Abstract
Background: Tuberculous pericarditis is a rare manifestation of tuberculosis in developed countries and poses a significant diagnostic challenge due to its nonspecific clinical presentation. Immunocompromised individuals, particularly those receiving tumor necrosis factor-alpha (TNF-α) inhibitors, are at substantially increased risk of developing tuberculosis. Therefore, screening for tuberculosis infection and active disease is essential prior to initiating biological therapy.
Case Report: A woman in her 20s with ankylosing spondylitis had negative tuberculosis screening prior to starting adalimumab therapy. After approximately one year of treatment, she presented with a dry cough, exertional dyspnea, and fever. Imaging revealed diffuse miliary pulmonary nodules, focal consolidations, hilar and mediastinal lymphadenopathy, and a large pericardial effusion. Transthoracic echocardiography confirmed cardiac tamponade. Echocardiography-guided pericardiocentesis was performed, and pericardial fluid was submitted for microbiological analysis. Disseminated tuberculosis with pericardial involvement was microbiologically confirmed by positive Xpert MTB/RIF and mycobacterial culture results from pericardial fluid, sputum, and stool specimens. The patient was transferred to a specialist unit and started on first-line antituberculous therapy combined with corticosteroids to prevent constrictive pericarditis. She demonstrated progressive clinical and radiological improvement, including complete resolution of the pericardial effusion and negative follow-up sputum microscopy, and was discharged to complete therapy with outpatient follow-up.
Conclusions: This case demonstrates that severe extrapulmonary tuberculosis, including tuberculous pericarditis complicated by life-threatening cardiac tamponade, can occur in patients receiving TNF-α inhibitors despite negative pre-treatment screening. It highlights the limitations of baseline tuberculosis testing, underscores the need for sustained clinical vigilance during biological therapy, and illustrates successful management with first-line antituberculous therapy combined with corticosteroids.
Keywords
Cardiac tamponade, Tuberculous pericarditis, Adalimumab, Case report
Introduction
Immunocompromised patients are at a markedly increased risk of tuberculosis compared with the general population [1]. The introduction of tumor necrosis factor-alpha (TNF-α) inhibitors has significantly improved outcomes in inflammatory diseases. However, TNF-α blockade impairs granuloma maintenance and may predispose to tuberculosis reactivation or rapid disease progression. Therefore, all national guidelines recommend that, prior to initiating biologic therapy, patients undergo screening for tuberculosis infection, preferably with an interferon-γ release assay (IGRA) or, alternatively, a tuberculin skin test (TST). In addition, active disease must be excluded through clinical evaluation, imaging, and microbiological testing [2]. Nevertheless, cases of active tuberculosis continue to be reported during biologic therapy, and uncommon but severe organ-specific complications may occur, underscoring the need for ongoing vigilance and long-term safety monitoring.
In developed countries, tuberculous pericarditis accounts for fewer than 4% of all cases of pericardial disease. Pericardial involvement may manifest as pericardial effusion, effusive–constrictive pericarditis, or constrictive pericarditis. According to the European Society of Cardiology guidelines, a definitive diagnosis of tuberculous pericarditis requires demonstration of Mycobacterium tuberculosis in pericardial fluid or pericardial tissue, either by culture or polymerase chain reaction [3].
This case report describes a woman in her 20s receiving TNF-α inhibitor therapy (adalimumab) who, despite negative tuberculosis screening prior to treatment, developed disseminated tuberculosis complicated by tuberculous pericarditis and life-threatening cardiac tamponade within one year of therapy initiation.
