fail to predict protein stability, localization, or activity. This disconnect is especially relevant in aggressive tumors where regulatory pathways are disrupted [9]. Accordingly, localization-aware assessment may capture prognostic information overlooked by expression-centric approaches.
Clinical Signals Supporting Localization-Dependent Prognostic Value
Analyses of public transcriptomic datasets suggest context-dependent associations between NUB1 expression and outcome, with adverse prognostic signals emerging primarily in ER-negative and triple-negative subsets. These findings imply biological specificity but are insufficient to define clinical utility [10,11].
Tissue-based immunohistochemical analyses extend these observations. Heterogeneous nuclear and cytoplasmic NUB1 patterns are observed that do not simply mirror transcript levels. Importantly, loss of cytoplasmic NUB1 identifies a subgroup of ER-negative tumors with significantly poorer outcomes, whereas nuclear localization alone lacks equivalent prognostic value.
These findings suggest that disruption of a cytoplasmic proteostasis-linked restraint mechanism may characterize a particularly aggressive ER-negative phenotype [11]. While the precise substrates and pathways remain to be elucidated, the localization–outcome association itself is clinically observable and reproducible within cohorts [10,11].
ER-Negative Specificity: Biological Plausibility
The apparent restriction of prognostic relevance to ER-negative disease warrants consideration. ER-negative tumors are characterized by heightened genomic instability, reliance on stress-adaptive pathways, and limited therapeutic options [7,12]. In this context, loss of cytoplasmic proteostasis regulators such as NUB1 may disproportionately impair cellular control mechanisms, conferring a selective growth advantage.
By contrast, ER-positive tumors benefit from hormone-driven regulatory networks that may buffer or override such effects. This distinction offers a plausible biological rationale for the ER-negative specificity of localization-dependent prognostic signals.
Integrating Case-Based and Population-Level Evidence
Localization-based biomarkers offer practical advantages. Immunohistochemistry is already embedded in diagnostic pathology, and compartment-specific scoring is routine for several established markers. Incorporating cytoplasmic NUB1 assessment would therefore require standardization rather than technological innovation.
However, clinical adoption must be contingent on comparative validation. Specifically, the prognostic signal attributed to cytoplasmic NUB1 should be tested against established ER-negative risk classifiers, including tumor grade, stage, proliferative indices, and immune markers, to determine whether localization adds independent or incremental prognostic value.
Critically, the potential value of cytoplasmic NUB1 does not lie in replacing established ER-negative risk classifiers, but in complementing them. Conventional prognostic variables such as tumor grade, proliferative indices, immune infiltration, and stage capture structural or microenvironmental features, whereas NUB1 localization may reflect intracellular stress handling and proteostasis capacity. We hypothesize that localization-aware biomarkers operate orthogonally to existing classifiers, identifying biologically aggressive tumors that are otherwise indistinguishable by standard pathology (Figure 1). Formal comparative modelling will be required to determine whether cytoplasmic NUB1 adds independent or incremental prognostic value; however, its signal suggests a distinct axis of tumor vulnerability that warrants systematic evaluation.
Future Directions
Next steps are clear and achievable, including independent validation of cytoplasmic NUB1 as a prognostic marker in larger ER-negative and triple-negative cohorts, standardization of immunohistochemical scoring thresholds suitable for routine pathology practice, and mechanistic interrogation of whether altered NUB1 localization modulates stress tolerance, proliferation, or treatment response. Importantly, these efforts should position NUB1 as a test case for localization-aware biomarker development rather than an endpoint.
Conclusion
Taken together, available evidence supports reframing NUB1 as a demonstrator protein illustrating the prognostic relevance of subcellular localization in ER-negative breast cancer. This perspective highlights localization as a neglected yet actionable dimension of tumor biology. If validated comparatively, localization-aware biomarkers may refine prognostic stratification and inform clinical decision-making in aggressive breast cancer subtypes where unmet need remains high.
References
2. Ramasamy A. Increasing statistical power and generalizability in genomics microarray research. Doctoral dissertation, University of Oxford; 2009.
3. Arshad M, Abdul Hamid N, Chan MC, Ismail F, Tan GC, Pezzella F, et al. NUB1 and FAT10 Proteins as Potential Novel Biomarkers in Cancer: A Translational Perspective. Cells. 2021 Aug 24;10(9):2176.
4. Hosono T, Tanaka T, Tanji K, Nakatani T, Kamitani T. NUB1, an interferon-inducible protein, mediates anti-proliferative actions and apoptosis in renal cell carcinoma cells through cell-cycle regulation. Br J Cancer. 2010 Mar 2;102(5):873–82.
5. Sohocki MM, Bowne SJ, Sullivan LS, Blackshaw S, Cepko CL, Payne AM, et al. Mutations in a new photoreceptor-pineal gene on 17p cause Leber congenital amaurosis. Nat Genet. 2000 Jan;24(1):79–83.
6. Kito K, Yeh ET, Kamitani T. NUB1, a NEDD8-interacting protein, is induced by interferon and down-regulates the NEDD8 expression. J Biol Chem. 2001 Jun 8;276(23):20603–9.
7. Zhang D, Wu P, Zhang Z, An W, Zhang C, Pan S, et al. Overexpression of negative regulator of ubiquitin-like proteins 1 (NUB1) inhibits proliferation and invasion of gastric cancer cells through upregulation of p27Kip1 and inhibition of epithelial-mesenchymal transition. Pathol Res Pract. 2020 Aug;216(8):153002.
8. van der Spuy J, Cheetham ME. The Leber congenital amaurosis protein AIPL1 modulates the nuclear translocation of NUB1 and suppresses inclusion formation by NUB1 fragments. J Biol Chem. 2004 Nov 12;279(46):48038–47.
9. Kamitani T, Kito K, Fukuda-Kamitani T, Yeh ET. Targeting of NEDD8 and its conjugates for proteasomal degradation by NUB1. J Biol Chem. 2001 Dec 7;276(49):46655-60.
10. Arshad M, Abdullah AR, Ismail F, Pezzella F, Yahaya A, Tan GC, et al. Mechanism and Predictive Role of NUB1 Protein in Oestrogen Receptor Pathway of FEC-Treated Breast Cancer Patients. Biomedicines. 2025 May 27;13(6):1307.
11. Tan KL, Haider S, Zois CE, Hu J, Turley H, Leek R, et al. Low cytoplasmic NUB1 protein exerts hypoxic cell death with poorer prognosis in oestrogen receptor negative breast cancer patients. Transl Oncol. 2024 Nov;49:102106.
12. Du D, Zhang W, Zhang D, Liu L, Li J, Chen Z, et al. NUB1 reduction promotes PCNA-mediated tumor growth by disturbing the PCNA polyubiquitination/NEDDylation in hepatocellular carcinoma cells. Cell Death Dis. 2025 Mar 31;16(1):228.