Abstract
Background: Hemodialysis (HD) patients experience severe physical deconditioning due to extremely low physical activity, prolonged sedentary behavior, and progressive muscle wasting. These changes accelerate frailty, impair functional independence, reduce quality of life (QoL), and increase hospitalization and mortality risks. Sarcopenia is now recognized as a key predictor of poor outcomes in this population.
Objective: To synthesize evidence on physical activity, sedentary behavior, muscle wasting, and exercise interventions in HD patients, and to evaluate the physiological and functional benefits of interdialytic exercise.
Methods: A structured narrative review was conducted using PubMed, Scopus, Web of Science, and Google Scholar for studies published from 2002–2025. Eligible studies included observational studies, randomized controlled trials, clinical trials, and systematic reviews involving adult HD patients. Data were extracted and thematically synthesized.
Results: HD patients exhibit extremely low physical activity and spend approximately 70–90% of their day sedentary, contributing to rapid muscle loss, frailty, reduced functional capacity, cognitive decline, and impaired QoL. Both intradialytic and interdialytic exercise programs significantly improve aerobic capacity, muscle strength, dialysis adequacy, inflammation markers, and patient-reported outcomes. Interdialytic training allows higher exercise intensity and volume, resulting in superior gains in VO2 max, mobility, and muscle performance. Home-based and supervised programs are feasible and safe, though adherence is limited by fatigue, depression, and access barriers.
Conclusion: Physical inactivity and sedentary lifestyles profoundly worsen musculoskeletal and functional health in HD patients. Structured exercise, particularly intradialytic and interdialytic training, provides meaningful physiological and clinical benefits. Exercise should be integrated as a core component of routine HD care to preserve function, enhance QoL, and improve long-term outcomes.
Keywords
Hemodialysis, Muscle wasting, Physical inactivity, Intradialytic
Introduction
Hemodialysis (HD) patients experience profound disturbances in physical functioning, largely driven by low physical activity levels, prolonged sedentary behavior, and progressive muscle wasting. These interrelated problems significantly accelerate frailty, reduce functional independence, impair quality of life, and increase the risk of hospitalization and mortality. Muscle atrophy in HD arises from a combination of catabolic inflammation, metabolic acidosis, hormonal disturbances, inadequate nutrition, and the inherent physiological stress of the dialysis process. Recent evidence also highlights the role of sarcopenia as a major predictor of poor outcomes across the chronic kidney disease (CKD) spectrum [1]. Globally, the prevalence of sarcopenia in CKD patients is rising, and HD individuals represent one of the highest-risk groups due to chronic inflammation, reduced mobility, and inadequate dietary protein intake. A recent systematic review demonstrated that sarcopenia affects a substantial proportion of dialysis patients and is strongly linked with impaired physical performance and mortality [2]. Physical inactivity further exacerbates these complications. Many HD patients spend more than half their day sitting or lying down, contributing to rapid functional decline and worsening cardiovascular health. Exercise interventions have emerged as a powerful therapeutic tool capable of counteracting these adverse effects. Evidence from randomized controlled trials shows that intradialytic exercise, performed during dialysis sessions, may improve survival, exercise capacity, and overall physical performance [3]. Mechanistic studies also reveal that regular exercise can modulate inflammation, enhance mitochondrial function, and improve muscle protein synthesis, highlighting the physiological pathways that support better health outcomes [4]. Beyond intradialytic exercise, home-based programs have gained attention for their feasibility and positive impact on physical function and activity levels in HD patients. A meta-analysis confirmed that structured home-based programs can effectively increase mobility and reduce functional decline, making them a valuable option for real-world dialysis care [5]. Moreover, repeated evidence demonstrates that exercise confers systemic physiological benefits through improved cardiovascular function, endothelial health, and enhanced metabolic flexibility, further reinforcing its role in CKD management [4]. Large multicenter investigations emphasize that integrating exercise into routine HD care is both safe and clinically beneficial. A landmark study published in NEJM Evidence demonstrated that exercise during HD sessions leads to significant improvements in physical function and may positively influence long-term outcomes [6]. Even simpler forms of movement, such as range of motion exercises performed twice weekly, have shown meaningful improvements in dialysis adequacy and quality of life in resource-limited settings, highlighting the broad applicability of exercise interventions [7].
Research Question
Primary research question
What is the relationship between physical activity levels, sedentary behavior, and muscle wasting in patients undergoing hemodialysis?
Secondary research question
- How prevalent are physical inactivity and sedentary lifestyles among hemodialysis patients?
- What mechanisms link sedentary behavior and low physical activity to muscle wasting (sarcopenia) in hemodialysis patients?
- How do physical activity levels influence muscle mass, strength, and functional capacity in this population?
- What types of exercise or physical activity interventions are effective in reducing muscle wasting in hemodialysis patients?
- What barriers and facilitators affect participation in physical activity among individuals on hemodialysis?
- How do changes in physical activity impact quality of life, morbidity, and mortality in hemodialysis patients?
Hypothesis regarding the review
Null hypothesis (H0)
There is no significant association between physical activity, sedentary behavior, and muscle wasting in patients undergoing hemodialysis.
Alternative hypothesis (H1)
Lower physical activity and higher sedentary behavior are significantly associated with greater muscle wasting and functional decline in patients undergoing hemodialysis.
Materials and Methods
Study design
This study followed a structured review design aimed at synthesizing evidence on physical activity, sedentary behavior, and related clinical outcomes in hemodialysis patients. A thematic synthesis approach was used to group studies based on major concepts such as physical activity levels, exercise interventions, muscle wasting, frailty, and quality of life. Only human studies involving adult hemodialysis patients were included.
Search strategy
A systematic search was conducted across major electronic databases, including PubMed, Scopus, Web of Science, and Google Scholar. Search terms included combinations of: “hemodialysis,” “physical activity,” “exercise,” “sedentary behavior,” “sarcopenia,” “functional capacity,” “frailty,” “quality of life,” and “CKD.” Boolean operators (AND/OR) were used to refine search results.
