Introduction
In breast-conserving therapy, whole-breast radiotherapy (WBRT) followed by a postoperative tumor bed boost is the standard approach for patients at increased risk of local recurrence [1–6]. However, postoperative boost delivery is limited by challenges in tumor bed delineation after surgery, particularly in the setting of oncoplastic reconstruction, and may prolong overall locoregional treatment time. Historical concerns regarding wound healing have limited the use of preoperative breast radiotherapy, though these concerns largely stem from experiences with whole-breast irradiation rather than limited-volume boost dosing [7,8].
We previously published "Preoperative Radiation Boost Results in Low Rates of Re-excision and Reduced Locoregional Treatment Time in Breast Cancer Patients," which highlighted these two critical findings from a phase 2 prospective clinical trial evaluating the feasibility of preoperative radiation boost in patients with breast cancer [9,10]. Our findings support a paradigm shift in boost sequencing by demonstrating that a preoperative radiation boost delivered before lumpectomy has low rates of wound complications, comparable to those of the standard of care. We also showed that the volume of the boost in the preoperative setting was significantly smaller than would be implemented in the postoperative setting [10].
We present in this article a more focused report of the findings from our original trial and the subsequent findings that we published later when the cohort was expanded [9,10].
Methods
Our prospective, single-arm phase II trial (NCT04871516) enrolled patients with mammographically detected, biopsy-proven, clinically node-negative early-stage invasive breast cancer or ductal carcinoma in situ (cTis–2, cN0, cM0) who are eligible for breast-conserving surgery [9,10]. The study was not blinded to any of the treating physicians. Target delineation used breast MRI fused with planning CT and mammographic data, with the biopsy clip serving as the epicenter for gross tumor volume definition. Target volumes for the boost were defined preoperatively, before tissue rearrangement or seroma formation. Patients received 13.32 Gy in 4 daily fractions using photons or electrons before lumpectomy with or without sentinel lymph node biopsy done 1–3 weeks post-boost, and finally, whole breast radiation therapy consisting of 36.63 Gy in 11 daily fractions was given 3-8 weeks after the final surgical procedure (Figure 1).
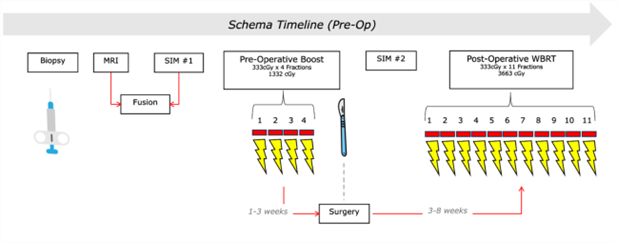
This hypofractionated regimen is consistent with previously validated institutional protocols [11]. Pathologically node-positive patients were allowed nodal irradiation at the radiation oncologist's discretion.
We also evaluated re-excision rates and time from diagnostic biopsy to completion of radiotherapy, compared with both a contemporary institutional cohort treated with standard postoperative boost sequencing and a prior institutional trial using identical fractionation with a postoperative boost [11].
Results
Of 55 patients who were enrolled in the original trial, 50 patients had complete data with at least 1-month follow-up and were evaluable for this analysis. The median age was 64 years old (range, 40-77 years). The median follow-up time was 12 months (range, 1-25 months). Twenty-five patients were White (50%) and the other 25 were African American, Asian, Hispanic, and others.
The cohort of patients for the trial was expanded. Re-excision data was available for a total of 89 patients. Preoperative radiation boost was associated with a marked reduction in re-excision rates, with only 3.4% requiring additional surgery for positive margins. This rate was significantly lower than the rate reported in the literature (17.2%, p=0.0005). Moreover, it was also significantly lower than the re-excision rate of the Canadian hypofractionation cohort (13.48%; p = 0.015) [11–14]. Cavity shave margins were performed in 47.2% of these patients compared to 75.3% in the Canadian cohort. Margins were defined per the 2013 published ASTRO/SSO guidelines [19].
From a treatment-delivery standpoint, the overall locoregional treatment time was significantly shortened, with a median of 109 days from biopsy to completion of radiotherapy. This represented a reduction of approximately 2–3 weeks compared with both historical and contemporary postoperative boost cohorts. The shortened timeline was primarily attributable to the ability to initiate radiotherapy earlier than surgery could be scheduled and to avoid postoperative delays related to re-excision.
