Abstract
Fear can be induced either through direct experience of threat or through observation of others’ distress. This capacity for observational fear provides a survival advantage by enabling vicarious, social learning of potential danger and forms a foundation for empathy. While the anterior cingulate cortex (ACC) has long been implicated in mediating observational fear, the neuromodulatory mechanisms that distinguish it from direct fear have been unclear. Our recent work demonstrates that the locus coeruleus (LC), the brain’s major noradrenergic nucleus, contributes in a lateralized manner to these distinct states. Specifically, the right LC→ACC pathway is required for sustaining vicarious freezing, but not for direct fear. This finding shifts the view of the LC from a diffuse arousal center to a differentiated neuromodulatory hub that separates self-referenced and socially acquired fear. Extending the principle of hemispheric specialization into neuromodulatory systems, the LC lateralization illustrates how ancient brainstem nuclei have been adapted to support the demands of social survival. This insight highlights a broader principle: neuromodulatory nuclei act as specialized contributors to distinct fear states.
Keywords
Vicarious fear, Locus coeruleus, Anterior cingulate cortex, Hemispheric lateralization, Empathy, Noradrenergic system
Introduction
Fear is a fundamental emotion that has evolved to protect organisms from imminent harm by mobilizing rapid defensive responses. In social animals, however, fear extends beyond the direct encounter with danger. Observing the distress of others can trigger defensive behaviors, a phenomenon known as vicarious or observational fear. This ability confers a survival advantage by allowing individuals to learn about threats without direct exposure, and it forms a biological foundation for empathy and social cohesion. Rodent studies have firmly established the anterior cingulate cortex (ACC) as a key structure for observational fear. Lesions or optogenetic manipulations of this region abolish the expression of observational freezing while sparing direct fear, and recordings demonstrate that cingulate theta oscillations synchronize with the basolateral amygdala during these experiences [1,2]. Human neuroimaging converges on the same regions, the ACC and the insula consistently activated during empathy for pain [3]. Yet the upstream modulatory systems that distinguish direct fear driven by one’s own experience from vicarious fear elicited by another’s suffering have remained less clear. One prime candidate for such a modulatory role is the locus coeruleus (LC), given its known projections to the ACC and its involvement in arousal and attention to salient stimuli.
Our study provides a decisive advance in this field. Using male mice, our work demonstrates that the LC, the brainstem hub of norepinephrine release, plays a lateralized role in differentiating direct from vicarious fear. Specifically, we identify the right LC→ACC projection as crucial for sustaining empathic freezing, while direct fear responses remain intact when this pathway is disrupted [4]. This discovery reframes the LC from a global arousal center into a lateralized neuromodulatory switch that distinguishes “my fear” from “your fear.”
The LC Beyond Arousal
For decades the LC was viewed primarily as a diffuse alarm system, releasing norepinephrine broadly across the brain during stress. Contemporary models, however, emphasize its role in adaptive gain. In this view, brief phasic bursts of the LC activity sharpen attention and enhance memory consolidation for salient events, whereas tonic firing elevates vigilance but diminishes precision [5]. Importantly, the LC neurons are not homogeneous: anatomical tracing and physiological studies have identified projection-defined ensembles that innervate distinct structures, including the hippocampus, motor cortex, amygdala, and prefrontal cortex [6]. Each subset can be independently recruited, allowing the LC to exert selective control over specific computations rather than acting as a uniform arousal knob. Within this framework, the ACC emerges as a privileged target. Importantly, if the LC exhibits such projection-specific specialization, it is plausible that these projections could also be lateralized in their function, allowing for further differentiation of behavioral states. Recent studies show that LC projections to the ACC are activated during socially salient events such as parental interactions, and that manipulation of this pathway alters social sensitivity [7]. These findings suggest that the LC is not only a regulator of arousal but also an active modulator of social cognition.
