Abstract
Purpose: Trastuzumab emtansine (T-DM1), an antibody-drug conjugate consisting of the humanized monoclonal antibody trastuzumab covalently linked to the cytotoxic agent DM1, is utilized for the management of human epidermal growth factor receptor-2 (HER2)-positive metastatic breast cancer (mBC), after progression on prior trastuzumab. Its regulatory approval preceded the era before pertuzumab approval. In the contemporary era, pertuzumab and/or trastuzumab is employed in routine clinical practice in the first-line (1L) setting. It is therefore relevant to consider currently utilized regimens to confirm their role in current clinical practice. The current study evaluated real-world evidence (RWE) for the efficacy and safety of T-DM1 in HER2-positive mBC and compare with evidence from the clinical trial program.
Methods: Searches were performed in PubMed, Embase, and Cochrane for eligible studies from inception until May 22, 2024. Screening was performed using AccuScreener module of SymproAI online software. Variables of interest included overall response rate (ORR), progression-free survival (PFS), overall survival (OS), and grade ≥3 adverse events (AEs).
Results: Fifty-five publications were identified; 19 from 12 clinical trials and 36 from 34 real-world studies. ORR ranged between 25.0–50.4% (clinical trials) and 14.3–80.0% (real-world studies). Corresponding median PFS was 4.3–9.6 months and 2.8–12.0 months while the median OS was 22.7–34.3 and 12.0–45.0 months, respectively. Furthermore, real-world data suggest the benefit of T-DM1 is more marked in patients without prior pertuzumab. Evidence from both trials and real-world studies support the use of T-DM1 in patients with brain/central nervous system (CNS) metastasis. Safety data from the studies demonstrated a consistent safety profile in terms of AEs (≥grade 3), with thrombocytopenia being the most frequent.
Conclusion: This review demonstrated T-DM1 as an effective treatment for HER2-positive mBC in the second-line (2L) or later setting and has an acceptable safety profile.
Keywords
Breast Cancer, HER2 Gene, Trastuzumab emtansine, Treatment outcome, Brain metastasis
Introduction
Human epidermal growth factor receptor-2 (HER2)-directed therapies have transformed the management of HER2-positive breast cancer (BC), a subtype previously associated with poor prognosis [1]. These therapies currently constitute part of recommended regimens for most lines of therapy for recurrent or metastatic HER2-positive BC [2,3]. There are three classes of approved HER2-directed therapies: anti-HER2 monoclonal antibodies (e.g., trastuzumab and pertuzumab) [4–6]; antibody-drug conjugates (ADC) (e.g., trastuzumab emtansine [T-DM1] and trastuzumab deruxtecan [T-DXd]) [7,8]; and tyrosine kinase inhibitors (TKIs; e.g., lapatinib, tucatinib and neratinib) [9]. In the contemporary era, the dual blockage of HER2 using trastuzumab and pertuzumab in combination with chemotherapy (CT) constitutes the standard first-line (1L) treatment regimen [10], while T-DM1 is used for the management of metastatic BC (mBC), and is indicated for use after disease progression on prior treatment with trastuzumab and a taxane [7].
Although the European Society for Medical Oncology (ESMO; and Pan Asian adapted ESMO clinical guidelines) and National Comprehensive Cancer Network clinical guidelines provide recommendations on the sequencing of HER2-directed therapies up to the third-line (3L) [2,3,11], some therapies may be unavailable in some regions or inappropriate for certain patients due to cost and safety profile.
The treatment landscape for HER2-positive metastatic breast cancer (mBC) has shifted significantly over the past five years with the introduction of newer therapies, such as pertuzumab. As a result, clinical trial data for agents like T-DM1, approved more than five years ago, may no longer align with the current clinical practices. These trials, conducted before pertuzumab’s approval, typically enrolled heavily pretreated patients who had progressed on one or more prior HER2-directed therapies. Today’s patient populations and treatment approaches differ, raising questions about whether the efficacy and safety outcomes from those historical trials still apply in modern real-world settings [12–14].
The clinical efficacy and safety of T-DM1, reported among clinical trials in the treatment of HER2-positive BC, have been systematically reviewed and meta-analyzed [15–17], however, limited reports have reviewed the findings from observational or real-world settings [18] or, have compared the findings of clinical trials with that of observational studies [19]. The growing integration of real-world evidence (RWE) in clinical practice is becoming crucial for complementing findings from randomized controlled trials (RCTs). In this context, T-DM1 has been established as an effective treatment for HER2-positive mBC. However, RWE on T-DM1’s effectiveness and safety in comparison to observations from clinical trials remains limited. To address the gap, this systematic literature review (SLR) aims to corroborate clinical trial results with RWE, with a specific focus on studies comparing the efficacy/effectiveness and safety of T-DM1 in patients with HER2-positive mBC.
Methods
This SLR was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis protocol (PRISMA-P) [20] and was registered on PROSPERO (ID: CRD42023410972). The PubMed, Embase, and Cochrane search strategies were designed to identify relevant publications on T-DM1 efficacy and safety. Searches were performed on 22nd May 2024, without geographic or time restrictions. The search strategies and outputs are detailed in Table S1.
Publications were screened to identify relevant evidence as per the eligibility for inclusion and exclusion criteria (Table S2). Studies incongruent with the protocol were excluded. The titles and abstracts of publications from the searches were screened using AccuScreener, a screening module [21] within SymproAI, an online Artificial Intelligence based Systematic Literature Management and Processing software for end-to-end automation of systematic reviews [22]. The AccuScreener output was cross-checked manually by a team of two expert researchers. The AccuScreener output was cross-checked manually by a team of two expert researchers. The full-text of included publications were reviewed by two researchers. A senior systematic reviewer authenticated the results and resolved any uncertainties at both the title/abstract and full-text screening steps. Data from the included publications were extracted by one researcher and reviewed by a second researcher. Extracted efficacy data included overall response rate (ORR), progression-free survival (PFS), overall survival (OS), and safety data included grade 3 or worse adverse events (AEs). The substantial methodological heterogeneity between included studies precluded formal meta-analysis, therefore, a robust descriptive approach was employed.
Quality appraisal with the risk of bias (RoB) analysis for all the included full-text publications were performed using validated Cochrane RoB tools in ROB Master module of SymproAI [22]. Quality assessment of randomized controlled trials (RCTs) was performed using the Cochrane Collaboration RoB 2.0 tool [23]. The RoB 2.0 assessed bias across five domains: (1) randomization process, (2) deviations from intended interventions, (3) missing outcome data, (4) measurement of the outcome, and (5) selection of the reported result. An overall RoB judgment was assigned to each study based on domain-specific assessments. The Cochrane ROBINS-I tool was used for non-randomized trials and real-world studies [23,24]. The ROBINS-I evaluated seven domains of potential bias: (1) bias due to confounding, (2) bias in participant selection, (3) bias in classification of interventions, (4) bias due to deviations from intended interventions, (5) bias due to missing data, (6) bias in outcome measurement, and (7) bias in selection of reported results. Each study was assigned an overall RoB rating based on the highest risk level identified across all domains.
Results
Overview of identified studies
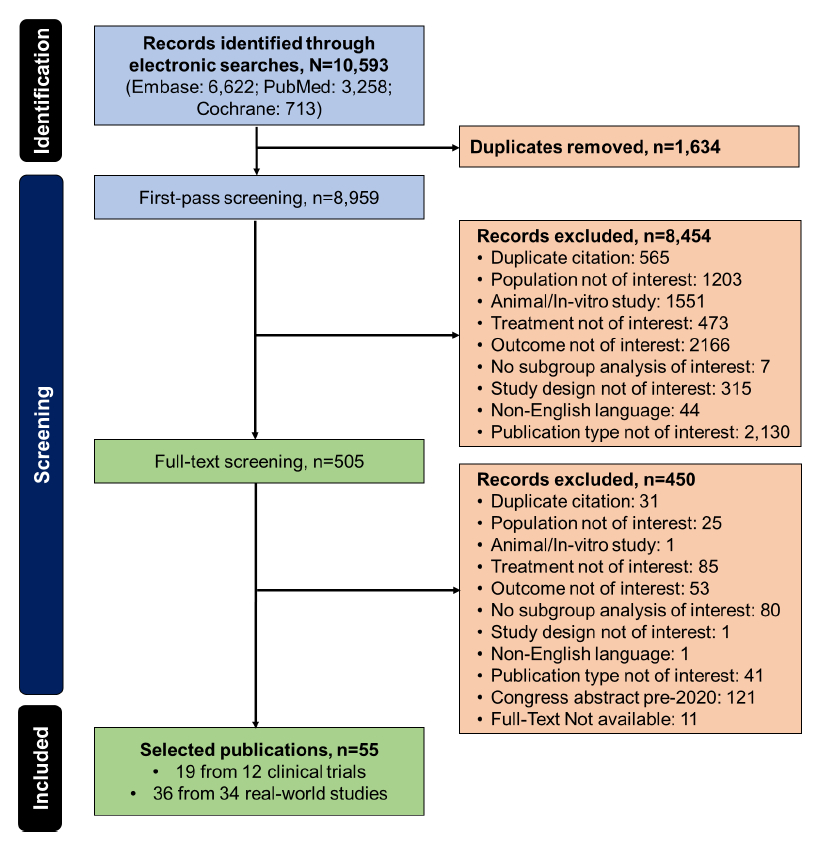
Overall, 55 publications (42 full-text articles and 13 congress abstracts) (Figure 1) that explored T-DM1 efficacy and safety were identified. Of these, 19 publications were from 12 different clinical trials. This included multiple publications from DESTINY-Breast03 (four publications) [25–28], EMILIA (four publications) [29–32], and KAMILLA (two publications) [33,34] while the remaining nine trials each described distinct studies [35–43]. A total of 36 publications were reported from real-world studies, of which a “field practice study” had three publications [44–46] and the remaining 33 studies each contributed a single publication [47–79]. The median age of patients ranged from 42 to 64.5 years across RWE studies and clinical trials. Where reported, ECOG performance status ranged from 0 to ≤2 (Tables 2–7). An overview of studies with multiple publications (primary and secondary) summarizing the overlapping cohorts across clinical trials (DESTINY Breast03, EMILIA, and KAMILLA) and real-world study (FIELD PRACTICE) has been presented as Table 1.
Figure 1. Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram of study selection.
|
Acronym |
Trial identifier |
Total publications |
Study design |
Primary publication details |
Secondary publications and its details |
Outcomes (details presented in tables) |
|
A. Clinical trials |
||||||
|
DESTINY Breast03 |
NCT03529110 |
Four |
Randomized, open-label, multi-center, active-controlled, phase 3 |
Cortés et al. 2022 [80]
|
Hurvitz et al. 2023 [26]
Hurvitz et al. 2024 [27]
Im et al. 2022 [28]
|
Efficacy (Table 2)
Brain metastases (Table 5)
Safety (grade ≥3) (Table 6)
Safety (Any grade) (Table S3)
|
|
EMILIA |
NCT00829166 |
Four |
Randomized, open-label, multi-centre, phase 3 |
Verma et al. 2012 [32]
|
Diéras et al. 2017 [29]
Krop et al. 2015 [31]
Im et al. 2021 [30]
|
Efficacy (Table 2)
Brain metastases (Table 5)
Safety (grade ≥3) (Table 6)
Safety (Any grade) (Table S3)
|
|
KAMILLA |
NCT01702571 |
Two |
Single-arm, On-going, phase 3b |
Wuerstlein et al. 2022 [34]
|
Montemurro et al. 2020 [33]
|
Efficacy (Table 2)
Brain metastases (Table 5)
Safety (grade ≥3) (Table 6)
Safety (Any grade) (Table S3)
|
|
B. Real–world study |
||||||
|
Field-practice study |
NR |
Three |
Retrospective analysis |
Fabi et al. 2017 [45]
|
Fabi et al. 2017 [46]
Fabi et al. 2018 [44]
|
Efficacy as per prior LoT (Table 3)
Efficacy as per prior pertuzumab regimens (Table 4)
Brain metastases (Table 5)
Safety (grade ≥3) (Table 7)
Safety (Any grade) (Table S4)
|
|
Abbreviations: 1L: First line of treatment; LoT: Line of Treatment. |
||||||
Among the 19 publications from clinical trials, 15 investigated HER2 positive mBC patients with/without brain metastasis [25,26,28–30,32,34–40,42,43], three reported subgroup analysis on brain metastasis [27,31,33] while one reported only safety outcomes [41].