The search was limited to peer-reviewed English-language articles published between 2002 and 2025. Reference lists of included studies were also screened to identify additional relevant articles.
Eligibility criteria
Inclusion criteria
Peer-reviewed journal articles, Population: Adult CKD stage 5 / ESRD on MHD, Studies addressing physical activity, sedentary behavior, muscle mass, and muscle function.
Exclusion criteria
Pediatric CKD studies, Conference abstracts without full-text availability, Editorials or letters with no data.
Study selection
Titles and abstracts were screened independently. Full-text articles were reviewed and selected based on relevance to: physical activity or exercise in hemodialysis, sedentary time and health outcomes, muscle mass, sarcopenia, or frailty, exercise interventions (intradialytic or home-based), associations with hospitalization, QoL, inflammation, or mortality. Initially 119 articles were consulted. A final total of 32 articles were included after applying inclusion–exclusion criteria (Figure 1).
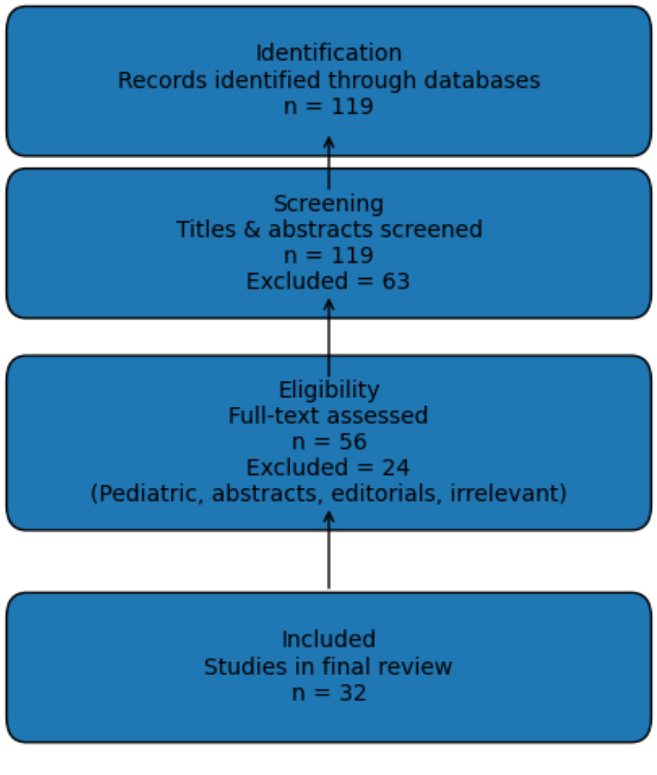
Figure 1: Flow-chart of the screened articles reviewed for the study
Results
Table 1 presents the detailed search strategy employed across the individual electronic databases, namely PubMed, Scopus, Web of Science, and Google Scholar. The table outlines the specific search criteria used for each database, including Boolean operators and keyword combinations. The primary keywords applied in the search process were Hemodialysis, Muscle Wasting, Physical Inactivity, and Intradialytic, ensuring comprehensive retrieval of relevant literature addressing the association between physical activity, sedentary behavior, and muscle health in hemodialysis patients.
|
S. No. |
Database |
Search criteria |
|
1. |
PubMed |
Core Concepts (hemodialysis and physical activity) |
|
2. |
Scopus |
TITLE-ABS-KEY fields with Boolean logic |
|
3. |
Web of Science |
The main field is TS = Topic, which searches Title + Abstract + Author Keywords + Keywords Plus. |
|
4. |
Google Scholar |
"Hemodialysis" and "physical activity" or exercise and "sedentary lifestyle" or sedentary were pasted in the Scholar search bar. |
Table 2 provides the bibliographic details of the studies included in the review. A total of 16 citations were incorporated after applying the predefined inclusion and exclusion criteria. For each selected study, the table reports the title of the article, names of the authors, journal of publication, year of publication, and the database from which the article was retrieved. This table offers a structured overview of the evidence base underpinning the review.
|
Citation number |
Title |
Authors |
Journal |
Publication year |
Database |
|
[3] |
The effect of intradialytic exercise on dialysis patient survival: a randomized controlled trial. |
Tabibi MA, Cheema B, Salimian N, Corrêa HL, Ahmadi S. |
BMC Nephrol. |
2023 |
doi: 10.1186/s12882-023-03158-6. PMID: 37069527; PMCID: PMC10108498.
|
|
[6] |
Exercise during Hemodialysis in Patients with Chronic Kidney Failure. |
Anding-Rost K, von Gersdorff G, von Korn P, Ihorst G, Josef A, Kaufmann M, Huber M, Bär T, Zeißler S, Höfling S, Breuer C, Gärtner N, Haykowsky MJ, Degenhardt S, Wanner C, Halle M. |
NEJM Evid. |
2023 |
doi: 10.1056/EVIDoa2300057. Epub 2023 Jun 17. PMID: 38320198.
|
|
[8] |
Improving dialysis adequacy and quality of life in patients undergoing hemodialysis with twice a week range of motion exercise. |
Rochmawati E, Utomo EK, Makiyah SNN. |
Ther Apher Dial. |
2022 |
doi: 10.1111/1744-9987.13701. Epub 2021 Jun 22. PMID: 34129271.
|
|
[10] |
Exercise training for adults undergoing maintenance dialysis. |
Bernier-Jean A, Beruni NA, Bondonno NP, Williams G, Teixeira-Pinto A, Craig JC, Wong G. |
Cochrane Database Syst Rev. |
2022 |
doi: 10.1002/14651858.CD014653. PMID: 35018639; PMCID: PMC8752366. |
|
[11] |
Physical Activity and Quality of Life in Hemodialysis Patients and Healthy Controls: A Cross-Sectional Study. |
Filipcic T, Bogataj Š, Pajek J, Pajek M. |
Int J Environ Res Public Health. |
2021 |
doi: 10.3390/ijerph18041978. PMID: 33670745; PMCID: PMC7922560.