Dissociable Mechanisms of Direct and Vicarious Fear
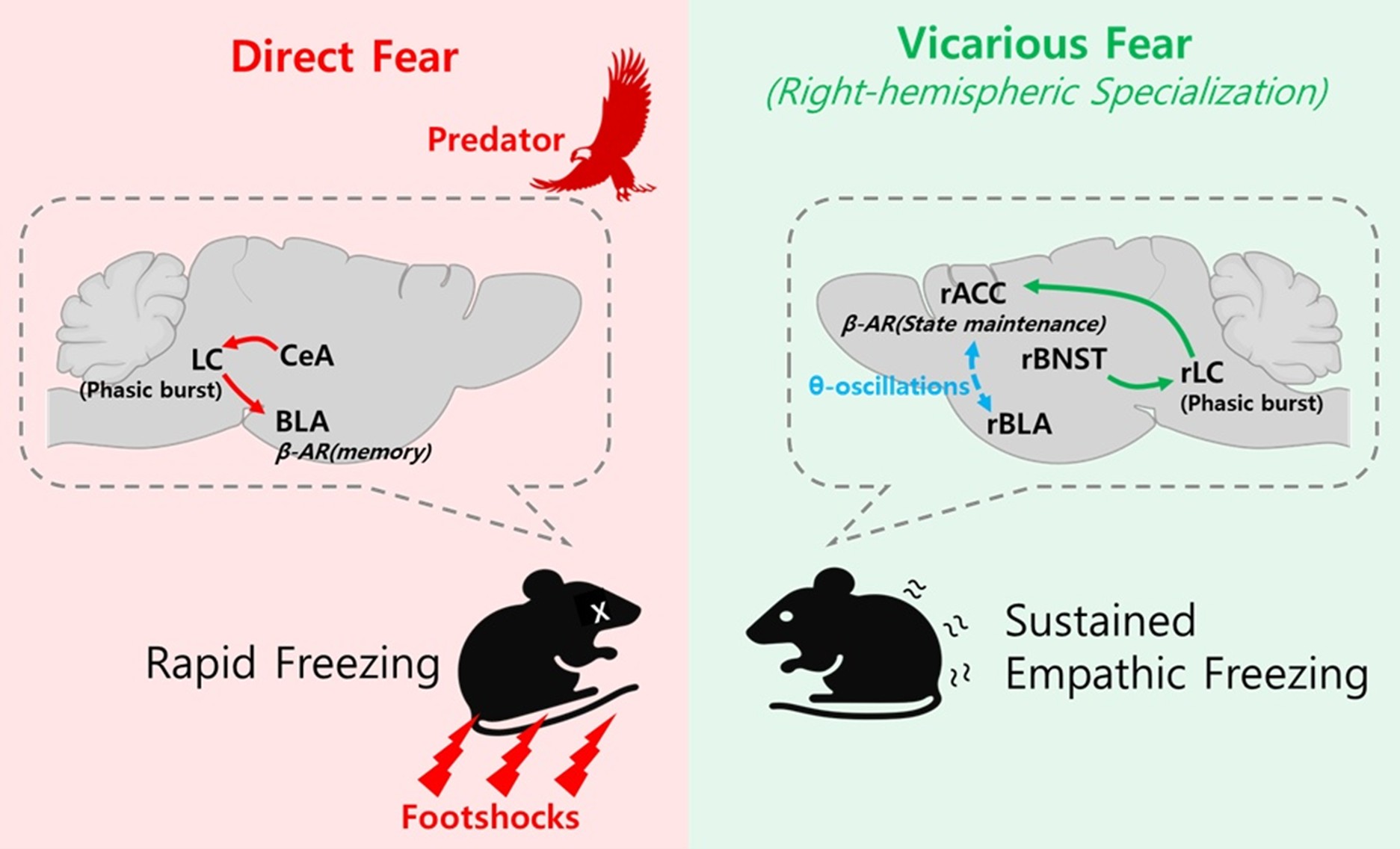
The central insight from our study is that the LC operates in a lateralized manner to support distinct fear states. Silencing the right hemispheric LC→ACC projection abolished the maintenance of observational freezing in mice but did not affect direct fear responses [4]. This dissociation indicates that direct fear is primarily mediated by rapid sensory input to the amygdala and its downstream hypothalamic and brainstem effectors, whereas vicarious fear requires sustained neuromodulatory support from the right LC to the right ACC (Figure 1). This lateralization is consistent with prior evidence from rodent observational fear paradigms, where unilateral lesion experiments demonstrated that only the right ACC, but not the left, is required for the expression of observational freezing [8]. Further, genetic investigations revealed that SST+ interneurons in the ACC regulate OFL in a cell-type–specific manner via Neurexin-3 (Nrxn3); selective deletion of Nrxn3 in SST interneurons markedly enhanced observational fear, underscoring the role of local inhibitory control in shaping empathic processing [9]. Additionally, 5-7 Hz rhythmic oscillations in the right cingulo-amygdala circuit have been shown to be essential for empathic freezing [10]. Together, these findings suggest that the maintenance of vicarious fear depends on both specialized right-hemisphere networks and balanced local inhibitory microcircuits within the ACC.