Efficacy
Clinical trials
Fifteen publications from 11 clinical trials (including five phase 2, five phase 3 clinical trials, and one T-PAS expanded-access study) reported efficacy outcomes for T-DM1 utilized as second-line (2L) therapy in patients who had progressed on prior treatment with ≥1 taxane and trastuzumab or other anti-HER2 regimen (Table 2).
|
Study (Primary / Secondary Publication) |
Study details (Country, Study name, design) |
Population; N; eligibility criteria |
Median follow-up (months) |
ORR, % (95% CI) |
mPFS, months (95% CI) |
mOS, months (95% CI) |
|
A. DESTINY-Breast03 (three publications) |
||||||
|
Cortés et al. 2022 [80] (Primary) |
Global
DESTINY-Breast03 (NCT03529110)
Randomized, open-label, multi-centre, active-controlled, phase 3 |
N=524; prior taxane + trastuzumab
T-DM1, n: 263 prior anti-HER2 therapy
Patient subgroups
|
|
T-DM1
T-Dxd
|
T-DM1: 6.8 (5.6–8.2) T-DXd: NE (18.5–NE) HR: 0.28 (95% CI, 0.22-0.37)
TDM1 vs T-Dxd
|
NR |
|
Hurvitz et al. 2023 [26] (Secondary) |
|
T-DXd, n: 261 prior anti-HER2 therapy
Patient subgroups
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
T-DXd: 106 (40.6) |
|
|
|
|
|
Im et al. 2022 [28] (Secondary) |
Asia |
Asians, n: 309
|
NR |
NR |
Asians
|
NR |
|
B. EMILIA (three publications) |
||||||
|
Verma et al. 2012 [32] (Primary)
|
Global
EMILIA (NCT00829166)
Randomized, open-label, multi-centre, phase 3 |
N=991; prior taxane + trastuzumab
|
First interim analysis (for all parameter): 13
Second interim analysis (Only for OS: 19 |
|
|
|
|
Diéras et al. 2017 [29] (Secondary) |
|
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
ECOG NA, n (%)
Lapatinib plus capecitabine: 8 (2) |
|
NR |
NR |
|
|
Im et al. 2021 [30] (Secondary) |
Asia |
N=158; Asians with prior taxane + trastuzumab
|
|
|
|
|
|
C. Other trials |
||||||
|
Wang et al. 2023 [40] (Primary)
|
China
ELAINA (NCT03084939)
Randomized, open-label bridging, multi-centre, phase 3 |
N=200; prior taxane + trastuzumab
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
|
Overall
For OS
|
|
|
|
|
Wildiers et al. 2022 [42] (Primary)
|
Europe
EORTC 75111-10114 ETF/BCG
Randomized, open-label, multi-centre, investigator-initiated, phase 2 |
N=40; prior trastuzumab + pertuzumab without or with metronomic oral cyclophosphamide
Median age, years
WHO PS 0, n (%)
WHO PS 1, n (%)
WHO PS 2, n (%)
WHO PS 3, n (%)
|
33.7 |
25.0 |
NR |
NR |
|
Wuerstlein et al. 2022 [34] (Primary)
|
Global and Asian
KAMILLA (NCT0170257)
Single-arm, multi-centre, phase 3b |
N=2,183; prior anti-HER2 + CT
Median age, years
ECOG PS 0, n (%)
ECOG PS 1, n (%)
ECOG PS 2, n (%)
ECOG PS missing, n (%)
Total (by prior LoT)
|
|
|
Total (by prior LoT)
|
Total (by prior LoT)
|
|
Krop et al. 2017 [38] (Primary)
|
Global
TH3RESA (NCT01419197)
Randomized, open-label, multi-centre, phase 3 |
N=602; ≤2 prior HER2-directed therapy (trastuzumab and lapatinib and a taxane)
Median age, years
ECOG PS 0, n (%)
ECOG PS 1, n (%)
ECOG PS 2, n (%)
|
30.5 |
NR |
NR |
Stratified HR: 0·68 (95% CI, 0·54–0·85); p=0·0007 |
|
Kashiwaba et al. 2016 [37] (Primary)
|
Japan
JO22997
Single-arm, multi-centre, phase 2 |
N=73; ≥1 prior CT + trastuzumab
Median age, years: 58
n (%)
|
|
38.4 (28.8–48.6)
|
5.6 (4.6–8.2) |
30.5 (25.2–not reached) |
|
Yardley et al. 2015 [43] (Primary)
|
USA
T-PAS (TDM4884g/NCT01120561)
Single-arm, multi-centre, expanded access |
N=215; ≤2 prior HER2-directed therapy (trastuzumab and lapatinib)
Median age, years: 53
n (%)
|
5.9 |
Among the 172 patients with measurable disease: 25.6 (19.2–32.8) |
Of the 202 patients with available efficacy data: 6.8 (5.7–7.6) |
NR |
|
Gupta et al. 2013 [36] (Primary)
|
NR
TDM4688g/NCT00943670
Single-arm, multi-centre, phase 2 |
N=51; prior trastuzumab
Single-agent T-DM1 with HER2-positive disease, n: 38
Median age, years: 53.5
n (%)
|
NR |
|
4.3 (4.0–6.7) |
NR |
|
Krop et al. 2012 [39] (Primary)
|
NR
TDM437g
Single-arm, phase 2 |
N=110; ≤2 prior HER2-directed therapy (trastuzumab, lapatinib, an anthracycline, a taxane, and capecitabine)
Median age, years: 52.5
n (%)
|
17.4 |
34.5 (26.1–43.9)
|
6.9 (4.2–8.4) |
NR |
|
Burris et al. 2011 [35] (Primary)
|
USA
TDM4258g
Single-arm, phase 2 |
N=112; ≥1 prior HER2-targeted therapy
Median age, years: 54.5
n (%)
|
≥12 |
25.9 (18.4–34.4) |
4.6 (3.9–8.6) |
NR |
|
Abbreviations: CI: Confidence Interval; CT: Chemotherapy; HER2: Human Epidermal Growth Factor Receptor 2; HR: Hazard Ratio; ITT: Intent-to-Treat; LoT: Line of Therapy; mOS: Median Overall Survival; mPFS: Median Progression-Free Survival; NE: Not Estimable; NR: Not Reported; ORR: Overall Response Rate; T-DM1: Trastuzumab Emtansine; T-DXd: Trastuzumab Deruxtecan; TKI: Tyrosine Kinase Inhibitor: TP: Trastuzumab: Pertuzumab; TPM: Trastuzumab: Pertuzumab: Metronomic Cyclophosphamide |
||||||
The single-arm phase 2 trials involved heavily pre-treated patients who previously received HER2-directed therapies; across the studies, the median number of prior lines of therapy (LoT) ranged between 3.0–8.5 in the metastatic setting [35–37,39]. The randomized two-arm phase 3 trials and an expanded access study evaluated patients that previously received HER2-directed therapies: trastuzumab plus taxane in EMILIA, DESTINY and ELAINA [25–32,40], and trastuzumab plus lapatinib in TH3RESA and T-PAS [38,43]. An additional phase 3b safety trial (KAMILLA) only included patients with prior anti-HER2 treatment [33,34].
The ORR with T-DM1 in phase 3 studies ranged between 29.3–50.4% [26,30,34,40] and between 25.0–38.4% in phase 2 studies [35–37,39,42]. In the T-PAS expanded access study involving patients with measurable disease, ORR was 25.6% [43]. In comparison, T-DXd treatment was associated with a higher ORR of 79.0% in patients pre-treated with a taxane and trastuzumab with consistent rates observed in subgroups (79.6% and 79.8% in de-novo and recurrent populations, respectively) [25,26].
Median progression-free survival (mPFS) observed in clinical trials of T-DM1 ranged between 4.3–9.6 months [26,32,34–37,39,40,43]. In the phase 3 trials, mPFS was 9.6 months (versus 6.4 months for lapatinib plus capecitabine; 9.3 months in the Asian subgroup) in EMILIA [30,32] and 6.8 months in KAMILLA [34], the latter being consistent with DESTINY and T-PAS (each 6.8 months; versus not reached for T-DXd in DESTINY) [43,80]. Further subgroup analysis of KAMILLA revealed that the Asian cohort had a lower mPFS (5.7 months) compared with the global cohort (6.8 months) [34]. The phase 2 trials reported a mPFS that ranged between 4.3–6.9 months [36,39].
Median overall survival (mOS) observed in the phase 3 trials [26,29,30,34,38,40], ranged between 22.7–34.3 months and was not reached in DESTINY-BREAST03 trial [26]. T-DM1 mOS was 34.3 months (versus 22.7 months for lapatinib plus capecitabine) in EMILIA [30], 22.7 months (versus 15.8 months for physician’s choice) in TH3RESA [38], 27.2 months in KAMILLA [34], and was not reached (versus not reached for T-DXd) in DESTINY [26]. Only one phase 2 trial reported a mOS of 30.5 months in patients treated with T-DM1 [37].
Real-world studies
Thirty-six publications (34 real-world studies) reported data on T-DM1 therapy in patients who had progressed after ≥1 prior anti-HER2 regimen.
T-DM1 efficacy in pertuzumab pre-treated patients
The ORR ranged between 14.3–57.1% in six studies [46,48,54,56,66,78]. This appeared to be lower in pertuzumab-pretreated patients (14.3–40.2%) compared to pertuzumab-naïve patients (30.0–57.1%) [46,48,78]. A study by Fabi et al. (2020) also showed significantly lower ORR in pertuzumab pre-treated compared with naïve patients (33.3% vs. 57.1%; p=0.03) [46].
In patients who received T-DM1 as 2L therapy following 1L pertuzumab, mPFS ranged between 2.8–10.5 months (Table 3) [46,48,54,56,66,70–74,78], with one study suggested a potential association between longer 1L treatment duration and better mPFS (5.4 versus 10.8 months for <12 versus ≥12 months of 1L therapy, respectively; p=0.003) [71]. Compared with pertuzumab-naïve patients, 2L T-DM1 was generally associated with significantly shorter mPFS in pertuzumab pre-treated patients (7.8–12.0 versus 2.8–7.0 months, respectively; p<0.05) [46,72,74,78]. Seven studies demonstrated considerable variability in mOS (12.0–45.0 months) [48,55,65,66,71,74,78]. A trend towards a shorter mOS in pertuzumab pre-treated compared to pertuzumab-naïve patients (12.0–24.0 versus 14.4–45.0 months, respectively) was a consistent finding [48,65,74,78], although only one study (PANHER) reported a significant difference (24.0 vs. 45.0 months, p=0.002) [74]. Only two studies reported that prior pertuzumab treatment did not significantly impact mPFS (8.7 vs 8.3 months [51] and 6.18 vs 5.98 months [61]; p>0.05) or mOS (18.5 vs 20.6 months, p=0.86) [51].
|
Study details (Country, Study name, design, data source and study duration) |
Population, N; eligibility criteria |
Median follow-up (months) |
ORR, % (95% CI) |
mPFS, months (95% CI) |
mOS, months (95% CI) |
|
He et al. 2024 [61]
China
Multicentric, observational
March 2023–June 2023 |
N=101
Previous treatment, n (%)
Median age, years: 51
n (%)
Prior LoTs, n (%)
|
NR |
NR |
Prior pertuzumab
Prior pyrotinib
|
NR |
|
Peters et al. 2023 [73]
UK
Real-world analysis
2016–2021 |
N=85; 1L pertuzumab
Median age, years: 58
|
NR |
NR |
6.5 |
NR |
|
Moinard-Butot et al. 2022 [71]
France
Retrospective study
2008–2016 |
N=233; 1L taxane + pertuzumab + trastuzumab
Median age:
|
20.8 |
NR |
Overall: 7.1 (5.7–9.0)
1L treatment duration:
|
|
|
Al Rabadi et al. 2021 [48]
USA
Retrospective study
2013–2018 |
N=33, 1L pertuzumab
Median age, years
|
17 |
|
Pertuzumab-pretreated vs. naive: 9.5 (2.9–not reached) vs. 7.3; HR: 0.66 (95% CI: 0.30–1.47), p=0.310
|
Pertuzumab-pretreated vs. naive: Not reached vs. 14.4; HR: 0.56 (95% CI: 0.22–1.46), p=0.230 |
|
Daniels et al. 2021 [55]
Australia
Population-based retrospective study
2015–2019 |
N=309; 1L pertuzumab /trastuzumab
Median age, years: 58 |
30.7 |
NR |
NR |
18.9 (IQR: 7.9–29.5)
|
|
Migeotte et al. 2021 [70]
Belgium
Multi-centre observational retrospective study
2009–2016 |
N=51; Prior pertuzumab
Median age, years
|
NR |
NR |
Pts receiving T-DM1 with vs without prior taxane-trastuzumab-pertuzumab: 3.55 (2.27–11.38) vs 9.50 (6.15–12.26); p=0.144 |
NR |
|
Pizzuti et al. 2021 [74]
Italy
PANHER – multi-centre, observational study with retrospective design
2000–2020 |
N=1,328; 1L pertuzumab / trastuzumab; non-pertuzumab regimens
Median age, years: 52
ECOG PS, median (range): 1 (1-3) |
52 |
NR |
Overall: 8.0 1L pertuzumab: 7 (5.1–8.9) 1L trastuzumab or pertuzumab-naïve: 12.0 (5.8–18.2), p=0.01 |
1L pertuzumab: 24.0 (19.0–29.0) 1L pertuzumab naïve: 45.0 (36.0–54.0); p=0.002 |
|
Battisti et al. 2020 [51]
UK
Retrospective review
2014–2018 |
Median age, years: 55
n (%)
|
NR |
64.1 |
Overall: 8.7 (7.0–10.1)
|
Overall: 20.4 (17.0–22.1)
|
|
Conte et al. 2020 [54]
Italy
Retrospective/prospective observational multi-centre study
2000–2018 |
N=77; 1L taxane + pertuzumab / trastuzumab
Median age, years: 50
|
7 |
27.1 |
6.3 (4.8–7.7) |
Not reached
|
|
del Prete et al. 2020 [56]
Italy
Multi-centre, retrospective study |
N=135; 1L taxane + pertuzumab / trastuzumab
Median age, years: 56 |
10.3 |
20.7 |
10.5 (8.6–12.7) |
NR |
|
Martinez-Garcia et al. 2020 [66]
Spain
TDM1RM – retrospective observational study
2015–2020 |
N=52; 1L taxane + pertuzumab / trastuzumab
Median age, years: 52
%
|
33 |
42 |
8.4 (6.9–9.9) |
23.6 (17.5–29.7) |
|
Lupichuk et al. 2019 [65]
Canada
Retrospective study
2013–2016 |
N=104, 1L pertuzumab; pertuzumab-naïve
Median age, years
|
29.7 (14.0–57.5) |
NR |
NR |
Pertuzumab naïve vs. pretreated: 23.2 vs. 14.1; aHR: 0.60 (95% CI: 0.35–1.03), p=0.065 |
|
Noda-Narita et al. 2019 [72]
Japan
Retrospective study
2014–2017 |
N=42; 1L pertuzumab / trastuzumab or trastuzumab alone
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
ECOG 2, n (%)
|
4.8 |
NR |
|
NR |
|
Fabi et al. 2017 [46]
Italy
Multi-centre field-practice retrospective study
2012–2016 |
N=107; 1L taxane + pertuzumab/trastuzumab
Median age, years
|
NR |
1L pertuzumab: 33.3 1L trastuzumab: 57.1; p=0.03 |
1L pertuzumab vs. naive: 5.0 (4.3–5.7) vs.11.0 (7.8–14.2); HR: 2.02 (95% CI: 1.14–3.58), p=0.01 |
NR |
|
Vici et al. 2017 [78]
Italy
Retrospective observational study
2013–2016 |
N=245; 1L pertuzumab-trastuzumab-taxane
Median age, years: 56
%
|
15 |
|
|
|
|
Abbreviations: 1L: First-line; aHR: adjusted Hazard Ratio; BM: Bone Metastasis; CI: Confidence Interval; HER2: Human Epidermal Growth Factor Receptor 2; IQR: Interquartile Range; Lot: Line of Therapy; mOS: Median Overall Survival; mPFS: Median Progression-Free Survival; NR: Not Reported; ORR: Overall Response Rate; OS: Overall Survival; PFS: Progression Free Survival; pts: Patients; T-DM1: Trastuzumab Emtansine; UK: United Kingdom. |
|||||
T-DM1 efficacy in early versus later lines of therapy
Ten studies described T-DM1 efficacy in patients with HER2-positive mBC in early vs. later lines of therapy [45,50,53,59,62,64,68–70,77].