Case Description
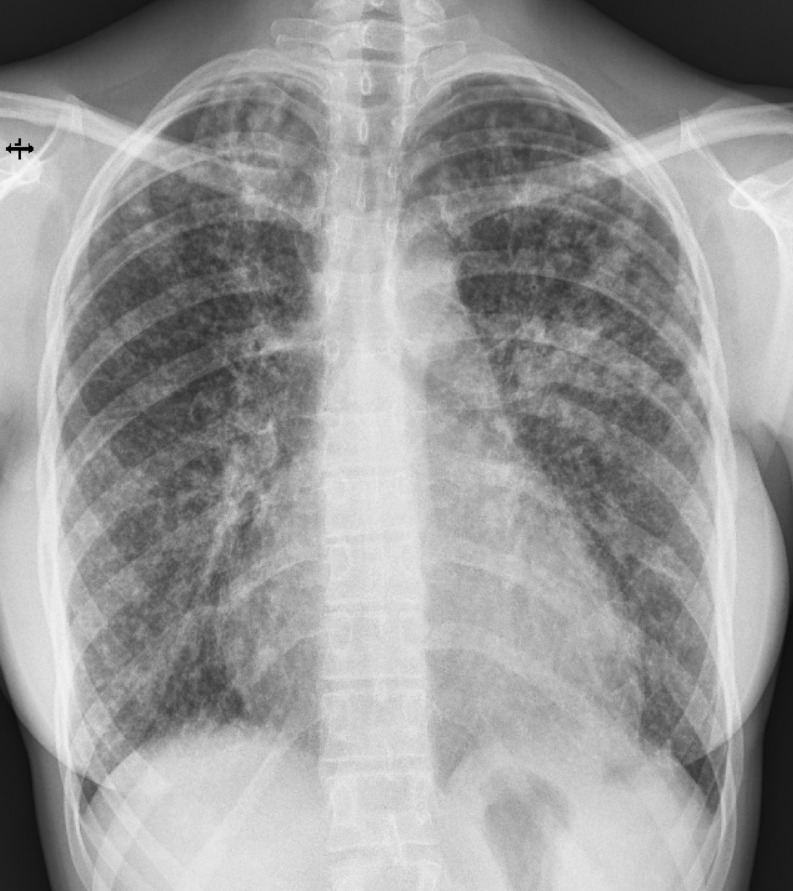
A woman in her 20s presented with a 3-week history of dry cough, exertional dyspnea, and fever, without night sweats or weight loss. She initially consulted her general practitioner, who prescribed antibiotic therapy and referred her for chest radiography. The chest X-ray demonstrated diffuse bilateral nodular opacities, consolidation in the left upper lobe, and cardiomegaly with an estimated cardiothoracic ratio of 0.64 (Figure 1). Based on these findings, she was referred to the Emergency Department of a tertiary care hospital the following day.
The patient’s past medical history was significant for ankylosing spondylitis, for which she had been receiving adalimumab every two weeks for approximately one year. Screening for latent tuberculosis infection with a QuantiFERON-TB Gold Plus interferon-gamma release assay (IGRA) before initiation of biologic therapy had been negative, and baseline non-contrast chest computed tomography (CT) performed several months earlier had shown no abnormalities. Given the negative baseline IGRA and unremarkable chest imaging, no prophylactic therapy for latent tuberculosis infection was initiated prior to and during adalimumab treatment. There was no known history of tuberculosis exposure, recent travel to high-prevalence regions, or relevant family history.
On admission, she was tachycardic at 120 beats per minute but hemodynamically stable. Electrocardiography demonstrated sinus tachycardia without other abnormalities. Laboratory testing revealed an elevated C-reactive protein (88.5 mg/L), while the leukocyte count remained within normal limits. Rapid urine antigen testing for Streptococcus pneumoniae, as well as SARS-CoV-2 and influenza A/B RNA testing, were negative.