|
|
[14] |
Exercise programme to improve quality of life for patients with end-stage kidney disease receiving haemodialysis: the PEDAL RCT. |
Greenwood SA, Koufaki P, Macdonald JH, Bulley C, Bhandari S, Burton JO, Dasgupta I, Farrington K, Ford I, Kalra PA, Kumwenda M, Macdougall IC, Messow CM, Mitra S, Reid C, Smith AC, Taal MW, Thomson PC, Wheeler DC, White C, Yaqoob M, Mercer TH. |
Health Technol Assess. |
2021 |
doi: 10.3310/hta25400. PMID: 34156335; PMCID: PMC8256322. |
|
[15] |
Exercise training during hemodialysis sessions: Physical and biochemical benefits. |
Torres E, Aragoncillo I, Moreno J, Vega A, Abad S, García-Prieto A, Macias N, Hernandez A, Godino MT, Luño J. |
Ther Apher Dial. |
2020 |
doi: 10.1111/1744-9987.13469. Epub 2020 Jan 27. PMID: 31886624.
|
|
[16] |
Exercise during hemodialysis and health promoting behaviors: a clinical trial. |
Dashtidehkordi A, Shahgholian N, Attari F. PMC6425622.
|
BMC Nephrol. |
2019 |
doi: 10.1186/s12882-019-1276-3. PMID: 30890122; PMCID. |
|
[17] |
Effects of exercise programs on physical function and activity levels in patients undergoing hemodialysis: a randomized controlled trial. |
Perez-Dominguez B, Casaña-Granell J, Garcia-Maset R, Garcia-Testal A, Melendez-Oliva E, Segura-Orti E. |
Eur J Phys Rehabil Med. |
2021 |
doi: 10.23736/S1973-9087.21.06694-6. Epub 2021 Apr 7. PMID: 33826277.
|
|
[24] |
Benefits and Barriers to and Desired Outcomes with Exercise in Patients with ESKD. |
Moorman D, Suri R, Hiremath S, Jegatheswaran J, Kumar T, Bugeja A, Zimmerman D. |
Clin J Am Soc Nephrol. |
2019 |
doi: 10.2215/CJN.09700818. Epub 2019 Jan 29. PMID: 30696660; PMCID: PMC6390914.
|
|
[26] |
Dialysis exercise team: the way to sustain exercise programs in hemodialysis patients. |
Capitanini A, Lange S, D'Alessandro C, Salotti E, Tavolaro A, Baronti ME, Giannese D, Cupisti A. |
Kidney Blood Press Res. |
2014 |
doi: 10.1159/000355787. Epub 2014 Jul 29. PMID: 25117740. |
|
[27] |
Comparative associations of muscle mass and muscle strength with mortality in dialysis patients. |
Isoyama N, Qureshi AR, Avesani CM, Lindholm B, Bàràny P, Heimbürger O, Cederholm T, Stenvinkel P, Carrero JJ. |
Clin J Am Soc Nephrol. |
2014 |
doi: 10.2215/CJN.10261013. Epub 2014 Jul 29. PMID: 25074839; PMCID: PMC4186520. |
|
[28] |
Associations of self-reported physical activity types and levels with quality of life, depression symptoms, and mortality in hemodialysis patients: the DOPPS. |
Lopes AA, Lantz B, Morgenstern H, Wang M, Bieber BA, Gillespie BW, Li Y, Painter P, Jacobson SH, Rayner HC, Mapes DL, Vanholder RC, Hasegawa T, Robinson BM, Pisoni RL. |
Clin J Am Soc Nephrol. |
2014 |
doi: 10.2215/CJN.12371213. Epub 2014 Oct 2. PMID: 25278548; PMCID: PMC4186524.
|
|
[29] |
Exercise training for adults with chronic kidney disease. |
Heiwe S, Jacobson SH. |
Cochrane Database Syst Rev. |
2011 |
doi: 10.1002/14651858.CD003236.pub2. PMID: 21975737; PMCID: PMC10183198. |
|
[30] |
Physical activity and mortality in chronic kidney disease (NHANES III). |
Beddhu S, Baird BC, Zitterkoph J, Neilson J, Greene T. PMC2798872. |
Clin J Am Soc Nephrol. |
2009 |
doi: 10.2215/CJN.01970309. Epub 2009 Oct 9. PMID: 19820134; PMCID. |
|
[31] |
Progressive exercise for anabolism in kidney disease (PEAK): a randomized, controlled trial of resistance training during hemodialysis. |
Cheema B, Abas H, Smith B, O'Sullivan A, Chan M, Patwardhan A, Kelly J, Gillin A, Pang G, Lloyd B, Singh MF. |
J Am Soc Nephrol. |
2007 |
doi: 10.1681/ASN.2006121329. Epub 2007 Apr 4. PMID: 17409306.
|
Table 3 summarizes the key characteristics and findings of the included studies focusing on physical activity and muscle wasting in hemodialysis patients. The 16 selected citations are presented with essential methodological details, including sample size, study design (e.g., observational study, randomized controlled trial, systematic review), intervention or primary variables examined, and the principal outcomes or findings. This table enables comparison across studies and highlights patterns in evidence regarding the impact of physical inactivity and exercise interventions on muscle health and functional capacity.