Where reported, most studies showed a decline in ORR, mPFS, and mOS in more heavily pre-treated patients versus those receiving T-DM1 as 2L therapy (Table 4). Early T-DM1 treatment showed better ORR (2L: 20.0–44.4%) [45,50,64,77] compared with heavily treated patients (≥4L: 17.1–20.3%) [50,68] which was also reflected in mPFS (2L: 7.7–12.0 months) compared with later lines (≥4L: 2.7–8.0 months) [45,50,59,69]. A similar pattern was also reported in studies that compared 1–4L versus 5–8L (12.9 vs. 6.6 months) [68] or 1–3L versus 4–10L (9.21 vs. 7.29 months) [70]. Consistently, one study reported a mOS of 41 months (2L) versus 17 months (5L) [50]. One-year OS rates were reported in two studies; this was 80% (2L) compared with 39% (≥4L) in one study [69], and 91% (1–4L) compared with 64% (5–8L) in another [68].
Seven other studies reported ORR (21.9–73.3%), mPFS (4.0–10.0 months) and mOS (17.3–not reached) with T-DM1 therapy in any LoT (Table 4) [49,52,57,60,63,67,79].
|
Study details (Country, Study name, design, data source and study duration) |
Population, N; eligibility criteria |
Median follow-up (months) |
ORR, % (95% CI) |
mPFS, months (95% CI), unless stated otherwise |
mOS, months (95% CI), unless stated otherwise |
|
A. Different line of therapy (LoT) |
|||||
|
Bahceci et al. 2021 [50]
Turkey
Retrospective multi-centre study |
N=414; prior trastuzumab /trastuzumab + pertuzumab /trastuzumab + lapatinib /CT
Median age, years: 47
T-DM1 lines, n
|
67.3±42.6
Post-T-DM1: 13.7±7.9 |
Last status at response
|
|
|
|
Migeotte et al. 2021 [70]
Belgium
Retrospective multi-centre study
2009–2016 |
T-DM1 lines, n
Median age, years
|
NR |
NR |
Overall: 9.01 (4.87–11.41)
|
NR |
|
Tataroglu Ozyukseler et al. 2021 [77]
Turkey
Single-centre retrospective, observational study
2016–2019 |
N=78; 1L trastuzumab-based regimens
Median age, years: 52
%
|
12.9 (7.4–19.8) |
Objective response rate: 25.6 |
7.8 (IQR, 3.7–19.5) |
21.1 (IQR, 8.7–30.9) |
|
Michel et al. 2020 [69]
Germany
PRAEGNANT – Ongoing prospective study |
N=76; 1L pertuzumab
Median age, years: 54.6
%
T-DM1 lines, n
|
3.1 |
NR |
Overall: 3.5 (2.8–7.8)
|
Overall: 22.5 (16.9–not reached)
|
|
Fabi et al. 2017 [45]
Italy
FIELD-PRACTICE – multi-centre study
2012–2016 |
N=282; Prior CT / hormonal therapies / anti-HER2 therapy
Median age, years: 51
T-DM1 lines, n
|
Overall: 10 For OS: 8 |
|
|
Not reached |
|
Michel et al. 2015 [68]
Germany
Retrospective chart review
2010–2014 |
N=23; Prior trastuzumab / lapatinib / pertuzumab
Median age, years: 54.6
%
Prior therapies, n (%)
T-DM1 lines, n (%):
|
285 (21–1016) days |
All: 26.0
|
Overall: 8.4 (5.7–12.9)
|
NR |
|
B. Second and further LoT (2L+) |
|||||
|
Baek et al. 2024 [49]
Korea
KCSG BR19-15 –retrospective, non-interventional study
2017–2018 |
Median age, years: 58
n (%)
Prior therapy, %
|
16.8 with advanced line of TD-M1 (2L+) |
35 |
6.6 (6.1–7.3)
aHR for prior pertuzumab use: 1.204 (95% CI: 0.936–1.55), p=0.149 |
Not reached
aHR for prior pertuzumab use:1.623 (95% CI: 1.022–2.578), p=0.040 |
|
Bian et al. 2023 [52]
China
Retrospective study
2013–2022 |
Median age, years: 47 |
NR |
|
|
NR |
|
Blotta et al. 2023 [53]
Brazil
Retrospective, medical records
2013–2023 |
N=73, Pts with T-DM1 (1L, 2L, 3L/3L+)
Median age, years: 57 |
NR |
82.6 |
12.7 |
53.9 |
|
Hsieh et al. 2023 [62]
Taiwan
Retrospective cohort study
2013–2022 |
N=32 (any LoT) Prior pertuzumab + trastuzumab, n: 12
Median age, years: 60 |
NR |
NR |
NR |
|
|
Ji et al. 2023 [63]
China
Real-world retrospectively study
2013–2022 |
N=144; Prior TKI, trastuzumab
Subgroup analysis
Median age, years
|
NR |
Objective response rate:
|
For all pts-
|
NR |
|
Martins et al. 2023 [67]
Portugal
Retrospective study
2017–2023 |
N=32; Prior Pertuzumab/trastuzumab plus taxane
Median age, years: 53
|
NR |
Objective response rate: 21.9 (9.3–40.0) |
4.4 (1.54–7.26) |
17.3 (10.6–24.54) |
|
Drago et al. 2022 [57]
USA
Retrospective study |
Median age, years
|
NR |
NR |
|
NR |
|
Gogia et al. 2022 [59]
India
Retrospective study
2015–2021 |
N=80; 1L anti HER2 therapy or relapse within 6 months of adjuvant trastuzumab
Median age, years: 49
ECOG PS 0-1: 80%
Prior therapy, %
T-DM1 line, n 2L: 80 (100%) |
18 |
80 |
10 |
27 |
|
Li et al. 2021 [64]
China
Real-world study
2013–2019 |
N=105; prior trastuzumab and lapatinib (any LoT)
Median age, years
|
NR |
Objective response rate, n (%)
|
Overall
|
NR |
|
Hardy-Werbin et al. 2019 [60]
Spain
Retrospective
2012–2016 |
N=15; Prior trastuzumab
Median age, years: 48
N (%)
|
NR |
Objective response rate: 73.3 |
10 (3.47–16.52) |
34 (16.96–51.04) |
|
Yeo et al. 2018 [79]
Hong Kong
Retrospective study
2013–2015 |
N=37; Prior anti-HER2 (trastuzumab, lapatinib or pertuzumab), CT or endocrine therapy
Median age, years: 52
N (%)
|
|
29.7 |
6.0 (3.3–9.8) |
Not reached |
|
Abbreviations: 1L: First-line; 2L: Second-line; 3L: Third-line; 4L: Fourth line; 5L: Fifth line; ADC: Antibody-Drug Conjugate; aHR: Adjusted Hazard Ratio; CI: Confidence Interval; CT: Chemotherapy; HER2: Human Epidermal Growth Factor Receptor 2; HR: Hazard Ratio; IQR: Interquartile Range; LoT: Line of Therapy; mBC: Metastatic Breast Cancer; mOS: Median Overall Survival; mPFS: Median Progression-Free Survival; NR: Not Reported; ORR: Overall Response Rate; OS: Overall Survival; PFS: Progression Free Survival; pts: Patients; T-DM1: Trastuzumab Emtansine; T-Dxd: Trastuzumab Deruxtecan; TKI: Tyrosine Kinase Inhibitor; UK: United Kingdom; US: United States |
|||||
Efficacy with or without brain/central nervous system metastasis
HER2-positive disease may progress and metastasize to the brain/central nervous system (CNS), which may impact treatment efficacy. In fact, evidence from trials that included patients with HER2-positive mBC with and without brain metastasis (BM) suggest mPFS and mOS are longer in the latter patient subgroup (Table 5) [26,27,33]. In patients with BM/CNS metastasis, mPFS and mOS ranged from 3.0–6.9 months and 18.9–26.8 months, respectively [26,27,31,33,43], which increased to 7.1 months and 30.0 months, respectively, in patients without BM [27,33]. Considering other 2L therapeutic options, T-DM1 was associated with prolonged mOS compared with capecitabine–lapatinib (26.8 vs. 12.9 months) although mPFS was similar (5.9 vs. 5.7 months) [31], while T-DXd yielded a better 1-year PFS rate of 72.0% (vs. 20.9% for T-DM1) [27]. Additionally, among patients with BM treated with T-DM1 (n=39), mPFS was longer in those without prior therapy for BM (5.6 months, n=19) compared to those with prior therapy for BMs (radiotherapy alone, surgery alone or both) (2.8 months, n=20) [27].
Similarly, evidence from six real-world studies also indicate a better mPFS and mOS in response to T-DM1 in patients without BM, although patients with BM also benefited from T-DM1 (Table 5) [44,45,60,61,71,75,77]. In patients with and without BM/CNS metastasis, mPFS ranged from 6–7 months and 8–13 months, respectively [44,45,60]; and mOS was 14–not reached and 32 months, respectively [44,60].