Wound complication rates were low (2% grade 3 wound dehiscence, 6% grade 2 wound infections, and 10% grade 2 radiation dermatitis), supporting the feasibility of preoperative limited-volume irradiation [10].
In 49 patients, physician-reported cosmetic outcomes were available based on the Harvard scale. At the last follow-up, 92% had good or excellent cosmetic outcomes, and 8% were reported to have a fair cosmetic outcome. No patient had a poor cosmetic outcome.
Recurrence rates have not been analyzed yet as the data has not matured to date. However, none has been observed so far.
Discussion
Besides the observed immediate clinical benefits of preoperative radiation, there is an underlying effect on tumor biology and the tumor microenvironment that can result in improvement in long-term local and possibly systemic outcomes. These effects can be a focus for further investigative studies. In a small pilot study, the YOUNGSTER trial, investigators found that there was a late increase in adaptive immunity gene expression along with a significant increase in the IGG signature after delivering a preoperative radiation boost (5 x 2.67 Gy) to patients with early stage breast cancer and who are eligible for this therapy postoperatively. Although TILs did not show a statistically significant increase, a trend was noted [15]. Other studies have also identified radiation-induced upregulation of genes related to immunity, inflammation, and cell death in early-stage breast cancer [16,17].
Preoperative boost delivery offers several theoretical and practical advantages. First, target volume definition is inherently more precise before surgical disruption, oncoplastic tissue rearrangement, or seroma evolution, potentially reducing geographic miss and unnecessary irradiation of normal breast tissue. Second, preoperative radiation is delivered in a non-hypoxic tumor microenvironment, which may enhance radiobiologic effectiveness. Third, the approach avoids the need for postoperative tumor bed localization, a well-recognized challenge in modern oncoplastic practice that can lead to boost omission or increased target volumes.
The hypofractionated boost dose used in this trial results in a moderate biologically effective dose, potentially mitigating fibrosis and cosmetic toxicity compared with higher single-fraction or hypofractionated preoperative boost regimens reported elsewhere. Although long-term local control and cosmetic outcomes are still maturing, the favorable surgical and logistical outcomes observed suggest that preoperative boost sequencing may represent a meaningful refinement of contemporary breast radiotherapy workflows.
Reoperation rates following breast conserving surgery range from 10–40%, with significant variation across institutions and surgeons [12–14]. Although more recent data show rates declining to approximately 12–20% following adoption of consensus guidelines [18,19]. Despite that our sample size is much smaller, the significantly lower rates of re-excision that we observed with our schema (3.4%) are very promising, especially given that cavity shave margins were excised in only 47.2% of the cases, indicating that preoperative radiation boost can be a viable tool to avoid re-operative procedures for margins, which, as multiple studies have previously shown, worsen cosmetic outcomes [19], increase the cost of care, lead to higher surgical complication rates, and cause psychological distress on the patients [20].
Preoperative radiation therapy can potentially be applied to a broader patient population than those with early-stage low-risk disease, with different schemas applied to other scenarios. The European Society for Therapeutic Radiology and Oncology (ESTRO) reviewed 27 phase I and phase II studies from 2000 onward, examining preoperative RT in various breast cancer settings, including early-stage low-risk disease, high-risk disease, and patients requiring mastectomy [21]. A 2025 narrative review in the International Journal of Radiation Oncology specifically focused on locally advanced breast cancer, highlighting encouraging outcomes with potential benefits in both clinical and cosmetic results [22]. We are currently prospectively evaluating preoperative partial breast irradiation and whether long-term outcomes are non-inferior to traditional adjuvant radiation therapy.
Preoperative radiation, in any schema, also has the potential to change how we define quality measures in locoregional treatment of breast cancer. Currently, the time from breast cancer diagnosis to surgical treatment [23,24], and separately to completion of radiation therapy [25–28] are regarded as surrogates for oncological outcomes. However, in patients receiving radiation therapy, and if part or all of the radiation therapy was delivered before surgical intervention, based on well-defined intervals proven to be feasible in prospective studies, the combined time from diagnosis to completion of surgical and radiation therapies might be a more appropriate measure.