Upstream pathways provide a mechanistic rationale for this segregation. The bed nucleus of the stria terminalis (BNST), a structure critically involved in processing uncertain and sustained threat as well as integrating visual threat cues, has been shown to regulate prolonged vigilance and anxiety-like states [11–14]. These properties suggest that the BNST is well positioned to modulate the LC activity during situations where threat is inferred rather than directly experienced. Since our fiber photometry recordings showed that only the right LC(rLC) → right ACC(rACC) circuit showed increased activity during observational fear [4], we hypothesize that socially observed, uncertain threats are relayed via the right BNST (rBNST) to preferentially activate the rLC, which in turn sustains activity in the rACC to maintain vicarious freezing. An important question for future research is whether this lateralization arises within the LC or reflects already-lateralized inputs from upstream structures such as the BNST. By contrast, the central amygdala (CeA) has been classically linked to phasic and predictable fear through its inhibitory microcircuits and descending projections to brainstem effectors [15,16]. Such distinctions highlight a division of labor in which the BNST supports anxiety-like states of uncertainty, while the CeA mediates acute defensive reactions.
Cellular and Circuit Mechanisms
The selective involvement of the right LC in observational fear can be understood at multiple mechanistic levels. At the receptor level, norepinephrine exerts distinct effects depending on arousal state. Activation of α2A-adrenergic receptors in the medial prefrontal cortex stabilizes recurrent network activity during moderate arousal, providing a mechanistic template for how noradrenergic modulation can maintain sustained representations of emotionally salient information [17]. By extension, similar receptor dynamics may operate in the ACC during observational fear, helping to stabilize empathic states once initiated. In the ACC, α1-adrenoceptor signaling did not significantly contribute to observational freezing [4], suggesting that α2A-mediated mechanisms, rather than α1-mediated excitation, may be critical for stabilizing empathic states during observational fear. Under conditions of higher arousal, norepinephrine engages β-adrenoceptors, which enhance neuronal excitability and modulate synaptic plasticity [18–21]. This mechanism appears particularly relevant for vicarious fear, since socially acquired threat cues are inherently indirect and less salient than direct aversive experiences. By boosting synaptic gain and facilitating long-term plasticity, β-adrenoceptor signaling reinforces socially derived threat memories, ensuring that observational fear is consolidated and maintained beyond the immediate context. By contrast, direct fear relies less on sustained neuromodulation and more on rapid transmission through the amygdala. Phasic sensory input quickly activates CeA inhibitory microcircuits, which in turn drive downstream hypothalamic and brainstem effectors to produce acute freezing [15,16]. In this state, β-adrenergic modulation of basolateral amygdala circuits facilitates the consolidation of directly experienced aversive memories [20–23], but the initiation of defensive responses can occur even without prolonged cortical involvement.
The distinct firing modes of LC neurons provide a mechanistic framework for understanding these differential roles [24]. Phasic LC bursts, characterized by brief high-frequency discharges in response to salient stimuli, transiently enhance cortical excitability and facilitate rapid memory encoding. Such brief, isolated phasic responses are well-suited for the immediate defensive responses characteristic of direct fear. By contrast, sustained tonic LC activity maintains elevated cortical arousal over extended periods, supporting general vigilance but with reduced precision. Our finding that the rLC→rACC pathway is required to sustain observational freezing suggests that this circuit operates through rhythmic phasic bursts of noradrenergic modulation, rather than continuous tonic discharge, to maintain sustained yet precise cortical engagement necessary for processing nuanced social cues.