|
Study (Primary/Secondary Publication) |
Study details (Country, Study name, design and duration) |
Population; N (%); eligibility criteria |
Median follow-up (months) |
ORR, % (95% CI) |
mPFS, months (95% CI) |
mOS, months (95% CI) |
|
A. Clinical studies |
||||||
|
I. DESTINY-Breast03 (two publications) |
||||||
|
Hurvitz et al. 2024 [27] (Secondary)
|
Global
DESTINY-Breast03 (NCT03529110)
Randomized, open-label, multi-centre, phase 3 |
N=524; prior anti-HER2 (trastuzumab and pertuzumab)
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
|
NR |
T-Dxd
T-DM1
|
Patients with BM at baseline
Patients without BM at baseline
Patients with prior BM therapy
Patients without prior BM therapy
|
NR |
|
|
|
|
|
NR |
NR |
Patients with BM
HR: 0·54 (95% CI, 0.29–1.03)
Patients without BM
HR: 0.66 (95% CI, 0.47–0.94) |
|
II. Other trials |
||||||
|
Fabi et al. 2017 [45] (Primary)
Fabi et al. 2018 [44] (Secondary)
|
Italy
FIELD-PRACTICE – multi-centre study
2012–2016 |
N=303; prior brain loco-regional treatment
Median age, years: 51
|
16 (1–55) |
BM: 35.1 Non-BM: 38.3 |
Overall population
2 LoT
3 LoT
4 LoT
|
Overall population
2 LoT
3 LoT
|
|
Montemurro et al. 2020 [33] (Secondary) |
Global
KAMILLA (NCT01702571)
Single-arm, ongoing, phase 3b |
N=2002; prior taxane, Trastuzumab, pertuzumab, Lapatinib, Endocrine therapy, Anthracyclines
Median age, years
ECOG PS 0
ECOG PS 1
ECOG PS 2
|
20.6 |
Patients with measurable BM at baseline (n=126) BOR: 21.4 (14.6–29.6) |
|
|
|
Krop et al. 2015 [31] (Secondary)
|
Global
EMILIA (NCT00829166)
Randomized, open-label, multi-centre, phase 3 |
N=991; prior taxane + trastuzumab stable
CNS metastasis at baseline
Median age, years
n (%) ECOG PS 0
ECOG PS 1
|
|
NR |
Stratified HR: 1.000 (95% CI, 0.542−1.844); p=0.9998
|
Stratified HR=0.696 (95% CI, 0.328−1.476); p=0.3378
|
|
Yardley et al. 2015 [43] (Primary) |
USA
T-PAS (TDM4884g / NCT01120561)
Single-arm, multi-centre, expanded access |
N=215; ≤2 prior HER2-directed therapy
Patients with BM, n: 26 (9.4)
Median age, years: 53
n (%)
|
5.9 |
Measurable, N=22 27.3 (10.7–50.2) |
6.9 (2.7–12.3) |
NR |
|
B. Real-world studies |
||||||
|
He et al. 2024 [61] (Primary)
|
China
Multicentric, observational
March 2023–June 2023 |
N=101; prior trastuzumab and taxane
Median age, years: 51
n (%)
|
7 |
NR |
BM: 6.1 (5.9–6.3) Non-BM: 4 |
NR |
|
Sanglier et al. 2023 [75] (Primary)
|
USA
Retrospective, observational study
Jan 1, 2011–Dec 31, 2020 |
N=214; no prior T-DM1 or LC treatment BM cohort treated with
Before weighting Median age, years
ECOG PS 0
ECOG PS 1
ECOG PS ≥2
ECOG PS missing
After weighting Median age, years
ECOG PS 0
ECOG PS 1
ECOG PS ≥2
ECOG PS missing
|
Prior to weighting: 30.7 After weighting: 28.8 |
NR |
Prior to weighting
HR: 0.57 (95% CI 0.42–0.78); p<0.001
After weighting
HR: 0.50 (95% CI 0.36–0.69); p<0.001 |
Prior to weighting
HR: 0.69 (95% CI 0.46–1.03), p=0.046
After weighting
HR: 0.55 (95% CI 0.34–0.89); p=0.013) |
|
Moinard-Butot et al. 2022 [71] (Primary)
|
France
Retrospective, 18 French Comprehensive Cancer Centre’s
2008–2016 |
Cohort A treated with T-DM1 N=233; taxane-based CT and dual HER2-blockage by trastuzumab + pertuzumab
Median age:
|
20.8 (0–61) |
NR |
NR |
Cohort A
|
|
Tataroglu Ozyukseler et al. 2021 [77] (Primary)
|
Turkey
Single-centre retrospective, observational study
2016–2019 |
N=78; ≥1 trastuzumab-based regimens
Median age, years: 52
%
|
12.9 (7.4–19.8) |
NR |
|
|
|
Hardy-Werbin et al. 2019 [60] (Primary) |
Spain
Retrospective
2012–2016 |
N=15; prior trastuzumab and taxane
Median age, years: 48
n (%)
|
NR |
80.0 |
BM: 6.0 (3.85–8.14) Non-BM: 13 (8.6–17.93) |
Not reached |
|
Abbreviations: BM: Brain Metastasis; BOR: Best Overall Response; CI: Confidence Interval; CNS: Central Nervous System; CT: Chemotherapy; HER2: Human Epidermal Growth Factor Receptor 2; HR: Hazard Ratio; ITT: Intent-to-Treat; LC: Lapatinib plus Capecitabine; LoT: Line of Therapy; mOS: Median Overall Survival; mPFS: Median Progression-Free Survival; NA: Not Applicable; NE: Not Evaluable; NR: Not Reported; ORR: Overall Response Rate; PFS: Progression Free Survival; T-DM1: Trastuzumab Emtansine; T-Dxd: Trastuzumab Deruxtecan; US: United States. |
||||||
Safety
Clinical trials
The pivotal phase 3 trials (DESTINY-Breast03, ELAINA, KAMILLA, EMILIA and TH3RESA), phase 2 studies, and the expanded-access study demonstrated a consistent T-DM1 safety profile in terms of types of grades 3 or worse AEs (Table 6). Thrombocytopenia emerged as one of the most common grade ≥3 AEs, occurring in 2.3%–10.2% of patients in most of the included trials [33,34,36,38,39,42,43]. Three trials reported higher grade ≥3 AE rates (14.0–29.7%) [26,29,41], while a notably higher (40.4%) incidence of thrombocytopenia was observed in the ELAINA trial [40].
|
Study (Primary/Secondary Publication) |
Study details (Country, Study name, design and duration) |
Population; N; eligibility criteria |
Median follow-up (months) |
Grade 3 AEs, n (%) |
Grade 4 AEs, n (%) |
Grade ≥3 AEs, n (%) |
|
A. Breast-03 Trial |
||||||
|
Cortés et al. 2022 [80] (Primary) |
Global
DESTINY-Breast03 (NCT03529110)
Randomized, open-label, multi-centre, active-controlled, phase 3 |
N=524; prior taxane + trastuzumab
T-DM1, n: 263 prior anti-HER2 therapy
Patient subgroups
T-DXd, n: 261 prior anti-HER2 therapy
Patient subgroups
Recurrent: 168
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
|
|
NR |
NR |
T-DXd (N = 257) vs T-DM1 (N = 261)
Haematologic toxicities
Non-haematologic toxicities
Grade ≥ 3 TEAEs leading to discontinuation: 13.6% vs 7.3% |
|
Hurvitz et al. 2023 [26] (Secondary) |
|
|
T-DM1: 26.5 |
T-Dxd (N=257) vs. T-DM1 (N=261)
Hematologic toxicities
Non-hematologic toxicities
Weight decreased: 6 (2) vs. 2 (<1) |
T-Dxd (N=257) vs. T-DM1 (N=261)
Hematologic toxicities Platelet count decreased: 2 (<1) vs. 7 (3)
Non-hematologic toxicities Neutrophil count decreased: 3 (1) vs. 0
|
T-Dxd (N=257) vs. T-DM1 (N=261)
Hematologic toxicities
Non-hematologic toxicities
Grade ≥3 TEAEs
Drug related Grade ≥3 TEAEs: 121 (47) vs. 110 (42) |
|
Im et al. 2022 [28] (Secondary) |
Asia |
Asians, n: 309; prior taxane + trastuzumab
|
NR |
NR |
No grade 4/5 drug-related ILD/pneumonitis reported |
NR |
|
B. EMILIA Trial |
||||||
|
Verma et al. 2012 [32] (Primary)
|
Global
EMILIA (NCT00829166)
Randomized, open-label, multi-centre, phase 3 |
N=991; prior taxane + trastuzumab
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
ECOG NA, n (%)
|
13 |
T-DM1: Left ventricular systolic dysfunction, n: 1 |
T-DM1: Gastrointestinal hemorrhage, n: 1 |
T-DM1 vs. Lapatinib plus capecitabine
Hematologic toxicities
Non-hematologic toxicities
Elevated AST: 21 (4.3) vs 4 (0.8) |
|
Diéras et al. 2017 [29] (Secondary) |
|
|
Lapatinib plus capecitabine: 41.9 (IQR 34.6–50.7) |
T-DM1 vs. Lapatinib plus capecitabine Hematologic toxicities
Non-Hematologic toxicities
Urticaria: 0 vs. 5 (1) |
T-DM1 vs. Lapatinib plus capecitabine Hematologic toxicities
Non-Hematologic toxicities
Pulmonary embolism: 0 vs. 6 (1.2) |
Grade 3 or worse AE
Hematologic toxicities
Non-Hematologic toxicities
|
|
Krop et al. 2015 [31] (Secondary) |
|
CNS metastasis at baseline; prior taxane + trastuzumab stable
Median age, years
n (%) ECOG PS 0
ECOG PS 1
Lapatinib plus capecitabine: 25 (51.0) |
For ITT population
Lapatinib plus capecitabine: 18.6 |
NR |
NR |
Patients with CNS metastases, n/N (%)
Lapatinib plus capecitabine: 31/49 (63.3%) |
|
Im et al. 2021 [30] (Secondary) |
Asia |
Asia N=158; Asians with prior taxane + trastuzumab Enrolled
Safety Evaluable
|
13 |
Hematologic toxicities (T-DM1 vs. Lapatinib plus capecitabine)
Asia
Global
|
Hematologic toxicities (T-DM1 vs. Lapatinib plus capecitabine)
Asia
Global
|
T-DM1 vs. Lapatinib plus capecitabine
|
|
C. KAMILLA Trial |
||||||
|
Montemurro et al. 2020 [33] (Primary) |
Global
KAMILLA (NCT01702571)
Single-arm, On-going, phase 3b |
N=2,002; prior taxane, Trastuzumab, pertuzumab, Lapatinib, Endocrine therapy, Anthracyclines
Median age, years
ECOG PS 0
ECOG PS 1
ECOG PS 2
|
20.6 |
BM at baseline Hematologic toxicities
Non-hematologic toxicities
No BM at baseline
Non-hematologic toxicities
|
None |
None |
|
Wuerstlein et al. 2022 [34] (Secondary) |
Global and Asia |
N=2,183; prior anti-HER2 + CT
Median age, years
ECOG PS 0, n (%)
ECOG PS 1, n (%)
ECOG PS 2, n (%)
ECOG PS missing, n (%)
|
|
Cohort 1 vs Cohort 2
Non-hematologic toxicities
Hematologic toxicities
|
Cohort 1 vs Cohort 2
Hematologic toxicities
|
Cohort 1 vs Cohort 2
Hematologic toxicities
Non-hematologic toxicities
Hematologic toxicities
|
|
D. Other Trials |
||||||
|
Wang et al. 2023 [40] (Primary) |
China
ELAINA (NCT03084939)
Randomized, open-label bridging, multi-centre, phase 3 |
N=200; prior taxane + trastuzumab
Median age, years
ECOG 0, n (%)
ECOG 1, n (%)
|
|
NR |
NR |
T-DM1 vs. Lapatinib plus capecitabine
Hematologic toxicities
Non-hematologic toxicities
|
|
Wildiers et al. 2022 [42] (Primary)
|
Europe
EORTC 75111-10114 ETF/BCG
Randomized, open-label, multi-centre, investigator-initiated, phase 2 |
N=40; prior trastuzumab + pertuzumab (TP) without or with metronomic oral cyclophosphamide (TPM)
Median age, years
WHO PS 0, n (%)
WHO PS 1, n (%)
WHO PS 2, n (%)
WHO PS 3, n (%)
|
33.7 |
NR |
NR |
Grade 3-5 (T-DM1)
All AEs: 18 (45)
Hematologic toxicities
Non-hematologic toxicities
Gastrointestinal
General disorders
Respiratory
Liver and kidney function
Special interest
|
|
Krop et al. 2017 [38] (Primary) |
Global
TH3RESA (NCT01419197)
Randomized, open-label, multi-centre, phase 3 |
N=602; ≤2 prior HER2-directed therapy (trastuzumab, lapatinib and taxane) Safety population
Median age, years
ECOG PS 0, n (%)
ECOG PS 1, n (%)
ECOG PS 2, n (%)
|
30.5 |
T-DM1 vs. Physician’s choice Hematologic toxicities
Non-Hematologic toxicities
|
T-DM1 vs. Physician’s choice Hematologic toxicities
|
Grade 3 or worse AE Hematologic toxicities
|
|
Watanabe et al. 2017 [41] (Primary)
|
Japan
JO29317
Single-arm, multi-centre, phase 2 |
N=232; prior CT and trastuzumab
Median age, years: 57
n (%)
|
NR |
Hematologic toxicities Thrombocytopenia: 60 (25.9)
Non-Hematologic toxicities Hepatotoxicity: 25 (10.8) |
Hematologic toxicities Thrombocytopenia: 9 (3.9) |
Grade ≥3: 109 (47.0)
Grade ≥3 AEs reported >2% of pts Hematologic toxicities
Non-Hematologic toxicities
|
|
Yardley et al. 2015 [43] (Primary) |
USA
T-PAS (TDM4884g / NCT01120561)
Single-arm, multi-centre, expanded access |
N=215; ≤2 prior HER2-directed therapy including trastuzumab and lapatinib + anthracycline and a taxane, and capecitabine or 5-fluorouracil
Median age, years: 53
n (%)
|
5.9 |
Hematologic toxicities Thrombocytopenia: 11 (5.1)
Non-Hematologic toxicities Peripheral neuropathy: 5 (2.3) |
Hematologic toxicities Thrombocytopenia: 6 (2.8) |
Overall, hemorrhagic event of grade 3 or greater: 4 (1.86)
Overall, Common TEAEs Grade ≥3 AEs occurring in >2% of patients Hematologic toxicities
Non-Hematologic toxicities
Patients Having Received ≤18 Cycles of T-DM1 vs. >18 Cycles of T-DM1
Grade ≥3 AE occurring in >1 patient Hematologic toxicities
|
|
Gupta et al. 2013 [36] (Primary) |
NR
TDM4688g / NCT00943670
Single-arm, multi-centre, phase 2 |
N=51; prior CT (including anthracycline, taxane [paclitaxel/docetaxel] and capecitabine) + trastuzumab + lapatinib
Median age, years: 53.5
n (%)
|
NR |
Hematologic toxicities T-DM1
Non-Hematologic toxicities T-DM1
Pertuzumab plus T-DM1
|
T-DM1 vs. Pertuzumab plus T-DM1: 0 vs. 3 (15) |
T-DM1 vs. Pertuzumab plus T-DM1: 17 (33.3) vs. 12 (60.0) |
|
Krop et al. 2012 [39] (Primary)
|
NR
TDM437g
Single-arm, phase 2 |
N=110; ≤2 prior HER2-directed therapy
Median age, years: 52.5
n (%)
|
17.4 |
Hematologic toxicities
Non-Hematologic toxicities
|
At least one grade 4 AE: 6 (5.45) |
Hematologic toxicities
Non-Hematologic toxicities
|
|
Burris et al. 2011 [35] (Primary)
|
USA
TDM4258g
Single-arm, phase 2 |
N=112; ≥1 prior HER2-targeted therapy
Median age, years: 54.5
n (%)
|
≥12 |
Hematologic toxicities Anemia: 3 (2.7)
Non-Hematologic toxicities
|
None |
NR |
|
Abbreviations: AEs: Adverse Events; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; BM: Brain Metastasis; CT: Chemotherapy; HER2: Human Epidermal Growth Factor Receptor 2; ILD: Interstitial Lung Disease; IQR: Interquartile Range; LVEF: Left Ventricular Ejection Fraction; MedDRA: Medical Dictionary for Regulatory Activities; NR: Not Reported; Pts: Patients; SAEs: Serious Adverse Events; T-DM1: Trastuzumab Emtansine; T-Dxd: Trastuzumab Deruxtecan; TEAEs: Treatment-Emergent Adverse Events; TKI: Tyrosine Kinase Inhibitor; TP: Trastuzumab plus Pertuzumab; TPM: Trastuzumab plus Pertuzumab and Metronomic Oral Cyclophosphamide; UTI: Urinary Tract Infection; WBC: White Blood Cell |
||||||
Liver toxicity/dysfunction was reported in 5.0–11.2% of patients in four studies [26,36,40,41], although lower incidences (≤5%) were observed in other studies [29,33,38,39,42,43]. Anemia in 2.7% to 7.0% of patients [26,29,33,35,38,43], grade 3 or worse anorexia (5%) [42], hypokalemia (8.9%) [35] and lymphopenia (15%) [42] were also reported. The incidence of other grade 3/4 AEs including neutropenia, fatigue, liver toxicity/dysfunction, vomiting, diarrhea, dyspnea, epistaxis, asthenia, hyperglycemia, pneumonia, headache, cellulitis and cardiac toxicity was consistently below <5% [26,29,33–36,38,39,42,43]. Other grade AEs reported among clinical trials are presented as supplemental Table S3.