|
Citation number |
Sample Size |
Study Design |
Intervention / Variable |
Key Findings |
|
[3] |
74 HD patients |
RCT |
Intradialytic exercise |
Improved survival and physical function. |
|
[6] |
235 HD Patients |
RCT |
Exercise during HD |
Improved strength, BP, and dialysis tolerance |
|
[7] |
60 HD Patients |
Clinical trial |
ROM exercise (2×/week) |
Better Kt/V and QoL. |
|
[10] |
Multiple RCTs |
Cochrane review |
Exercise training |
Improves walking capacity, QoL, strength. |
|
[11] |
79 HD Patients |
Cross-sectional |
Physical activity levels |
HD patients have lower physical activity and QoL than controls. |
|
[14] |
335 HD Patients |
RCT (PEDAL) |
QoL-focused exercise |
Modest improvement in physical function. |
|
[15] |
44 HD Patients |
Clinical trial |
Intradialytic cycling |
Improved strength, albumin, reduced inflammation. |
|
[16] |
70 HD Patients |
Clinical trial |
HD exercise + health behaviors |
Improved self-care behaviors & QoL. |
|
[17] |
88 HD Patients |
RCT |
Combined exercise program |
Increased activity levels & physical performance. |
|
[24] |
151 HD Patients |
Survey study |
Barriers to exercise |
Fatigue & lack of motivation common barriers. |
|
[26] |
N/A Review Article |
Observational |
Exercise team model |
Dedicated staff increases program adherence. |
|
[27] |
330 HD Patients |
Cohort |
Muscle mass vs strength |
Strength predicts mortality more strongly. |
|
[28] |
5763 HD Patients |
Cohort (DOPPS) |
Self-reported physical activity |
Higher physical activity linked to lower mortality & depression. |
|
[29] |
Multiple RCTs |
Cochrane review |
Exercise training |
Improves fitness, strength, and QoL in CKD/HD. |
|
[30] |
5800+HD Patients |
NHANES cohort |
Physical activity |
Higher physical activity reduces mortality in CKD. |
|
[31 |
49 HD Patients |
RCT |
Resistance training during HD |
Marked increase in muscle strength & mass. |
Table 4 presents a variable-wise conceptual synthesis of dialysis-related physical activity research. All 16 included studies are mapped according to the central concept examined (e.g., sarcopenia, sedentary behavior, intradialytic exercise), along with operational definitions, measurement tools employed (such as BIA, DXA, handgrip strength, 6MWT, accelerometry), summary of evidence, and their clinical relevance. This table integrates theoretical constructs with empirical findings and underscores the clinical importance of assessing and addressing physical inactivity and muscle wasting in the hemodialysis population.
|
Citation number |
Concept |
Definition |
Measurement Tools |
Evidence summary & clinical importance |
|
[7,10,11,28,30] |
Physical activity |
Any bodily movement that increases energy expenditure above resting level. |
IPAQ, Actigraphy, step count, physical activity logs |
HD patients show extremely low activity levels. Higher activity improves survival, functional capacity, QoL, and reduces hospitalization. |
|
[24] |
Sedentary behavior |
Waking behavior with energy expenditure ≤1.5 METs while sitting or reclining. |
Actigraphy, posture monitors, sedentary time tracking |
HD patients spend 70–90% of their day sedentarily. High sedentary time is linked to mortality, cardiovascular events, and frailty. |
|
[27] |
Muscle wasting / Sarcopenia |
Loss of muscle mass, strength, and function common in CKD/HD. |
BIA, DXA, handgrip strength, SPPB |
Highly prevalent in HD and predicts disability, hospitalization, falls, poor QoL, and mortality. |
|
[3,6,15–17,26,29,31] |
Exercise Intervention (intradialytic / home-based) |
Planned, structured physical activity performed during dialysis or at home. |
Exercise Logs, RPM, cycle ergometer output |
Improves aerobic capacity, strength, dialysis adequacy, BP control, and reduces depressive symptoms. Recommended as part of routine HD care. |
|
[14] |
Quality of Life (QoL) |
Patient’s perception of physical, mental, and social well-being. |
KDQOL-36, SF-36, WHOQoL |
Strongly associated with physical activity level. Exercise programs significantly improve QoL domains in HD patients. |
Effects of sedentary lifestyle
A sedentary lifestyle is highly prevalent among hemodialysis patients and contributes to rapid physical and cognitive decline. Long sitting hours, limited mobility during dialysis, and chronic fatigue accelerate muscle wasting, impair functional capacity, and worsen mental health. Sedentary behavior is also associated with poorer cognitive performance, as inactivity may reduce neuroplasticity and slow cerebral blood flow. Evidence shows that reduced engagement in physical or cognitive activities results in lower attention, memory, and executive function, all of which are already vulnerable in chronic kidney disease. [8,9].
Effects of physical activity
Physical activity provides multiple therapeutic benefits for hemodialysis patients by improving cardiovascular fitness, muscle mass, strength, and overall quality of life. Regular aerobic or resistance exercise enhances physical function, reduces inflammation, and counters sarcopenia by promoting protein synthesis and muscle preservation. Exercise training is also linked to better cognitive outcomes, increased independence in daily activities, and improved emotional well-being. Systematic reviews and controlled trials consistently demonstrate that structured exercise programs—including intradialytic and home-based training—can significantly improve physical performance and life quality in this population [10,11].
Interdialytic exercise
Interdialytic exercise refers to physical activity performed between hemodialysis sessions, as opposed to intradialytic exercise that takes place during treatment. This period represents a crucial window for rehabilitation because patients are physiologically more stable, have fewer movement restrictions, and can engage in a broader range of aerobic, resistance, and functional training activities. Interdialytic exercise is a core pillar of renal rehabilitation, integrating structured physical activity with nutrition and lifestyle modifications to counteract the severe deconditioning commonly observed in dialysis patients [12].
Rationale for interdialytic exercise
Patients on maintenance hemodialysis experience profound declines in muscle mass, cardiorespiratory fitness, and physical function. These impairments are driven by uremic inflammation, dietary inadequacy, metabolic acidosis, protein-energy wasting, and prolonged sedentary behavior between dialysis sessions. Interdialytic exercise provides a way to target these deficits using training intensities that are impossible to achieve during the constrained environment of hemodialysis. Evidence indicates that structured exercise during the non-dialysis days improves physiological reserve, increases daily energy expenditure, and promotes long-term behavioral change toward active living [13].