Conclusion
Despite some limitations such as a small number of patients, single arm design, and potential bias toward patient selection, this phase II experience demonstrates that preoperative radiation boost delivery is feasible, well-tolerated, and associated with improved surgical outcomes and reduced overall treatment duration. By enhancing target delineation, streamlining care, and reducing re-excision rates, preoperative boost sequencing warrants further investigation in randomized, multi-institutional trials—particularly in patients undergoing oncoplastic breast-conserving surgery.
References
2. Bartelink H, Horiot JC, Poortmans P, Struikmans H, Van den Bogaert W, Barillot I, et al. Recurrence rates after treatment of breast cancer with standard radiotherapy with or without additional radiation. N Engl J Med. 2001 Nov 8;345(19):1378–87.
3. Bartelink H, Maingon P, Poortmans P, Weltens C, Fourquet A, Jager J, et al. Whole-breast irradiation with or without a boost for patients treated with breast-conserving surgery for early breast cancer: 20-year follow-up of a randomised phase 3 trial. Lancet Oncol. 2015 Jan;16(1):47–56.
4. Bartelink H, Horiot JC, Poortmans PM, Struikmans H, Van den Bogaert W, Fourquet A, et al. Impact of a higher radiation dose on local control and survival in breast-conserving therapy of early breast cancer: 10-year results of the randomized boost versus no boost EORTC 22881-10882 trial. J Clin Oncol. 2007 Aug 1;25(22):3259–65.
5. Polgár C, Fodor J, Orosz Z, Major T, Takácsi-Nagy Z, Mangel LC, et al. Electron and high-dose-rate brachytherapy boost in the conservative treatment of stage I-II breast cancer first results of the randomized Budapest boost trial. Strahlenther Onkol. 2002 Nov;178(11):615–23.
6. Romestaing P, Lehingue Y, Carrie C, Coquard R, Montbarbon X, Ardiet JM, et al. Role of a 10-Gy boost in the conservative treatment of early breast cancer: results of a randomized clinical trial in Lyon, France. J Clin Oncol. 1997 Mar;15(3):963–8.
7. Riet FG, Fayard F, Arriagada R, Santos MA, Bourgier C, Ferchiou M, et al. Preoperative radiotherapy in breast cancer patients: 32 years of follow-up. Eur J Cancer. 2017 May;76:45–51.
8. Semiglazov VF, Topuzov EE, Bavli JL, Moiseyenko VM, Ivanova OA, Seleznev IK, et al. Primary (neoadjuvant) chemotherapy and radiotherapy compared with primary radiotherapy alone in stage IIb-IIIa breast cancer. Ann Oncol. 1994 Sep;5(7):591–5.
9. Chakraborty MA, Sherwani ZK, Narra LR, Abou Yehia Z, Ohri N, Cui T, et al. Preoperative Radiation Boost Results in Low Rates of Re-excision and Reduced Locoregional Treatment Time in Breast Cancer Patients. Ann Surg Oncol. 2025 Dec;32(13):9836–42.
10. Yehia ZA, Sherwani Z, Chakraborty M, Ohri N, Grann A, Eladoumikdachi F, et al. First Results of the Primary Outcome of a Phase 2 Prospective Clinical Trial to Assess the Feasibility of Preoperative Radiation Boost in Patients With Breast Cancer. Int J Radiat Oncol Biol Phys. 2025 Feb 1;121(2):333–40.
11. Ahlawat S, Haffty BG, Goyal S, Kearney T, Kirstein L, Chen C, et al. Short-Course Hypofractionated Radiation Therapy With Boost in Women With Stages 0 to IIIa Breast Cancer: A Phase 2 Trial. Int J Radiat Oncol Biol Phys. 2016 Jan 1;94(1):118–25.
12. Racz JM, Glasgow AE, Keeney GL, Degnim AC, Hieken TJ, Jakub JW, et al. Intraoperative Pathologic Margin Analysis and Re-Excision to Minimize Reoperation for Patients Undergoing Breast-Conserving Surgery. Ann Surg Oncol. 2020 Dec;27(13):5303–11.
13. McCahill LE, Single RM, Aiello Bowles EJ, Feigelson HS, James TA, Barney T, Engel JM, Onitilo AA. Variability in reexcision following breast conservation surgery. JAMA. 2012 Feb 1;307(5):467–75.