AEs most commonly led to dose reductions (≈5–30% in most studies; up to ~60% in a large phase IIIB safety cohort) while permanent treatment discontinuation (≈2–15% in most studies, and up to 25% in a small pertuzumab+T-DM1 cohort) and protocol-defined dose delays/interruptions were also reported [77–82]. In the updated DESTINY-Breast03 trial, treatment modifications occurred more often with T-DXd than with T-DM1, including drug-related discontinuation (20% vs 7%), dose reduction (25% vs 15%), and interruption (42% vs 17%), with pneumonitis/interstitial lung disease (ILD) among the most common causes of discontinuation on T-DXd [75]. In the DESTINY-Breast03 brain metastases subgroup (data cut-off 21 May 2021), overall treatment discontinuation was 48.6% with T-DXd and 82.0% with T-DM1 [76].
Real-world studies
Consistent with trial data, several real-world studies identified thrombocytopenia, liver toxicity/dysfunction, neutropenia, anemia, fatigue, and nausea as the most frequently reported grade 3 or worse AEs (Table 7).
|
Study details (Country, Study name, design, data source and study duration) |
Population; N; eligibility criteria |
Median follow-up (months) |
Grade 3 AEs, n (%) |
Grade 4 AEs, n (%) |
Grade ≥3 AEs, n (%) |
|
FIELD-PRACTICE study (two publications) |
|||||
|
Fabi et al. 2017 [45]
Italy
FIELD-PRACTICE – multi-centre study
2012–2016 |
N=303; Prior taxane / anthracycline based therapies
Median age, years: 51
|
10 |
Hematologic toxicities
Non-hematologic toxicities
|
Hematologic toxicities
Non-hematologic toxicities Transaminase: 1 (0.3) |
NR |
|
Fabi et al. 2017 [46]
|
N=107; 1L taxane + pertuzumab / trastuzumab pertuzumab / + trastuzumab, n: 34 trastuzumab, n: 73
Median age, years
|
NR |
T-DM1 post Pertuzumab / +trastuzumab vs. Trastuzumab
Hematologic toxicities Thrombocytopenia: 0 vs. 1 (1)
Non-hematologic toxicities Pertuzumab / +trastuzumab vs. Trastuzumab
Transaminases: 0 vs. 2 (3) Alopecia: 1 (3) vs. 1 (1) Asthenia: 0 vs. 2 (3) |
T-DM1 post Pertuzumab / +trastuzumab vs. Trastuzumab
Hematologic toxicities Thrombocytopenia: 0 vs. 1 (1) |
NR |
|
Real-world studies with single publication |
|||||
|
Baek et al. 2024 [49]
Korea
KCSG BR19-15, retrospective, non-interventional study
2017–2018 |
HER2-positive MBC treated with T-DM1, N=824; Prior pertuzumab CNS, n (%): 145 (17.6)
Median age, years: 58
n (%)
|
16.8 |
NR |
NR |
Grade 3 or 4 Hematologic toxicities
Non-hematologic toxicities
|
|
He et al. 2024 [61]
China
Multicentric, observational
March 2023–June 2023 |
N=101; Prior trastuzumab and pertuzumab
Median age, years: 51
n (%)
|
7 |
NR |
NR |
Grade 3-4 Early vs. Advanced stage Hematologic toxicities
|
|
Bian et al. 2023 [52]
China
Retrospective study
2013–2022 |
N=213; Prior TKI therapy
Median age, years: 47 |
NR |
NR |
NR |
Grade 3 or 4 hemaatologic toxicities ADC group
T-DM1 group
Non-hematologic toxicities ADC vs. T-DM1
|
|
Ji et al. 2023 [63]
China
Real-world retrospective study
2013–2022 |
N=144; TKI, Trastuzumab
Median age, years
|
NR |
NR |
NR |
Grade 3-4 T-DM1 vs. Novel anti-HER2 ADCs
Hematologic toxicities
Non-hematologic toxicities
|
|
Martins et al. 2023 [67]
Portugal
Retrospective study
2017–2023 |
N=32; 1L Pertuzumab / trastuzumab plus taxane
Median age, years: 53
|
NR |
NR |
Non-hematologic toxicities Hepatic and cardiac toxicity: 3 (9.3) |
No grade 5 AE |
|
Gogia et al. 2022 [59]
India
Retrospective study
2015–2021 |
N=80; Prior trastuzumab, pertuzumab
Median age, years: 49
ECOG PS 0-1: 80%
|
18 |
NR |
NR |
Grade 3/4 (overall): 20 (25) Hematologic toxicities
Non-hematologic toxicities
|
|
Bahceci et al. 2021 [50]
Turkey
Retrospective multi-centre study |
N=414; Prior CT and Anti-HER2
Median age, years: 47
|
|
NR |
NR |
Hematologic toxicities
Non-hematologic toxicities
|
|
Li et al. 2021 [64]
China
Real-world study
2013–2019 |
N=105; Prior trastuzumab and lapatinib
Median age, years
|
NR |
NR |
NR |
Hematologic toxicities
Non-Hematological toxicities
|
|
Tataroglu Ozyukseler et al. 2021 [77]
Turkey
Single-centre retrospective, observational study
2016–2019 |
N=78; 1L trastuzumab-based regimens
Median age, years: 52
%
|
12.9 |
Hematologic Toxicities
Non-hematologic toxicities
|
Hematologic Toxicities Thrombocytopenia: 4 (5.1)
Non-hematologic toxicities Increased AST and/or ALT: 2 (2.6) |
NR |
|
Battisti et al. 2020 [51]
UK
Retrospective review
2014–2018 |
Median age, years: 55
n (%)
|
NR |
Hematologic Toxicities
Non-hematologic toxicities
|
Hematologic Toxicities Thrombocytopenia: 1 (0.8)
|
Grade ≥3 (overall): 46 (35.9)
|
|
del Prete et al. 2020 [56]
Italy
Multi-centre retrospective study |
N=135; 1L taxane + pertuzumab/trastuzumab
Median age, years: 56 |
10.3 |
Hematologic toxicities Neutropenia: 1 (0.7) |
None |
NR |
|
Martinez-Garcia et al. 2020 [66]
Spain
TDM1RM study, retrospective observational study
2015–2020 |
N=52; 1L taxane + pertuzumab / trastuzumab
Median age, years: 52
%
|
33 |
Hematologic toxicities
Non-hematologic toxicities
|
NR |
NR |
|
Yeo et al. 2018 [79]
Hong Kong
Retrospective study
2013–2015 |
N=37; 2L+ prior anti-HER2 (trastuzumab / lapatinib / pertuzumab) / CT / endocrine therapy
Median age, years: 52
N (%)
|
15.6 |
Hematologic toxicities
Non-hematologic toxicities
|
Hematologic toxicities Thrombocytopenia: 2 (5.4) |
None |
|
Vici et al. 2017 [78]
Italy
Retrospective observational study
2013–2016 |
N=250; 1L pertuzumab
Median age, years: 56
%
|
15 |
Hematologic toxicities
Non-hematologic toxicities
|
NR |
NR |
|
Michel et al. 2015 [68]
Germany
Retrospective chart review
2010–2014 |
N=23, prior HER2-directed therapy (trastuzumab, lapatinib, pertuzumab), (neo-) adjuvant cytotoxic therapy
Median age, years: 54.6
%
|
9.3 |
SAEs
Hematologic toxicities
Non-hematologic toxicities
|
SAEs
Hematologic toxicities Elevated liver enzyme: 1 (4.3) |
No grade 5 toxicity |
|
Abbreviations: 1L: First-line; 2L: Second-line; 3L: Third-line; 4L: Fourth line; ADC: Antibody-Drug Conjugate; AEs: Adverse Events; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; BM: Brain Metastasis; CNS: Central Nervous System; CT: Chemotherapy; GGT: Gamma-Glutamyl Transferase; HER2: Human Epidermal Growth Factor Receptor 2; ILD: Interstitial Lung Disease; LVEF: Left Ventricular Ejection Fraction; mBC: Metastatic Breast Cancer; NR: Not Reported; Pts: Patients; SAEs: Serious Adverse Events; SS: Sum of Squares; T-DM1: Trastuzumab Emtansine; TEAEs: Treatment-Emergent Adverse Events; TKI: Tyrosine Kinase Inhibitor; UK: United Kingdom; US: United States; UTI: Urinary Tract Infection; vs: Versus |
|||||
Almost half of the included real-world publications reported incidences of grade 3 or worse thrombocytopenia [45,46,49–52,59,61,63,64,66,68,77–79], with considerable variation ranging between 1.2–29.0%. Grade 3/4 neutropenia and anemia were less frequent compared to thrombocytopenia; about 25% of the included real-world studies highlighted low immune cells/infection and low red blood cell counts as grade 3/4 AEs [45,49–51,56,59,61,63,64,66,77,79]. Of note, the frequency of neutropenia across studies ranged between 0.7–6.5% [45,49–51,56,61,63,64,66] and between 1.2–6.2% for anemia [49–51,59,61,63,66,77,79]. Four studies [50,61,63,77] reported leukopenia (0.4-6.5%) and one study [68] reported grade 3/4 infection (4.3%).
Grade 3/4 liver toxicity or dysfunction was the most frequently reported grade ≥3 non-hematological toxicity, reported in one third of studies [45,46,49–51,59,63,64,67,68,77–79]. Grade 3/4 elevations in liver enzymes including transaminases, aminotransferase, and gamma-glutamyl transferase along with bilirubin, alanine, and aspartate were also described, with incidence ranging between 0.4–12.5% [45,46,49,50,59,63,64,68,77–79]. Notably, one study reported that 19.5% of patients experienced liver toxicity [51]. Other non-hematological grade 3 or worse AEs included fatigue and nausea. The frequency of fatigue was less frequent (1.3–2.8%) in six studies [49–51,63,77,78], was 8.0%–12.5% in two other studies [59,64]; and one study reported fatigue in 21.7% of patients [68]. The incidence of nausea was observed to be between 0.6–6% as described in five studies [49,51,64,77,78]. Other less common grade 3/4 AEs (<5%) included diarrhea [49–51,77], alopecia [45,46], peripheral neuropathy [51,59], hypokalemia [50,79], asthenia [45,66], and mucositis [51]. Additionally, three studies linked minimal cardiotoxicity with T-DM1 treatment [47,50,67]. Other grade AEs reported in real-world studies are presented as supplemental Table S4.