At the projection level, the LC neurons innervating the ACC release norepinephrine in synchrony with socially salient cues, and experimental manipulations of this pathway bidirectionally alter social behaviors [7]. Oscillatory coordination provides an additional explanatory layer. Observational fear depends on the ACC-amygdala theta coupling [2,10], and the right ACC theta has been causally linked to empathic freezing. Critically, because the LC bursts are known to entrain cortical rhythms [25] and synchronized oscillations between prefrontal cortex and amygdala support fear expression [24], we propose that phasic bursts from the right LC serve as a pacemaker that reinforces theta-synchronized ensembles in the right ACC, thereby stabilizing vicarious freezing over time. This oscillatory entrainment model provides a unifying mechanism for how lateralized neuromodulation sustains socially acquired fear states.
Evolutionary Significance of Lateralized Locus Coeruleus
The observation that the LC itself is lateralized in function aligns with the broader principle of hemispheric specialization, a deeply conserved feature across vertebrate evolution. Brain asymmetry has been documented in species ranging from fish and amphibians to birds and mammals, and is thought to provide adaptive advantages by allowing parallel processing of distinct behavioral demands while reducing cross-hemispheric interference. Classic examples include left-hemisphere dominance for birdsong learning, lateralized visual threat detection in fish, and human language specialization [26,27]. These converging lines of evidence underscore that lateralization is not an evolutionary anomaly but a recurring design principle that enhances efficiency and flexibility of neural processing. Within this comparative framework, the emergence of lateralization in the LC is particularly striking. The LC is a phylogenetically ancient, compact nucleus that provides widespread neuromodulatory input across the brain. Notably, functional lateralization can emerge even in brainstem nuclei positioned near the midline; for instance, the dorsal raphe nucleus has been shown to exhibit asymmetric function despite its central location [28], demonstrating that anatomical position does not preclude hemispheric specialization. Its hemispheric specialization demonstrates that even highly conserved modulatory systems can acquire asymmetries in the service of increasingly complex behavioral demands. In rodents, the right LC→ACC pathway supports the maintenance of vicarious fear, suggesting that evolutionary pressures favored the segregation of socially acquired threat processing from circuits dedicated to direct survival responses. Such an arrangement provides a clear functional advantage. By lateralizing the neuromodulatory control of self-referenced versus socially referenced fear, the brain reduces competition between immediate defensive reactions and the sustained vigilance required for processing social cues. This division of labor exemplifies how evolution repurposes ancient nuclei for novel challenges, extending the principle of lateralization from sensory and motor domains into neuromodulatory systems to support novel social behaviors.
Future directions
Future research should aim to validate these findings across species. Human studies combining the LC imaging, the ACC electrophysiology, and pupil-based measures of arousal during empathy tasks could test the prediction of right-biased coupling. In rodents, projection- and receptor-specific experiments, for example using DREADDs or optogenetics to selectively manipulate β-adrenoceptor signaling in the ACC terminals of LC neurons, can parse the contributions of β versus α2A signaling to the maintenance of empathic freezing. Behavioral paradigms that manipulate the predictability of observed distress will allow investigators to assess how uncertainty recruits BNST→LC→ACC circuits [12–14]. Developmental and sex-specific studies are needed as well, since the LC stress reactivity is sexually dimorphic [29]. Given that sex differences in neuromodulatory responses to stress and social stimuli have been increasingly recognized, future work should examine whether the lateralized LC→ACC pathway exhibits similar functional asymmetry in female mice, or whether sex-specific circuits underlie observational fear. Future studies should also identify the specific neuron types within the BNST, BLA, and CeA that interact with the lateralized LC circuit using cell-type-specific genetic approaches. Additionally, given the known direct projections between BLA and CeA, investigating whether closed-loop circuits, such as BLA→CeA→LC→BLA, contribute to sustaining freezing states through recurrent amplification will be important for understanding circuit dynamics. Finally, future work should examine how glial cells shape the LC circuit plasticity under social stress. Astrocytes in the LC have been shown to become activated under conditions of maternal separation and social isolation, suggesting a potential role in stress-induced modulation of noradrenergic output [30,31]. Microglia have likewise emerged as key regulators of stress effects on memory, raising the possibility that they contribute to the remodeling of the LC circuits during socially relevant stress experiences [32].