14. Silva E, Tan M. The Impact of Margin Status on Breast Conservation Rates. Ann Surg Oncol. 2019 Nov;26(12):3931–38.
15. Gomà C, Mollà M, Oses G, Sanfeliu E, Rodríguez-Hernández A, González-Farré B, et al. Preoperative Radiation Therapy-Induced Molecular and Immune Modulation in Early-Stage Breast Cancer: Results From the YOUNGSTER trial. Int J Radiat Oncol Biol Phys. 2026 Jan 1;124(1):122–32.
16. Horton JK, Blitzblau RC, Yoo S, Geradts J, Chang Z, Baker JA, et al. Preoperative Single-Fraction Partial Breast Radiation Therapy: A Novel Phase 1, Dose-Escalation Protocol With Radiation Response Biomarkers. Int J Radiat Oncol Biol Phys. 2015 Jul 15;92(4):846–55.
17. Bosma SCJ, Hoogstraat M, van der Leij F, de Maaker M, Wesseling J, Lips E, et al. Response to Preoperative Radiation Therapy in Relation to Gene Expression Patterns in Breast Cancer Patients. Int J Radiat Oncol Biol Phys. 2020 Jan 1;106(1):174–81.
18. Marinovich ML, Noguchi N, Morrow M, Houssami N. Changes in Reoperation After Publication of Consensus Guidelines on Margins for Breast-Conserving Surgery: A Systematic Review and Meta-analysis. JAMA Surg. 2020 Oct 1;155(10):e203025.
19. Moran MS, Schnitt SJ, Giuliano AE, Harris JR, Khan SA, Horton J, et al. Society of Surgical Oncology-American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. Int J Radiat Oncol Biol Phys. 2014 Mar 1;88(3):553–64.
20. Heil J, Breitkreuz K, Golatta M, Czink E, Dahlkamp J, Rom J, et al. Do reexcisions impair aesthetic outcome in breast conservation surgery? Exploratory analysis of a prospective cohort study. Ann Surg Oncol. 2012 Feb;19(2):541–7.
21. D'Archi S, Carnassale B, Accetta C, De Lauretis F, Di Guglielmo E, Di Leone A, et al. Re-Excision After Positive Margins in Breast-Conserving Surgery: Can a Risk-Based Strategy Avoid Unnecessary Surgery? J Clin Med. 2025 Aug 18;14(16):5839.
22. Zamagni A, Montero Luis A, Mjaaland I, Chirila ME, Ciervide R, Machiels M, et al. ESTRO recommendations on preoperative radiation therapy in breast cancer: current and future perspectives - Endorsed by ASTRO. Radiother Oncol. 2026 Jan;214:111236.
23. Arif A, Khan AJ. Preoperative Radiation Therapy in Locally Advanced Breast Cancer: A Narrative Review. Int J Radiat Oncol Biol Phys. 2026 Mar 1;124(3):622–40.
24. Wiener AA, Hanlon BM, Schumacher JR, Vande Walle KA, Wilke LG, Neuman HB. Reexamining Time From Breast Cancer Diagnosis to Primary Breast Surgery. JAMA Surg. 2023 May 1;158(5):485–92.
25. Ungvari Z, Fekete M, Buda A, Lehoczki A, Munkácsy G, Scaffidi P, et al. Quantifying the impact of treatment delays on breast cancer survival outcomes: a comprehensive meta-analysis. Geroscience. 2026 Feb;48(1):1173–87.
26. Bleicher RJ, Moran MS, Ruth K, Edge SB, Dietz JM, Wilke LG, et al. The Impact of Radiotherapy Delay in Breast Conservation Patients Not Receiving Chemotherapy and the Rationale for Dichotomizing the Radiation Oncology Time-Dependent Standard into Two Quality Measures. Ann Surg Oncol. 2022 Jan;29(1):469–81.
27. Olivotto IA, Lesperance ML, Truong PT, Nichol A, Berrang T, Tyldesley S, et al. Intervals longer than 20 weeks from breast-conserving surgery to radiation therapy are associated with inferior outcome for women with early-stage breast cancer who are not receiving chemotherapy. J Clin Oncol. 2009 Jan 1;27(1):16–23.
28. Haviland JS, Bentzen SM, Bliss JM, Yarnold JR; START Trial Management Group. Prolongation of overall treatment time as a cause of treatment failure in early breast cancer: An analysis of the UK START (Standardisation of Breast Radiotherapy) trials of radiotherapy fractionation. Radiother Oncol. 2016 Dec;121(3):420–3.