Three studies reported T-DM1 discontinuation, primarily due to disease progression or AEs. Dzimitrowicz et al. (2016) [58] reported disease progression as predominant cause of treatment discontinuation (84.3%, 59/70 of cases), while 10% discontinued due to toxicity or poor tolerance. Similarly, Silva et al. (2023) [76] reported T-DM1 discontinuation majorly due to disease progression (90%, 36/40), specifically progression in bone, brain, and liver, and the remaining due to toxic side effects (10%, 4/40). Blotta et al. (2023) [53] reported T-DM1 discontinuation majorly due to AEs that included peripheral neuropathy (40%), pneumonitis (20%), and hepatic pseudocirrhosis (20%) (data not shown in the table).
Corroboration of clinical trial findings with real-world outcomes
Across pivotal trials and heterogeneous RWE cohorts, T-DM1 demonstrated concordant effectiveness and tolerability in HER2-positive mBC, supporting the corroboration of findings from clinical trials and routine clinical practice. ORR were comparable, with trials reporting ORRs of ~29–50% (29.3–29.6% in KAMILLA [34]; 34–35% in DESTINY-Breast03 [26,80]; 43.6–47.8% in EMILIA [29,32]; 50.4% in ELAINA [40]). In routine clinical practice, ORRs ranged from ~20–64%, including 20.7–44.5% in multiple post-pertuzumab/second-line datasets [51,54,56,66,78,79]. Similarly, mPFS ranged 6.8–9.6 months in key trials (6.8 months in Breast03 [26,80] and KAMILLA [34]; 7.0 months in ELAINA [40]; 9.6/9.3 months in EMILIA [30,32]) which was consistent with RWE estimates of ~6.0–10.5 months in similar settings [51,54,56,66,74,78,79]. The mOS in trials was ~30 months (29.9–34.3 months in EMILIA [29,30,32]; 27.2–29.5 months in KAMILLA [34]; 33.2 months in ELAINA [40]) while RWE cohorts reported a broad but overlapping range (~17 months to not reached), with several studies reported outcomes within the ~23–36+ month range [51,54,55,59,60,66,67,71,78,79]. For subgroups, brain/CNS metastases were consistently prognostic (shorter outcomes than non-CNS cohorts) while T-DM1 demonstrated measurable activity in the clinical trials and RWE [27,31,33,44,71,75,77].
Safety concordance was strong, with grade ≥3 thrombocytopenia and hepatic laboratory abnormalities as leading toxicities in trials [32,34,80] as well as RWE [49,52,59,63].
Quality assessment
A total of 42 references were assessed for RoB using Cochrane tools. Of these, seven randomized trials were assessed using RoB2.0 and 35 non-randomized trials were assessed using ROBINS-I.
Cochrane risk of bias (RoB 2.0) assessment for randomized studies
A total of seven publications [26,27,29,31,32,38,40] were assessed using the RoB 2.0 tool. Of these, five publications [26,27,29,38,40] were classified as high RoB, primarily due to shortcomings in the randomization process (domain 1) and outcome measurement (domain 4). Krop et al. (2015) [31] was judged to have some concerns due to uncertainties in randomization procedures, while Verma et al. (2012) [32] was the sole study with low RoB across all domains (Figure S1). The detailed assessments of each RCTs along with the checklist are provided as supplemental information – Appendix A.
Cochrane risk of bias in non-randomized studies of interventions (ROBINS-I)
A total of 35 publications (8 single-arm clinical and 27 real-world studies) were assessed using the ROBINS-I tool. Of these, the majority (85.7%, 30/35) were judged to have a serious RoB, primarily due to critical limitations such as uncontrolled confounding variables, incomplete adjustment for baseline differences, or substantial missing outcome data [34–37,43–46,48–51,54–56,58,60,61,63–65,68–72,74,77–79]. Four studies (11.4%) exhibited a moderate risk [33,39,41,75], and a single study with an overall low-risk showed minimal bias across all domains [47]. Among the four moderate-risk studies [33,39,41,75], limitations were primarily due to the confounding factors (Domain 1), deviations from interventions (domain 4), or missing data (domain 5) (Figure S2). The detailed assessments of each real-world studies along with the checklist are provided as supplemental information – Appendix B.
Discussion
The present SLR suggests that the real-world efficacy and safety outcomes of T-DM1 corroborate with those observed in clinical trials. Clinical trials have demonstrated response with T-DM1 therapy in patients previously treated with trastuzumab-based combination therapy and better efficacy than lapatinib plus capecitabine. Real-world studies have shown better response and survival outcomes with T-DM1 in pertuzumab-naïve patients and with early treatment compared to heavily treated patients. The safety outcomes highlighted thrombocytopenia and hepatic dysfunction as the most frequently reported grade ≥3 AEs. Although patients with CNS involvement often experience shorter PFS and OS, T-DM1 remains beneficial in this subgroup.
T-DM1 was initially approved for the treatment of HER2-positive mBC after prior treatment with trastuzumab and taxanes (considered a preferred 1L regimen option) [3]. Since the approval of pertuzumab as 1L therapy for HER2-positive mBC, guidelines have recommended its use in combination with trastuzumab as 1L therapy for HER2-positive mBC. Prospectively designed trials evaluating T-DM1 efficacy in patients previously exposed to pertuzumab are scarce. However, real-world studies identified in this SLR reported outcomes for T-DM-1 in population pre-treated with pertuzumab.
Across randomized trials and diverse real-world cohorts, T-DM1 showed consistent antitumor activity and a predictable safety profile, indicating that pivotal results translate to clinical practice [26,29,32,34,40,45,49,51,63,78,80]. Patterns of response, disease control, and survival in trials overlapped in observational datasets despite broader eligibility, variable imaging schedules, and heterogeneous pre-treatment histories [45,51,74,78,79]. Concordant subgroup findings, especially maintained activity despite poorer prognosis of patients with CNS disease further support generalizability [27,31,33,44,71,75,77]. Where reported, discontinuations were predominantly driven by progression, with toxicity-related stops being uncommon [45,51]. The gaps observed were best explained by contemporary sequencing and later-line, more refractory use rather than loss of intrinsic efficacy [46,72,74,78].
Published clinical trial data provide evidence for the efficacy and safety of T-DM1 for the treatment of HER2-positive mBC based on data from approximately 3,500 heavily pre-treated patients (median prior LoT of ≥3). The included trials generally required patients to have progressed on ≥1 or ≥2 HER2-directed regimens, largely including trastuzumab and TKIs. Real-world studies provide complimentary data highlighting earlier use of T-DM1, i.e., as 2L or 3L therapy following progression on both trastuzumab and pertuzumab. The real-world studies therefore include broader patient populations than the clinical trials and provide a consolidated evidence base to appraise T-DM1 treatment as part of currently utilized therapeutic regimens for HER2-positive mBC, in particular as 2L treatment after 1L pertuzumab.
Considering T-DM1 efficacy, the observed mPFS from clinical trials (4.3–9.6 months) [26,32,34–37,39,40,43] and real world studies (2.8–10.5 months) [46,48,54,56,66,70–74,78] were largely consistent. However, the observed mOS from real-world studies (12.0–45.0 months) was more variable compared to observations from the clinical trials (22.7–34.3 months), potentially due to the former encompassing a heterogeneous patient population with possibly greater variability in lines of treatment [30,38,74,78]. Similarly, ORR was more variable in the real-world studies (21.9–80.0%) than clinical trials (25.0–50.4%) [40,42,45,46,48,50,59,64,67,68]. Notwithstanding the variability of clinical outcomes, the observations suggest that T-DM1 is an effective treatment in a broad patient population within the licensed indication. Furthermore, although the benefit of T-DM1 is more marked in patients who have not received pertuzumab (including trastuzumab pre-treated patients) based on real-world observations, patients may still benefit from T-DM1 at 2L [46,74] or later LoT [49,60,63,79]. Real-world studies also indicate better outcomes when T-DM1 is used in earlier LoT [45,50,69], suggesting T-DM1 should be initiated following relapse on 1L or 2L HER2-directed therapy rather than later in the treatment sequence. Further evidence from both trials and real-world studies support the use of T-DM1 in patients with BM/CNS metastasis [31,33,43–45]; The observed mPFS in real-world studies (6.0–7.0 months) [44,45,60] and clinical trials (5.5–6.9 months) [31,33,43] were consistent. However, both trial and real-world data indicate that survival outcomes were better for patients without BM [33,44,45,60]. Of note, results of the phase 3 trial of T-DXd suggest that, where available, T-DXd may be a better option in patients with BM [27].
With regards to safety of T-DM1 regimen, safety data from real-world studies [45–47,49–52,56,59,61,63,64,66–68,77–79] and clinical trials [26,28–30,32–36,38–43] were consistent in terms of types of grade 3 or worse AEs, with the most frequently reported being hematological AEs (thrombocytopenia, neutropenia, anemia) and liver toxicity/dysfunction. Other non-hematological AEs such as fatigue and nausea occurred less frequently. Thrombocytopenia emerged as a source of concern that needs attention with the incidence ranging from 2.5% to 40.4% and 1.2% to 29.0% were reported from the clinical trials [26,29,30,33,34,36,38–43] and RWE studies [45,46,49–52,59,61,63,64,66,68,77–79] respectively. A geographical variation in thrombocytopenia incidence was also noted, with considerably lower rates (2% to 8%) reported in studies conducted in the US and Europe [35,45,46,51,66,68] compared to those from Asia (12.8% to 29.0%) [49,52,59,61,63,64,77,79].The observation may reflect regional differences in patient management, such as more proactive approaches to thrombocytopenia prevention and treatment in the US and Europe.
Overall, the analyzed evidence base demonstrated the efficacy and safety of T-DM1 within its licensed indication in the real-world setting. Despite the superior efficacy demonstrated with T-DXd and other emerging therapies [81,82], the clinical role of T-DM1 remains relevant due to its continued widespread global use, driven by factors such as accessibility, affordability in low- and middle-income settings, safety profile, and patient-specific therapeutic considerations. T-DM1 retains clinical relevance in settings where newer ADCs are unavailable, inaccessible, or contraindicated.
A comparison with previously published SLRs emphasizes the novelty and relevance of this review. Martin Huertas et al. (2021) [18] exclusively reviewed observational studies on T-DM1 monotherapy, excluding clinical trials and combination therapies, limiting the breadth of patient populations and treatment contexts assessed. Similarly, Zhang et al. (2021) and Yeh et al. (2022) focused exclusively on RCTs, thus excluding valuable real-world insights and broader applicability to diverse patient scenarios [15,17]. Omarini et al. (2021) [19] combinedly analyzed both clinical trials and real-world studies and confined their analysis to patients pre-treated with trastuzumab and pertuzumab, thus narrowing their population scope relative to our comprehensive assessment of both pertuzumab-naïve and pre-treated populations. Lastly, Liu et al. (2024) [16] incorporated RCTs and non-randomized trials but broadened their focus to include early-stage and locally advanced breast cancer, reducing specificity to metastatic disease and limiting their applicability to advanced stage settings.
In contrast, our review's strength lies in its comprehensive synthesis across a broader range of study designs (both clinical trials and observational studies). Additionally, our inclusion criteria encompass a wider spectrum of patients; those with varying degrees of prior HER2-targeted therapies, brain metastasis status, and distinct regional contexts, thereby enhancing the external validity and applicability of the findings. Furthermore, unlike the earlier reviews, our SLR explicitly analyzes subgroup variations by geographic regions, providing insights into population-specific efficacy and safety outcomes, particularly regarding thrombocytopenia incidence, which revealed important regional differences.
This review has however several limitations reflecting potential shortcomings in the identified literature. The trials were largely conducted in heavily pre-treated patients, and prior treatments might not reflect current treatment landscape which limits application to current clinical practice. Additionally, there are limitations inherent to real-world studies including heterogeneity in the patient population and the fact that these studies are not powered to demonstrate differences between treatment groups. Furthermore, we kept the comparison between studies as simple due to the diverse regimens and variable LoTs used in different studies. The limited follow-up, particularly in real-world studies also meant that OS data were not reported and/or may not be mature. Absence of OS and safety data in some studies also indicate the existence of some evidence gap. Only EMILIA trial, with a median follow-up of 47.8 months, reported long-term outcomes. Furthermore, the present analysis focused mainly on efficacy and safety data, without incorporating quality-of-life outcomes, which limits a comprehensive understanding of the long-term benefits of T-DM1. Given the methodological limitations and heterogeneity across studies – including differences in design, baseline patient characteristics (e.g., presence of brain metastases), lines of therapy, and comparators – a formal meta-analysis was not conducted, which restricts the ability to draw robust conclusions. Instead, the data were summarized descriptively. A large proportion of the included studies (83.3%, 35/42) demonstrated a high to serious risk of bias, primarily due to limitations in study design and potential confounding factors. These methodological limitations may have influenced the robustness and reliability of the reported findings.