Types of interdialytic exercise
A wide range of exercise modalities can be implemented safely in the interdialytic period. Aerobic training commonly includes walking, cycling, swimming, or home-based step exercises. Resistance training may involve elastic bands, dumbbells, bodyweight exercises, or machine-based strengthening programs. Functional exercises, such as balance work, mobility drills, and stair climbing, are also recommended to enhance independence in daily activities. Clinical practice guidelines emphasize combining aerobic and resistance components for maximal improvement in muscle and cardiorespiratory outcomes [12].
Evidence from clinical trials
The PEDAL randomized controlled trial demonstrated that a structured 6-month exercise program delivered outside dialysis sessions resulted in improvements in physical function, mental well-being, vitality, and patient-reported quality of life. Although individual results varied, the overall trend indicated that supervised interdialytic exercise programs are feasible and beneficial for people receiving hemodialysis. Importantly, the trial highlighted the need for personalized training loads and gradual progression to maximize adherence and safety [14]. Similarly, other studies have shown that exercise training, whether performed intra- or interdialytically, leads to substantial improvements in muscle strength, functional mobility, walking capacity, and biochemical markers of inflammation and metabolism. Interdialytic training, in particular, allows higher intensities and larger training volumes, contributing to greater gains in VO2 max, lower limb strength, and overall physical performance [15].
Physiological and functional benefits
Interdialytic exercise contributes to multiple clinically relevant outcomes. Regular participation enhances mitochondrial function, promotes muscle protein synthesis, reduces oxidative stress, and improves endothelial health. These physiological adaptations translate to measurable functional benefits, including improved gait speed, chair-stand performance, and balance, key predictors of survival and hospitalization in dialysis patients. Exercise also improves fatigue resistance, sleep quality, and mobility, allowing patients to maintain higher levels of independence and social participation [13].
Program design and safety considerations
Renal rehabilitation experts recommend starting interdialytic exercise programs with low-to-moderate intensities and gradually increasing frequency and duration over several weeks. Pre-exercise assessment should include evaluation of cardiovascular risk, musculoskeletal status, and dialysis-related complications. Patients with intradialytic hypotension, severe anemia, uncontrolled hypertension, or acute illness may require temporary modification or postponement of exercise plans. Continuous communication between nephrologists, physical therapists, and dialysis staff ensures safe progression and adherence [12].
Adherence and implementation challenges
Despite the clear benefits, adherence to interdialytic exercise programs remains a challenge. Fatigue, depression, lack of motivation, transportation issues, and limited access to supervised exercise facilities contribute to poor long-term engagement. The PEDAL trial emphasized that personalized, flexible, and motivationally tailored programs significantly improve adherence. Home-based models supported by telehealth, wearable devices, and remote monitoring are emerging as effective solutions to overcome these barriers [14].
Key findings
This information was organized into structured tables summarizing: Key studies on physical activity and muscle wasting in hemodialysis patients (Table 1). Key studies variable-wise summary of concepts in dialysis-related physical activity research (Table 2).
Data synthesis
A thematic synthesis approach was used. Studies were grouped into themes such as “sedentary behavior,” “physical activity benefits,” “muscle wasting,” “exercise interventions,” and “QoL outcomes.” Findings were compared, contrasted, and summarized. Trends, gaps, and future directions were identified. No statistical meta-analysis was performed because the review aimed to narratively summarize findings due to heterogeneity between study methods.
Discussion
Hemodialysis (HD) patients represent a population with profound impairments in physical activity, muscle health, and overall functional capacity. The evidence summarized in the present review demonstrates that reduced physical activity and prolonged sedentary behavior are not merely lifestyle characteristics in this group, but central pathological contributors to muscle wasting, frailty, poor quality of life (QoL), and increased mortality [2,27,28,30]. The interaction between inactivity, sarcopenia, and adverse outcomes forms a self-perpetuating cycle that significantly compromises long-term prognosis in individuals undergoing maintenance HD.
Physical activity levels in hemodialysis patients: Across observational and cohort studies, HD patients consistently exhibit extremely low levels of physical activity compared with healthy controls [11,28]. Objective monitoring confirms that patients spend the majority of their waking hours sedentary [24]. Large cohorts such as DOPPS and NHANES demonstrate a strong inverse relationship between physical activity and mortality, depression, and hospitalization [28,30]. These findings indicate that physical activity is not only a marker of health but an independent determinant of survival in HD patients.
Lower activity levels are also associated with impaired functional capacity, as assessed by the six-minute walk test (6MWT), sit-to-stand, and TUG tests [10,14]. This decline in physical function directly limits independence and social participation, reinforcing the importance of routine physical activity assessment in HD care [13].
Sedentary behavior and its clinical implications: HD patients are characterized by excessive sedentary time, often exceeding 70–90% of the day [24]. Sedentary behavior has emerged as an independent risk factor for adverse outcomes, distinct from lack of exercise. Prolonged sitting during dialysis and non-dialysis hours contributes to metabolic dysregulation, cardiovascular risk, and further muscle deconditioning [20,21].
High sedentary burden is strongly associated with frailty, cardiovascular events, and mortality [22,27]. Given that dialysis sessions impose unavoidable immobility, strategies that incorporate light movement and intradialytic exercise are essential to counteract sedentary physiology [3,7,15,25].
Muscle wasting and sarcopenia in hemodialysis: Muscle wasting and sarcopenia are highly prevalent in HD patients and represent a core pathological process linking inactivity to poor outcomes [2,9]. Loss of muscle mass and strength arises from uremic toxicity, inflammation, protein-energy wasting (PEW), metabolic acidosis, and physical inactivity [18]. Notably, muscle strength predicts mortality more strongly than muscle mass alone [27], highlighting the clinical relevance of neuromuscular function.
Assessments using BIA, DXA, handgrip strength, and SPPB consistently identify high rates of sarcopenia [11,31]. Sarcopenia is associated with increased falls, disability, hospitalization, and reduced QoL [2,9,27].