Conclusion
By identifying the right LC→ACC pathway as essential for sustaining vicarious but not direct fear, our study redefines the role of the LC in emotional processing. Traditionally regarded as a diffuse arousal system, the LC is now revealed to possess lateralized specializations that differentially support distinct modes of fear. This lateralization highlights a fundamental principle: that the social brain does not simply recycle circuits evolved for individual survival, but requires specialized neuromodulatory streams to maintain empathic states.
The implications of this finding extend in several directions. At the mechanistic level, it raises new questions about how receptor-specific noradrenergic signaling and glial interactions shape the stability of socially acquired fear memories. At the circuit level, it points to the LC as a hub where limbic and cortical inputs are differentially integrated to serve self-referenced versus other-referenced fear. At the systems level, it illustrates how lateralization, a principle often discussed in the context of higher cognition such as language, extends down to brainstem nuclei to sculpt social behavior.
Ultimately, the discovery that the LC is not merely an arousal switch but a lateralized gatekeeper of social fear underscores a broader principle: neuromodulatory nuclei are not uniform regulators but specialized and differentiated contributors to the architecture of distinct fear states.
Acknowledgements
This work was supported by the Institute for Basic Science (IBS-R001-Y3-2025-a00, Young Scientist Fellowship).
References
2. Jeon D, Kim S, Chetana M, Jo D, Ruley HE, Lin SY, et al. Observational fear learning involves affective pain system and Cav1.2 Ca2+ channels in ACC. Nat Neurosci. 2010 Apr;13(4):482–8.
3. Lamm C, Decety J, Singer T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. Neuroimage. 2011 Feb 1;54(3):2492–502.
4. Kim JH, Choi DE, Shin HS. The lateralized LC-NAergic system distinguishes vicarious versus direct fear in mice. Nat Commun. 2025 Mar 10;16(1):2364.
5. Aston-Jones G, Cohen JD. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu Rev Neurosci. 2005;28:403–50.
6. Chandler DJ, Gao WJ, Waterhouse BD. Heterogeneous organization of the locus coeruleus projections to prefrontal and motor cortices. Proc Natl Acad Sci U S A. 2014 May 6;111(18):6816–21.
7. Corona A, Choe J, Muñoz-Castañeda R, Osten P, Shea SD. A circuit from the locus coeruleus to the anterior cingulate cortex modulates offspring interactions in mice. Cell Rep. 2023 Jul 25;42(7):112771.
8. Kim S, Mátyás F, Lee S, Acsády L, Shin HS. Lateralization of observational fear learning at the cortical but not thalamic level in mice. Proc Natl Acad Sci U S A. 2012 Sep 18;109(38):15497–501.
9. Keum S, Kim A, Shin JJ, Kim JH, Park J, Shin HS. A Missense Variant at the Nrxn3 Locus Enhances Empathy Fear in the Mouse. Neuron. 2018 May 2;98(3):588–601.e5.
10. Kim SW, Kim M, Baek J, Latchoumane CF, Gangadharan G, Yoon Y, et al. Hemispherically lateralized rhythmic oscillations in the cingulate-amygdala circuit drive affective empathy in mice. Neuron. 2023 Feb 1;111(3):418–29.e4.
11. Goode TD, Ressler RL, Acca GM, Miles OW, Maren S. Bed nucleus of the stria terminalis regulates fear to unpredictable threat signals. Elife. 2019 Apr 4;8:e46525.
12. Lebow MA, Chen A. Overshadowed by the amygdala: the bed nucleus of the stria terminalis emerges as key to psychiatric disorders. Mol Psychiatry. 2016 Apr;21(4):450–63.
13. Avery SN, Clauss JA, Blackford JU. The Human BNST: Functional Role in Anxiety and Addiction. Neuropsychopharmacology. 2016 Jan;41(1):126–41.
14. Brinkmann L, Buff C, Feldker K, Neumeister P, Heitmann CY, Hofmann D, et al. Inter-individual differences in trait anxiety shape the functional connectivity between the bed nucleus of the stria terminalis and the amygdala during brief threat processing. Neuroimage. 2018 Feb 1;166:110–16.