Conclusion
The findings from this review collectively demonstrate that T-DM1 can be an effective therapeutic option for the treatment of HER2-positive mBC in the 2L or later setting and has an acceptable safety profile. T-DM1 may be preferable to regimens that do not include HER2-directed therapy and should be considered as early as possible in the treatment sequence, including in patients with BM, particularly when T-DXd is not a treatment option due to cost or availability. However, further research is warranted.
Conflict of Interest
All authors declare that they have no conflicts of interest.
Acknowledgement
The authors would like to express their sincere gratitude to all colleagues who contributed to this study.
Author Contributions
All authors contributed significantly to the work. Their specific roles are outlined as follows – Conceptualization: Kunal Jain, Rajinder Chawla and Amit Kishore; Data Curation: Bivek Shah, Amit Kishore; Formal Analysis: Bivek Shah, Amit Kishore; Investigation: Bivek Shah; Methodology: Bivek Shah, Amit Kishore; Project Administration: Rajinder Chawla, Amit Kishore; Software: Bivek Shah, Amit Kishore; Supervision: Rajinder Chawla, Amit Kishore; Validation: Amit Kishore, Kunal Jain; Visualization: Bivek Shah, Amit Kishore; Writing – Original Draft: Bivek Shah, Sola Lawal, Kunal Jain, Amit Kishore; Writing – Review and Editing: Kunal Jain, Rajinder Chawla, Sola Lawal, Amit Kishore.
Supplementary Information
Supplementary Table S1. Search strategies for (A) PubMed, (B) Embase and (C) Cochrane database.
Supplementary Table S2. Eligibility criteria for identification of relevant literature.
Supplementary Table S3. Other grade AEs with T-DM1 in pivotal HER2-postive advance/metastatic breast cancer clinical trials.
Supplementary Table S4. Other grade AEs with T-DM1 in pivotal HER2-postive advance/metastatic breast cancer in real-world studies.
Supplementary Figure S1. ROB 2.0 based risk of bias assessed in Randomized studies as (A) traffic plot and (B) unweighted summary bar plot.
Supplementary Figure S2. ROBINS-I based risk of bias assessed in non-Randomized studies as (A–C) traffic plots and (D) unweighted summary bar plot.
Appendix A. Quality assessment using Cochrane ROB 2.0
Appendix B. Quality assessment using Cochrane ROBINS-I.
References
2. Gennari A, André F, Barrios CH, Cortés J, de Azambuja E, DeMichele A, et al. ESMO Guidelines Committee. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann Oncol. 2021 Dec;32(12):1475–95.
3. NCCN. NCCN Clinical Practice Guidelines in Oncology, Breast Cacner. v4 2023, March 2023, Available at https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf Accessed Sept 2023.
4. Roche GmbH. Herceptin (trastuzumab) SmPC. Available at: https://www.ema.europa.eu/en/documents/product-information/herceptin-epar-product-information_en.pdf. Accessed December 2023.
5. Roche GmbH. Perjeta (pertuzumab) SmPC. Available at: https://www.ema.europa.eu/en/documents/product-information/perjeta-epar-product-information_en.pdf. Accessed December 2023.
6. Li N, Yang L, Zhao Z, Du T, Liang G, Li N, et al. Antibody-drug conjugates in breast cancer: current evidence and future directions. Exp Hematol Oncol. 2025 Mar 20;14(1):41.
7. Roche GmbH. Kadcyla (ado-trastuzumab emtansine) SmPC Setp 2018 Available at: https://www.ema.europa.eu/en/documents/product-information/kadcyla-epar-product-information_en.pdf Accessed Sept 2023.
8. Daiichi Sankyo Europe GmbH. ENHERTU® (fam-trastuzumab deruxtecan-nxki). Prescribing information. July 2023. available at: https://www.ema.europa.eu/en/documents/product-information/enhertu-epar-product-information_en.pdf Accessed Sept 2023.
9. Novartis. Tyverb (Lapatinib) SmPC. Available at: https://www.ema.europa.eu/en/documents/product-information/tyverb-epar-product-information_en.pdf. Accessed December 2023.
10. Schettini F, Conte B, Buono G, De Placido P, Parola S, Griguolo G, et al. T-DM1 versus pertuzumab, trastuzumab and a taxane as first-line therapy of early-relapsed HER2-positive metastatic breast cancer: an Italian multicenter observational study. ESMO Open. 2021 Apr;6(2):100099.
11. Im SA, Gennari A, Park YH, Kim JH, Jiang ZF, Gupta S, et al. Pan-Asian adapted ESMO Clinical Practice Guidelines for the diagnosis, staging and treatment of patients with metastatic breast cancer. ESMO Open. 2023 Jun;8(3):101541.
12. FDA approves new treatment option for patients with HER2-positive breast cancer who have progressed on available therapies [press release]. 2019.
13. Jelovac D, Emens LA. HER2-directed therapy for metastatic breast cancer. Oncology (Williston Park). 2013 Mar;27(3):166–75.
14. Stanowicka-Grada M, Senkus E. Anti-HER2 Drugs for the Treatment of Advanced HER2 Positive Breast Cancer. Curr Treat Options Oncol. 2023 Nov;24(11):1633–50.
15. Zhang X, Leng J, Zhou Y, Mao F, Lin Y, Shen S, et al. Efficacy and Safety of Anti-HER2 Agents in Combination With Chemotherapy for Metastatic HER2-Positive Breast Cancer Patient: A Network Meta-Analysis. Front Oncol. 2021 Aug 19;11:731210.
16. Liu F, Li Y, Yang D, Tang L, Yang Q, Jiang M, et al. Meta-analysis of the clinical efficacy and safety of T-DM1 in the treatment of HER2-positive breast cancer. Indian J Cancer. 2024 Jan 1;61(1):146–55.
17. Yeh Y, Chen C, Ko Y. Effectiveness and cost-effectiveness of trastuzumab emtansine in women with HER2-positive locally advanced or metastatic breast cancer: A systematic review and meta-analysis. J Cancer Res Ther. 2022 Jul-Sep;18(4):1061–72.
18. Martin Huertas R, Fernández Abad M, Corral de la Fuente E, Serrano Domingo JJ, Martínez Jáñez N. Prolonged Responses With Trastuzumab Emtasine Treatment of Human Epidermal Growth Factor Receptor 2-positive Metastatic Breast Cancer Refractory to Trastuzumab and Pertuzumab: Systematic Review of Evidence. Clin Breast Cancer. 2021 Oct;21(5):391–8.
19. Omarini C, Piacentini F, Sperduti I, Cerma K, Barbolini M, Canino F, et al. T-DM1 efficacy in trastuzumab-pertuzumab pre-treated HER2 positive metastatic breast cancer patients: a meta-analysis. BMC Cancer. 2022 Jun 7;22(1):623.
20. Shamseer L, Moher D, Clarke M, Ghersi D, Liberati A, Petticrew M, et al; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015 Jan 2;350:g7647.
21. AccuScreener. Accessed at: https://accuscript.ai/, accessed on 23rd May 2024. Accessed May 26, 2024.
22. SymproAI. An artificial intelligence based systematic literature management and processing software. Available at: https://sympro.ai/.
23. Higgins JP, Savović J, Page MJ, Sterne JA. Revised Cochrane risk-of-bias tool for randomized trials (RoB 2). Cochrane Handbook for Systematic Reviews of Interventions. Published August 22, 2019.
24. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. bmj. 2016;355.
25. Cortés J, Im SA, Iwata H, Hamilton EP, Hurvitz SA, Egorov A, et al. "236P Subgroup analysis by disease history and prior treatments of patients (pts) with HER2-positive (HER2+) metastatic breast cancer (MBC) from DESTINY-Breast03, a randomized phase III study of trastuzumab deruxtecan (T-DXd) vs trastuzumab emtansine (T-DM1)." Annals of Oncology 33 (2022): S645–46.
26. Hurvitz SA, Hegg R, Chung WP, Im SA, Jacot W, Ganju V, et al. Trastuzumab deruxtecan versus trastuzumab emtansine in patients with HER2-positive metastatic breast cancer: updated results from DESTINY-Breast03, a randomised, open-label, phase 3 trial. Lancet. 2023 Jan 14;401(10371):105–17.
27. Hurvitz SA, Kim SB, Chung WP, Im SA, Park YH, Hegg R, et al. Trastuzumab deruxtecan versus trastuzumab emtansine in HER2-positive metastatic breast cancer patients with brain metastases from the randomized DESTINY-Breast03 trial. ESMO Open. 2024 May;9(5):102924.
28. Im S-A, Xu B, Kim S-B, Chung W-P, Park YH, Kim M-H, et al. PS2-1 Trastuzumab deruxtecan vs T-DM1 in HER2+ mBC in Asian subgroup: Results of the randomized phase 3 study DESTINY-Breast03." Annals of Oncology 33 (2022): S464–65.
29. Diéras V, Miles D, Verma S, Pegram M, Welslau M, Baselga J, et al. Trastuzumab emtansine versus capecitabine plus lapatinib in patients with previously treated HER2-positive advanced breast cancer (EMILIA): a descriptive analysis of final overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2017 Jun;18(6):732-42. doi: 10.1016/S1470-2045(17)30312-1. Epub 2017 May 16. Erratum in: Lancet Oncol. 2017 Aug;18(8):e433.
30. Im SA, Park I, Sohn JH, Im YH, Lee SC, Chang HK, et al. "284P Trastuzumab emtansine (T-DM1) in Asian patients with previously treated HER2-positive locally advanced (LA) or metastatic breast cancer (MBC): data from the phase III EMILIA study." Annals of Oncology 32 (2021): S488–89.
31. Krop IE, Lin NU, Blackwell K, Guardino E, Huober J, Lu M, et al. Trastuzumab emtansine (T-DM1) versus lapatinib plus capecitabine in patients with HER2-positive metastatic breast cancer and central nervous system metastases: a retrospective, exploratory analysis in EMILIA. Ann Oncol. 2015 Jan;26(1):113–19.
32. Verma S, Miles D, Gianni L, Krop IE, Welslau M, Baselga J, et al; EMILIA Study Group. Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med. 2012 Nov 8;367(19):1783–91.
33. Montemurro F, Delaloge S, Barrios CH, Wuerstlein R, Anton A, Brain E, et al. Trastuzumab emtansine (T-DM1) in patients with HER2-positive metastatic breast cancer and brain metastases: exploratory final analysis of cohort 1 from KAMILLA, a single-arm phase IIIb clinical trial☆. Ann Oncol. 2020 Oct;31(10):1350–58.
34. Wuerstlein R, Ellis P, Montemurro F, Antón Torres A, Delaloge S, Zhang Q, et al. Final results of the global and Asia cohorts of KAMILLA, a phase IIIB safety trial of trastuzumab emtansine in patients with HER2-positive advanced breast cancer. ESMO Open. 2022 Oct;7(5):100561.
35. Burris HA 3rd, Rugo HS, Vukelja SJ, Vogel CL, Borson RA, Limentani S, et al. Phase II study of the antibody drug conjugate trastuzumab-DM1 for the treatment of human epidermal growth factor receptor 2 (HER2)-positive breast cancer after prior HER2-directed therapy. J Clin Oncol. 2011 Feb 1;29(4):398–405.
36. Gupta M, Wang B, Carrothers TJ, LoRusso PM, Chu YW, Shih T, et al. Effects of Trastuzumab Emtansine (T-DM1) on QT Interval and Safety of Pertuzumab Plus T-DM1 in Patients With Previously Treated Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer. Clin Pharmacol Drug Dev. 2013 Jan;2(1):11–24.
37. Kashiwaba M, Ito Y, Takao S, Doihara H, Rai Y, Kanatani K, et al. A multicenter Phase II study evaluating the efficacy, safety and pharmacokinetics of trastuzumab emtansine in Japanese patients with heavily pretreated HER2-positive locally recurrent or metastatic breast cancer. Jpn J Clin Oncol. 2016 May;46(5):407–14.
38. Krop IE, Kim SB, Martin AG, LoRusso PM, Ferrero JM, Badovinac-Crnjevic T, et al. Trastuzumab emtansine versus treatment of physician's choice in patients with previously treated HER2-positive metastatic breast cancer (TH3RESA): final overall survival results from a randomised open-label phase 3 trial. Lancet Oncol. 2017 Jun;18(6):743–54.
39. Krop IE, LoRusso P, Miller KD, Modi S, Yardley D, Rodriguez G, et al. A phase II study of trastuzumab emtansine in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer who were previously treated with trastuzumab, lapatinib, an anthracycline, a taxane, and capecitabine. J Clin Oncol. 2012 Sep 10;30(26):3234–41.
40. Wang X, Li W, Yin Y, Tong Z, Zhang Q, Zheng H, et al. Primary results of ELAINA: a randomized, multicenter, open-label, phase III study of the efficacy and safety of trastuzumab emtansine vs. lapatinib plus capecitabine in Chinese patients with HER2-positive locally advanced or metastatic breast cancer who have received prior trastuzumab-based therapy. Transl Breast Cancer Res. 2023 Jan 31;4:3.
41. Watanabe J, Ito Y, Saeki T, Masuda N, Takano T, Takao S, et al. Safety Evaluation of Trastuzumab Emtansine in Japanese Patients with HER2-Positive Advanced Breast Cancer. In Vivo. 2017 May-Jun;31(3):493–500.