Role of exercise interventions: Strong evidence from RCTs and systematic reviews supports the safety and efficacy of structured exercise interventions in HD patients [5,10,21,25,29]. Intradialytic cycling, resistance training, and range-of-motion exercises significantly improve muscle strength, aerobic capacity, dialysis adequacy (Kt/V), and blood pressure control [3,6,7,15,31].
Combined exercise and nutritional strategies provide superior improvements in muscle mass and function compared with either alone [1,18]. Exercise also yields psychological benefits, including reductions in depressive symptoms and improvements in cognitive performance [8,22], reinforcing its role as a multidimensional therapeutic intervention.
Functional capacity and quality of life: Functional capacity is a strong predictor of hospitalization and survival in HD patients [10,14]. Poor function limits independence and diminishes QoL. Evidence from Cochrane reviews and large RCTs such as the PEDAL trial shows that exercise leads to meaningful improvements in walking ability and perceived health status [10,14,29].
QoL is strongly associated with physical activity levels [11,28]. Patients engaging in regular exercise report better physical, mental, and social well-being [7,16,25], underlining the holistic benefits of addressing inactivity and muscle wasting.
Frailty, sleep and cognitive health: Frailty is highly prevalent in HD patients and reflects cumulative deficits in strength and physiological reserve [20]. Physical inactivity and sarcopenia are major contributors to frailty progression [27]. Importantly, even frail patients benefit from tailored exercise programs [20,32].
Sleep quality is frequently impaired in HD patients. Exercise interventions improve sleep duration and quality [19], which may further support recovery and muscle health. Additionally, exercise preserves cognitive function and may protect against dialysis-related cognitive decline [8,22].
Barriers and implementation challenges: Despite strong evidence, exercise is not routinely embedded in HD care. Common barriers include fatigue, lack of motivation, and fear of injury [24]. Studies indicate that dedicated multidisciplinary exercise teams and staff engagement significantly improve adherence [26], emphasizing the need for system-level integration of renal rehabilitation.
Clinical and Research Implications: Overall, physical inactivity, sedentary behavior, and muscle wasting are central, modifiable determinants of outcomes in HD patients. Routine screening for physical activity, sarcopenia, and functional capacity should be standard practice [13,19]. Individualized, progressive exercise programs combined with nutritional support should be prescribed as part of comprehensive renal rehabilitation [1,18,32].
Future research should focus on optimizing exercise dose, minimizing sedentary time, and developing scalable models for implementing intradialytic and home-based programs across diverse dialysis settings.
Limitations
- Predominance of observational data- Many associations between physical activity, sedentary behavior, sarcopenia, and outcomes are derived from cohort and cross-sectional studies, which limit causal inference. Large cohort studies such as Dialysis Outcomes and Practice Patterns Study analyses by Lopes AA et al. (2014) [28] and the National Health and Nutrition Examination Survey study by Beddhu S et al. (2009) [30] demonstrated strong associations between physical activity and mortality, while acknowledging that causality cannot be inferred. More recent evidence continues to recognize this limitation. The 2024 global meta-analysis by Duarte MP et al. (2024) [2] notes substantial heterogeneity and observational dominance in sarcopenia research. Similarly, the 2025 network meta-analysis by Zhang L et al. (2025) [1] emphasizes that many underlying studies are non-interventional, limiting causal conclusions.
- Heterogeneity of measures- Physical activity, sedentary lifestyle, and sarcopenia were assessed using different tools (self-report vs. accelerometry; BIA vs. DXA; various strength tests), reducing comparability across studies. Substantial variability exists in assessment methods for physical activity, sedentary behavior, and sarcopenia. Earlier reviews such as Sabatino A et al. (2021) [9] and Bakker EA et al. (2020) [13] highlighted inconsistencies in diagnostic criteria (BIA vs DXA), strength assessments, and functional tests. Post-2022 literature continues to emphasize this issue. Duarte et al. (2024) [2] explicitly report wide methodological heterogeneity affecting pooled sarcopenia prevalence estimates. Likewise, Bernier-Jean et al. (2022) [10] in the updated Cochrane review underline inconsistent outcome measures across RCTs, limiting comparability and meta-analytic synthesis.
- Small and short RCTs- Most exercise trials in HD patients have modest sample sizes and relatively short intervention periods, limiting conclusions about long-term sustainability and survival effects. Exercise RCTs in HD patients are frequently limited by modest sample sizes and short intervention periods. Early RCTs such as the PEAK trial by Cheema B et al. (2007) [31] and the Cochrane review by Heiwe S & Jacobson (2011) [29] acknowledged limited power for long-term outcomes.
More recent RCT syntheses continue to recognize this constraint. The 2022 Cochrane review by Bernier-Jean et al. (2022) [10] and the 2023 home-based exercise meta-analysis by Battaglia Y et al. (2023) [5] both emphasize short follow-up durations and insufficient power to determine effects on mortality and cardiovascular endpoints. Even contemporary trials such as Anding-Rost K et al. (2023) [6] acknowledge the need for longer-term outcome evaluation. - Selection bias- Participants in exercise studies are often healthier and more motivated than the general HD population, which may overestimate feasibility and benefits. Exercise trial participants often represent healthier, more motivated subsets of the HD population. Foundational discussions by Kouidi E (2002) [32] and Wang CJ & Johansen (2019) [20] questioned whether frail dialysis patients are adequately represented in exercise research.
Recent systematic reviews reinforce this concern. Bernier-Jean et al. (2022) [10] and Battaglia et al. (2023) [5] report selective recruitment of relatively stable patients, potentially overestimating feasibility and benefit. - Limited data on dose–response- The optimal type, intensity, frequency, and duration of exercise for different HD subgroups remain unclear. The optimal type, frequency, intensity, and duration of exercise remain unclear. Earlier meta-analyses such as Sheng K et al. (2014) [25] and Lu Y et al. (2019) [23] identified wide variability in exercise prescriptions and insufficient dose–response evaluation.