15. Ciocchi S, Herry C, Grenier F, Wolff SB, Letzkus JJ, Vlachos I, et al. Encoding of conditioned fear in central amygdala inhibitory circuits. Nature. 2010 Nov 11;468(7321):277–82.
16. Haubensak W, Kunwar PS, Cai H, Ciocchi S, Wall NR, Ponnusamy R, et al. Genetic dissection of an amygdala microcircuit that gates conditioned fear. Nature. 2010 Nov 11;468(7321):270–6.
17. Wang M, Ramos BP, Paspalas CD, Shu Y, Simen A, Duque A, et al. Alpha2A-adrenoceptors strengthen working memory networks by inhibiting cAMP-HCN channel signaling in prefrontal cortex. Cell. 2007 Apr 20;129(2):397–410.
18. Arnsten AF. Stress signalling pathways that impair prefrontal cortex structure and function. Nat Rev Neurosci. 2009 Jun;10(6):410–22.
19. Perez DM, editor. The Adrenergic Receptors. Totowa, NJ: Humana Press; 2006.
20. Ferry B, Roozendaal B, McGaugh JL. Basolateral amygdala noradrenergic influences on memory storage are mediated by an interaction between beta- and alpha1-adrenoceptors. J Neurosci. 1999 Jun 15;19(12):5119–23.
21. Villain H, Benkahoul A, Drougard A, Lafragette M, Muzotte E, Pech S, et al. Effects of Propranolol, a β-noradrenergic Antagonist, on Memory Consolidation and Reconsolidation in Mice. Front Behav Neurosci. 2016 Mar 14;10:49.
22. Strange BA, Dolan RJ. Beta-adrenergic modulation of emotional memory-evoked human amygdala and hippocampal responses. Proc Natl Acad Sci U S A. 2004 Aug 3;101(31):11454–8.
23. Tully K, Li Y, Tsvetkov E, Bolshakov VY. Norepinephrine enables the induction of associative long-term potentiation at thalamo-amygdala synapses. Proc Natl Acad Sci U S A. 2007 Aug 28;104(35):14146–50.
24. Karalis N, Dejean C, Chaudun F, Khoder S, Rozeske RR, Wurtz H, et al. 4-Hz oscillations synchronize prefrontal-amygdala circuits during fear behavior. Nat Neurosci. 2016 Apr;19(4):605–12.
25. Sara SJ, Bouret S. Orienting and reorienting: the locus coeruleus mediates cognition through arousal. Neuron. 2012 Oct 4;76(1):130–41.
26. Bisazza A, Rogers LJ, Vallortigara G. The origins of cerebral asymmetry: a review of evidence of behavioural and brain lateralization in fishes, reptiles and amphibians. Neurosci Biobehav Rev. 1998 May;22(3):411–26.
27. Corballis MC. Evolution of cerebral asymmetry. Prog Brain Res. 2019;250:153–78.
28. Crespi F, Jouvet M. Has the raphe dorsalis nucleus an asymmetric function? Exp Brain Res. 1984;56(3):403–9.
29. Martinez-Muniz GA, Wood SK. Sex Differences in the Inflammatory Consequences of Stress: Implications for Pharmacotherapy. J Pharmacol Exp Ther. 2020 Oct;375(1):161–74.
30. Nakamoto K, Aizawa F, Kinoshita M, Koyama Y, Tokuyama S. Astrocyte Activation in Locus Coeruleus Is Involved in Neuropathic Pain Exacerbation Mediated by Maternal Separation and Social Isolation Stress. Front Pharmacol. 2017 Jun 28;8:401.
31. Miguel-Hidalgo JJ. Astroglia in the Vulnerability to and Maintenance of Stress-Mediated Neuropathology and Depression. Front Cell Neurosci. 2022 Apr 22;16:869779.
32. Sanguino-Gómez J, Buurstede JC, Abiega O, Fitzsimons CP, Lucassen PJ, Eggen BJL, et al. An emerging role for microglia in stress-effects on memory. Eur J Neurosci. 2022 May;55(9-10):2491–518.