42. Wildiers H, Meyskens T, Marréaud S, Lago LD, Vuylsteke P, Curigliano G, et al. Long term outcome data from the EORTC 75111–10114 ETF/BCG randomized phase II study: Pertuzumab and trastuzumab with or without metronomic chemotherapy for older patients with HER2-positive metastatic breast cancer, followed by T-DM1 after progression. Breast. 2022 Aug;64:100–111.
43. Yardley DA, Krop IE, LoRusso PM, Mayer M, Barnett B, Yoo B, et al. Trastuzumab Emtansine (T-DM1) in Patients With HER2-Positive Metastatic Breast Cancer Previously Treated With Chemotherapy and 2 or More HER2-Targeted Agents: Results From the T-PAS Expanded Access Study. Cancer J. 2015 Sep-Oct;21(5):357–64.
44. Fabi A, Alesini D, Valle E, Moscetti L, Caputo R, Caruso M, et al. T-DM1 and brain metastases: Clinical outcome in HER2-positive metastatic breast cancer. Breast. 2018 Oct;41:137–43.
45. Fabi A, De Laurentiis M, Caruso M, Valle E, Moscetti L, Santini D, et al. Efficacy and safety of T-DM1 in the 'common-practice' of HER2+ advanced breast cancer setting: a multicenter study. Oncotarget. 2017 Mar 18;8(38):64481–89.
46. Fabi A, Giannarelli D, Moscetti L, Santini D, Zambelli A, Laurentiis M, et al. Ado-trastuzumab emtansine (T-DM1) in HER2+ advanced breast cancer patients: does pretreatment with pertuzumab matter? Future Oncol. 2017 Dec;13(30):2791–97.
47. Acibuca A, Sezer A, Yilmaz M, Sumbul AT, Demircan S, Muderrisoglu IH, et al. Cardiotoxicity of trastuzumab emtansine (T-DM1): a single-center experience. J Int Med Res. 2021 Dec;49(12):3000605211053755.
48. Al Rabadi LS, Cook MM, Kaempf AJ, Saraceni MM, Savin MA, Mitri ZI. Ado-trastuzumab for the treatment of metastatic HER2-positive breast cancer in patients previously treated with Pertuzumab. BMC Cancer. 2021 Oct 27;21(1):1150.
49. Baek SK, Jeong JH, Jung K, Ahn HK, Kim MH, Sohn J, et al; Korean Cancer Study Group (KCSG). A nationwide real-world study for evaluation of effectiveness and safety of T-DM1 in patients with HER2-positive metastatic breast cancer in Korea (KCSG BR19-15). Ther Adv Med Oncol. 2024 Jan 28;16:17588359231225029.
50. Bahçeci A, Paydaş S, Ak N, Ferhatoğlu F, Saip PM, Seydaoğlu G, et al. Efficacy and Safety of Trastuzumab Emtansine in Her2 Positive Metastatic Breast Cancer: Real-World Experience. Cancer Invest. 2021 Jul-Aug;39(6-7):473–81.
51. Battisti NML, Rogerson F, Lee K, Shepherd S, Mohammed K, Turner N, et al. Safety and efficacy of T-DM1 in patients with advanced HER2-positive breast cancer The Royal Marsden experience. Cancer Treat Res Commun. 2020;24:100188.
52. Bian L, Li F, Ji C, Li J, Yuan Y, Zhang H, et al. ADC vs HP dual-antibody for HER2-positive advanced breast cancer after failed to TKI. J Clin Oncol. 2023;41:e13036.
53. Audi Blotta D, Gomes JR, Moreira SB, Colosimo DF, Araujo J, Cavalcanti Gonçalves Ferreira C, et al. Real-world outcomes of long-term efficacy of T-DM1 after discontinuation due to limiting toxicity in patients with HER2-positive metastatic breast cancer. J Clin Oncol. 2023;41:e13034.
54. Conte B, Fabi A, Poggio F, Blondeaux E, Dellepiane C, D'Alonzo A, et al; Gruppo Italiano Mammella (GIM) study group. T-DM1 Efficacy in Patients With HER2-positive Metastatic Breast Cancer Progressing After a Taxane Plus Pertuzumab and Trastuzumab: An Italian Multicenter Observational Study. Clin Breast Cancer. 2020 Apr;20(2):e181–7.
55. Daniels B, Kiely BE, Tang M, Houssami N, Lord SJ, Pearson SA. Trastuzumab emtansine for HER2-positive metastatic breast cancer: Outcomes from a whole-of-population Australian cohort. Breast. 2021 Aug;58:106–12.
56. Prete SD, Montella L, Arpino G, Buono G, Buonerba C, Dolce P, et al. Second line trastuzumab emtansine following horizontal dual blockade in a real-life setting. Oncotarget. 2020 Jun 2;11(22):2083–91.
57. Joshua Z Drago, Patricia R Pereira, Anton Safonov, Antonio Marra, Yi Rao, Bo Liu, et al; Abstract P2-13-19: Statin modulation of antibody drug conjugate activity in breast cancer models and patients. Cancer Res 15 February 2022; 82 (4_Supplement): P2–13–19.
58. Dzimitrowicz H, Berger M, Vargo C, Hood A, Abdelghany O, Raghavendra AS, et al. T-DM1 Activity in Metastatic Human Epidermal Growth Factor Receptor 2-Positive Breast Cancers That Received Prior Therapy With Trastuzumab and Pertuzumab. J Clin Oncol. 2016 Oct 10;34(29):3511–17.
59. Gogia A, Gupta A, Deo SV, Sharma D, Mathur S. Safety, efficacy and survival outcome of Ado-trastuzumab emtansine (T-DM1) in patients with metastatic HER2-positive breast cancer: An Indian experience. J Clin Oncol. 2022;40:e13008.
60. Hardy-Werbin M, Quiroga V, Cirauqui B, Romeo M, Felip E, Teruel I, et al. Real-world data on T-DM1 efficacy - results of a single-center retrospective study of HER2-positive breast cancer patients. Sci Rep. 2019 Sep 4;9(1):12760.
61. He M, Zhao W, Wang P, Li W, Chen H, Yuan Z, et al. Efficacy and safety of Trastuzumab Emtansine in treating human epidermal growth factor receptor 2-positive metastatic breast cancer in Chinese population: a real-world multicenter study. Front Med (Lausanne). 2024 Apr 29;11:1383279.
62. Hsieh W, Chang CL. Retrospective observational study of outcomes in HER2-positive metastatic breast cancer patients treated with trastuzumab emtansine in single center. JCO Glob Oncol. 2023;9:15.
63. Ji C, Li F, Yuan Y, Zhang H, Bian L, Zhang S, et al. Novel Anti-HER2 Antibody-Drug Conjugates Versus T-DM1 for HER2-Positive Metastatic Breast Cancer After Tyrosine Kinase Inhibitors Treatment. Oncologist. 2023 Oct 3;28(10):e859–66.
64. Li F, Xu F, Li J, Wang T, Bian L, Zhang S, et al. Pyrotinib versus trastuzumab emtansine for HER2-positive metastatic breast cancer after previous trastuzumab and lapatinib treatment: a real-world study. Ann Transl Med. 2021 Jan;9(2):103.
65. Lupichuk S, Cheung WY, Stewart D. Pertuzumab and Trastuzumab Emtansine for Human Epidermal Growth Factor Receptor-2-Positive Metastatic Breast Cancer: Contemporary Population-Based Outcomes. Breast Cancer (Auckl). 2019 Oct 9;13:1178223419879429.
66. Martinez-Garcia J, Boix AP, Henarejos PS, Lucas MJ, Saura AS, Luengo M, et al. 291P Trastuzumab emtansine in HER2-positive metastatic breast cancer after pertuzumab and trastuzumab: TDM1RM Study. Annals of Oncology. 2020 Sep 1;31:S358.
67. Martins CH, Pereira MV, Rocha JM, Oliveira C, Peixoto M, Rodrigues JR, et al. PR128 TRANSTUZUMAB EMTANSINE IN HER-2 POSITIVE METASTATIC BREAST CANCER: A RETROSPECTIVE STUDY IN A PORTUGUESE CENTRAL HOSPITAL. The Breast. 2023 Oct 1;71:S59–60.
68. Michel LL, Bermejo JL, Gondos A, Marmé F, Schneeweiss A. T-DM1 as a New Treatment Option for Patients with Metastatic HER2-positive Breast Cancer in Clinical Practice. Anticancer Res. 2015 Sep;35(9):5085–90.
69. Michel LL, Hartkopf AD, Fasching PA, Kolberg HC, Hadji P, Tesch H, et al. Progression-Free Survival and Overall Survival in Patients with Advanced HER2-Positive Breast Cancer Treated with Trastuzumab Emtansine (T-DM1) after Previous Treatment with Pertuzumab. Cancers (Basel). 2020 Oct 17;12(10):3021.
70. Migeotte A, Dufour V, van Maanen A, Berliere M, Canon JL, Taylor D, et al. Impact of the line of treatment on progression-free survival in patients treated with T-DM1 for metastatic breast cancer. BMC Cancer. 2021 Nov 11;21(1):1204.
71. Moinard-Butot F, Saint-Martin C, Pflumio C, Carton M, Jacot W, Cottu PH, et al. Efficacy of trastuzumab emtansine (T-DM1) and lapatinib after dual HER2 inhibition with trastuzumab and pertuzumab in patient with metastatic breast cancer: Retrospective data from a French multicenter real-life cohort. Breast. 2022 Jun;63:54–60.
72. Noda-Narita S, Shimomura A, Kawachi A, Sumiyoshi-Okuma H, Sudo K, Shimoi T, et al. Comparison of the efficacy of trastuzumab emtansine between patients with metastatic human epidermal growth factor receptor 2-positive breast cancers previously treated with combination trastuzumab and pertuzumab and with trastuzumab only in Japanese population. Breast Cancer. 2019 Jul;26(4):492–8.
73. Peters S, Tang HC, Shafiq T, Karim S, Khan S, Trivedi S, et al. A UK Real-world Evaluation of Efficacy and Toxicity of Second-line Trastuzumab Emtansine and Third-line Trastuzumab Deruxtecan in HER2-positive Advanced Breast Cancer. Clinical Oncology. 2023 Jun 1;35(6):e419.
74. .Pizzuti L, Krasniqi E, Sperduti I, Barba M, Gamucci T, Mauri M, et al. PANHER study: a 20-year treatment outcome analysis from a multicentre observational study of HER2-positive advanced breast cancer patients from the real-world setting. Ther Adv Med Oncol. 2021 Nov 29;13:17588359211059873.
75. Sanglier T, Shim J, Lamarre N, Peña-Murillo C, Antao V, Montemurro F. Trastuzumab emtansine vs lapatinib and capecitabine in HER2-positive metastatic breast cancer brain metastases: A real-world study. Breast. 2023 Jun;69:441–50.
76. Silva D, Duarte H, Leite-Silva P, Ferro JF, Prospero I, Barbosa D, et al. Is T-DM1 cardiotoxicity close monitoring relevant in palliative breast cancer patients? A single center experience using equilibrium radionuclide ventriculography (ERNV). European Journal of Nuclear Medicine and Molecular Imaging. 2023 Sep 1;50(Suppl 1):S617.
77. Tataroglu Ozyukseler D, Basak M, Ay S, Koseoglu A, Arıcı S, Oyman A, et al. Prognostic factors of ado-trastuzumab emtansine treatment in patients with metastatic HER-2 positive breast cancer. J Oncol Pharm Pract. 2021 Apr;27(3):547–54.
78. Vici P, Pizzuti L, Michelotti A, Sperduti I, Natoli C, Mentuccia L, et al. A retrospective multicentric observational study of trastuzumab emtansine in HER2 positive metastatic breast cancer: a real-world experience. Oncotarget. 2017 May 25;8(34):56921–31.
79. Yeo W, Luk MY, Soong IS, Yuen TY, Ng TY, Mo FK, Chan K, Wong SY, Tsang J, Leung C, Suen JJ, Ngan RK. Efficacy and tolerability of trastuzumab emtansine in advanced human epidermal growth factor receptor 2-positive breast cancer. Hong Kong Med J. 2018 Feb;24(1):56–62.
80. Cortés J, Kim SB, Chung WP, Im SA, Park YH, Hegg R, et al; DESTINY-Breast03 Trial Investigators. Trastuzumab Deruxtecan versus Trastuzumab Emtansine for Breast Cancer. N Engl J Med. 2022 Mar 24;386(12):1143–54.
81. Modi S, Jacot W, Iwata H, Park YH, Vidal Losada M, Li W, et al. Trastuzumab deruxtecan in HER2-low metastatic breast cancer: long-term survival analysis of the randomized, phase 3 DESTINY-Breast04 trial. Nat Med. 2025 Dec;31(12):4205–13.
82. Schettini F, Nucera S, Pascual T, Martínez-Sáez O, Sánchez-Bayona R, Conte B, et al. Efficacy and safety of antibody-drug conjugates in pretreated HER2-low metastatic breast cancer: A systematic review and network meta-analysis. Cancer Treat Rev. 2025 Jan;132:102865