More recent syntheses, including Bernier-Jean et al. (2022) [10] and Zhang et al. (2025) [1], reiterate that heterogeneity in intervention protocols precludes firm recommendations regarding optimal dosage, particularly across HD subgroups. - Implementation gaps- Few studies evaluate real-world scalability, cost-effectiveness, and integration of exercise programs into routine dialysis workflows. Despite robust efficacy data, exercise remains poorly integrated into routine dialysis workflows. Early discussions by Capitanini A et al. (2014) [26] emphasized the need for structured dialysis exercise teams. Moorman D et al. (2019) [24] documented patient-reported barriers including fatigue and lack of support. More recent reviews, including Lambert et al. (2022) [19] and Battaglia et al. (2023) [5], highlight persistent gaps in real-world scalability, sustainability, and cost-effectiveness.
- Underrepresentation of frail and comorbid patients- The most vulnerable HD patients are often excluded from trials, limiting generalizability. Frailty and multimorbidity are highly prevalent in HD populations, yet vulnerable patients are frequently excluded from trials. Foundational analyses by Isoyama N et al. (2014) [27] and Wang & Johansen (2019) [20] emphasized the prognostic impact of muscle weakness and frailty, while noting limited representation in interventional research. Recent reviews such as Duarte et al. (2024) [2] and Zhang et al. (2025) [1] continue to report wide variability in risk profiles and limited inclusion of severely frail individuals, thereby restricting generalizability.
Conclusion
This review was guided by the following hypotheses:
Null Hypothesis (H0): There is no significant association between physical activity, sedentary behavior, and muscle wasting in patients undergoing hemodialysis.
Alternative Hypothesis (H1): Lower physical activity and higher sedentary behavior are significantly associated with greater muscle wasting and functional decline in patients undergoing hemodialysis.
Based on the accumulated evidence synthesized in this review, the null hypothesis (H0) is rejected, and the alternative hypothesis (H1) is supported.
Consistent findings from large observational cohorts such as the Dialysis Outcomes and Practice Patterns Study and analyses of National Health and Nutrition Examination Survey data demonstrate strong inverse associations between physical activity levels and mortality, hospitalization, depressive symptoms, and reduced quality of life. These studies show that lower physical activity is not merely a correlate of illness severity but an independent determinant of adverse outcomes. Furthermore, comparative analyses of muscle mass and strength in dialysis populations, such as those by Isoyama N and colleagues, demonstrate that reduced muscle strength is a powerful predictor of mortality, reinforcing the clinical relevance of functional decline.
The review also highlights that HD patients spend a substantial proportion of their waking hours sedentary, often exceeding 70–90%, and that this sedentary burden is independently associated with frailty progression, cardiovascular risk, and diminished functional performance. The convergence of evidence linking inactivity, sarcopenia, and poor outcomes establishes a self-perpetuating cycle that significantly compromises long-term prognosis.
Importantly, randomized controlled trials—including early resistance training studies such as the PEAK trial led by Cheema B and more recent trials such as the PEDAL RCT conducted by Greenwood SA—demonstrate that structured exercise interventions significantly improve muscle strength, aerobic capacity, walking performance, and perceived health status. Systematic reviews and meta-analyses further confirm improvements in dialysis adequacy (Kt/V), blood pressure regulation, and psychological well-being. These interventional findings provide mechanistic and clinical corroboration of the observed associations, supporting a causal pathway between increased physical activity and improved muscle and functional outcomes.
Therefore, the collective evidence supports the alternative hypothesis that lower physical activity and higher sedentary behavior are significantly associated with greater muscle wasting and functional decline in patients undergoing maintenance hemodialysis.
From a clinical standpoint, intradialytic and home-based exercise interventions represent effective therapeutic strategies capable of interrupting this adverse cycle. Integrating physical education professionals or exercise physiologists into multidisciplinary dialysis teams enables individualized prescription, safe supervision, and sustained patient engagement. Such a structured rehabilitation model has the potential to reduce functional decline, enhance treatment satisfaction, and improve long-term survival and quality of life in this vulnerable population.
Declaration by Authors
The authors hereby declared that this research is original and had not been sent to any other journal for publication.
Acknowledgement
The authors thank to the University Authority for providing all logistic support in the study.
Funding Agencies
None.
Conflict of Interest
The authors declared no conflict of interest.
References
2. Duarte MP, Almeida LS, Neri SGR, Oliveira JS, Wilkinson TJ, Ribeiro HS, et al. Prevalence of sarcopenia in patients with chronic kidney disease: a global systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2024 Apr;15(2):501–12.
3. Tabibi MA, Cheema B, Salimian N, Corrêa HL, Ahmadi S. The effect of intradialytic exercise on dialysis patient survival: a randomized controlled trial. BMC Nephrol. 2023 Apr 17;24(1):100.
4. Bishop NC, Burton JO, Graham-Brown MPM, Stensel DJ, Viana JL, Watson EL. Exercise and chronic kidney disease: potential mechanisms underlying the physiological benefits. Nat Rev Nephrol. 2023 Apr;19(4):244–56.
5. Battaglia Y, Amicone M, Mantovani A, Combe C, Mitra S, Basile C, et al. Home-based exercise in patients on maintenance dialysis: a systematic review and meta-analysis of randomized clinical trials. Nephrol Dial Transplant. 2023 Oct 31;38(11):2550–61.
6. Anding-Rost K, von Gersdorff G, von Korn P, Ihorst G, Josef A, Kaufmann M, et al. Exercise during Hemodialysis in Patients with Chronic Kidney Failure. NEJM Evid. 2023 Sep;2(9):EVIDoa2300057.
7. Rochmawati E, Utomo EK, Makiyah SNN. Improving dialysis adequacy and quality of life in patients undergoing hemodialysis with twice a week range of motion exercise. Ther Apher Dial. 2022 Feb;26(1):140–6.
8. Bogataj Š, Mesarič KK, Pajek M, Petrušič T, Pajek J. Physical exercise and cognitive training interventions to improve cognition in hemodialysis patients: A systematic review. Front Public Health. 2022 Oct 14;10:1032076.
9. Sabatino A, Cuppari L, Stenvinkel P, Lindholm B, Avesani CM. Sarcopenia in chronic kidney disease: what have we learned so far? J Nephrol. 2021 Aug;34(4):1347–72.
10. Bernier-Jean A, Beruni NA, Bondonno NP, Williams G, Teixeira-Pinto A, Craig JC, et al. Exercise training for adults undergoing maintenance dialysis. Cochrane Database Syst Rev. 2022 Jan 12;1(1):CD014653.
11. Filipčič T, Bogataj Š, Pajek J, Pajek M. Physical Activity and Quality of Life in Hemodialysis Patients and Healthy Controls: A Cross-Sectional Study. Int J Environ Res Public Health. 2021 Feb 18;18(4):1978.
12. Hoshino J. Renal Rehabilitation: Exercise Intervention and Nutritional Support in Dialysis Patients. Nutrients. 2021 Apr 24;13(5):1444.
13. Bakker EA, Zoccali C, Dekker FW, Eijsvogels TMH, Jager KJ. Assessing physical activity and function in patients with chronic kidney disease: a narrative review. Clin Kidney J. 2020 Sep 8;14(3):768–79.
14. Greenwood SA, Koufaki P, Macdonald JH, Bulley C, Bhandari S, Burton JO, et al. Exercise programme to improve quality of life for patients with end-stage kidney disease receiving haemodialysis: the PEDAL RCT. Health Technol Assess. 2021 Jun;25(40):1–52.
15. Torres E, Aragoncillo I, Moreno J, Vega A, Abad S, García-Prieto A, et al. Exercise training during hemodialysis sessions: Physical and biochemical benefits. Ther Apher Dial. 2020 Dec;24(6):648–54.
16. Dashtidehkordi A, Shahgholian N, Attari F. "Exercise during hemodialysis and health promoting behaviors: a clinical trial". BMC Nephrol. 2019 Mar 19;20(1):96.
17. Perez-Dominguez B, Casaña-Granell J, Garcia-Maset R, Garcia-Testal A, Melendez-Oliva E, Segura-Orti E. Effects of exercise programs on physical function and activity levels in patients undergoing hemodialysis: a randomized controlled trial. Eur J Phys Rehabil Med. 2021 Dec;57(6):994–1001.
18. Hanna RM, Ghobry L, Wassef O, Rhee CM, Kalantar-Zadeh K. A Practical Approach to Nutrition, Protein-Energy Wasting, Sarcopenia, and Cachexia in Patients with Chronic Kidney Disease. Blood Purif. 2020;49(1-2):202–11.
19. Lambert K, Lightfoot CJ, Jegatheesan DK, Gabrys I, Bennett PN. Physical activity and exercise recommendations for people receiving dialysis: A scoping review. PLoS One. 2022 Apr 28;17(4):e0267290.
20. Wang CJ, Johansen KL. Are dialysis patients too frail to exercise? Semin Dial. 2019 Jul;32(4):291–6.
21. Bogataj Š, Pajek M, Pajek J, Buturović Ponikvar J, Paravlic A. Exercise-Based Interventions in Hemodialysis Patients: A Systematic Review with a Meta-Analysis of Randomized Controlled Trials. J Clin Med. 2019 Dec 24;9(1):43.
22. Chu NM, McAdams-DeMarco MA. Exercise and cognitive function in patients with end-stage kidney disease. Semin Dial. 2019 Jul;32(4):283–90.
23. Lu Y, Wang Y, Lu Q. Effects of Exercise on Muscle Fitness in Dialysis Patients: A Systematic Review and Meta-Analysis. Am J Nephrol. 2019;50(4):291–302.
24. Moorman D, Suri R, Hiremath S, Jegatheswaran J, Kumar T, Bugeja A, et al. Benefits and Barriers to and Desired Outcomes with Exercise in Patients with ESKD. Clin J Am Soc Nephrol. 2019 Feb 7;14(2):268–76.
25. Sheng K, Zhang P, Chen L, Cheng J, Wu C, Chen J. Intradialytic exercise in hemodialysis patients: a systematic review and meta-analysis. Am J Nephrol. 2014;40(5):478–90.
26. Capitanini A, Lange S, D'Alessandro C, Salotti E, Tavolaro A, Baronti ME, et al. Dialysis exercise team: the way to sustain exercise programs in hemodialysis patients. Kidney Blood Press Res. 2014;39(2-3):129–33.
27. Isoyama N, Qureshi AR, Avesani CM, Lindholm B, Bàràny P, Heimbürger O, et al. Comparative associations of muscle mass and muscle strength with mortality in dialysis patients. Clin J Am Soc Nephrol. 2014 Oct 7;9(10):1720–8.
28. Lopes AA, Lantz B, Morgenstern H, Wang M, Bieber BA, Gillespie BW, et al. Associations of self-reported physical activity types and levels with quality of life, depression symptoms, and mortality in hemodialysis patients: the DOPPS. Clin J Am Soc Nephrol. 2014 Oct 7;9(10):1702–12.
29. Heiwe S, Jacobson SH. Exercise training for adults with chronic kidney disease. Cochrane Database Syst Rev. 2011 Oct 5;2011(10):CD003236.
30. Beddhu S, Baird BC, Zitterkoph J, Neilson J, Greene T. Physical activity and mortality in chronic kidney disease (NHANES III). Clin J Am Soc Nephrol. 2009 Dec;4(12):1901–6.
31. Cheema B, Abas H, Smith B, O'Sullivan A, Chan M, Patwardhan A, et al. Progressive exercise for anabolism in kidney disease (PEAK): a randomized, controlled trial of resistance training during hemodialysis. J Am Soc Nephrol. 2007 May;18(5):1594–601.
32. Kouidi E. Exercise training in dialysis patients: why, when, and how? Artif Organs. 2002 Dec;26(12):1009–13.