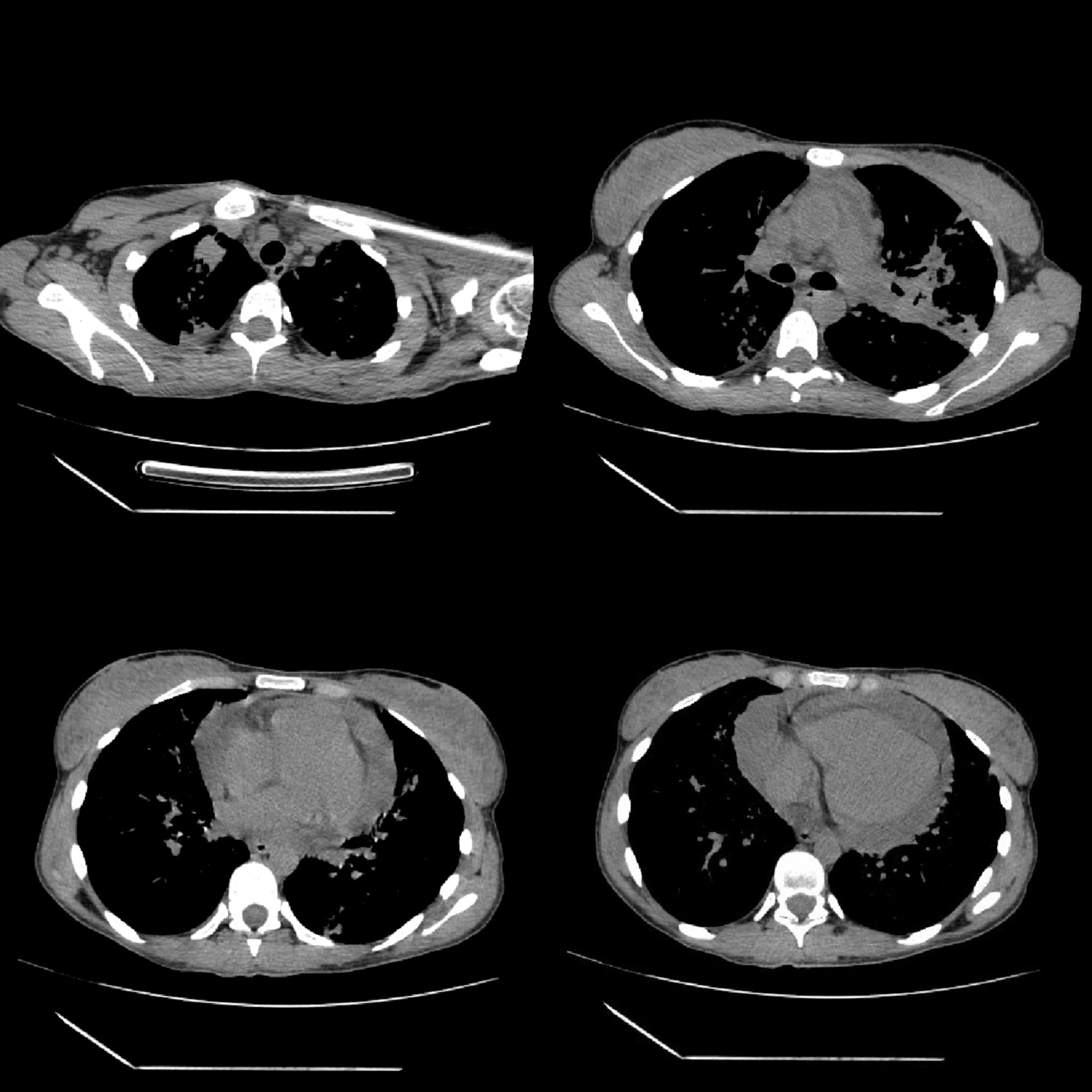
Non-contrast chest CT demonstrated diffuse miliary nodularity with focal consolidations in the right upper lobe segment S1 and left upper lobe segment S1, accompanied by hilar and mediastinal lymphadenopathy and a large pericardial effusion measuring up to 4 cm in thickness (Figures 2 and 3). A cardiology consultation was obtained. Screening transthoracic echocardiography (TTE) demonstrated a pericardial effusion measuring 3.8 cm, with evidence of cardiac tamponade. Although the patient remained hemodynamically stable, the large pericardial effusion with echocardiographic tamponade physiology warranted urgent echocardiography-guided pericardiocentesis to prevent progression to obstructive shock. The procedure reduced the effusion to 2.0 cm with resolution of tamponade, and a pericardial drain was left in situ. Pericardial fluid, sputum, urine, and stool samples were collected and tested for Mycobacterium tuberculosis (MTB) using smear microscopy, the Xpert MTB/RIF assay, and culture. MTB was detected by the Xpert MTB/RIF assay (positive for MTB DNA without rifampicin resistance) in multiple specimens, including sputum, pericardial fluid, and feces. Smear microscopy was negative in all samples except for scant acid-fast bacilli (AFB) detected in a single sputum specimen (Table 1).
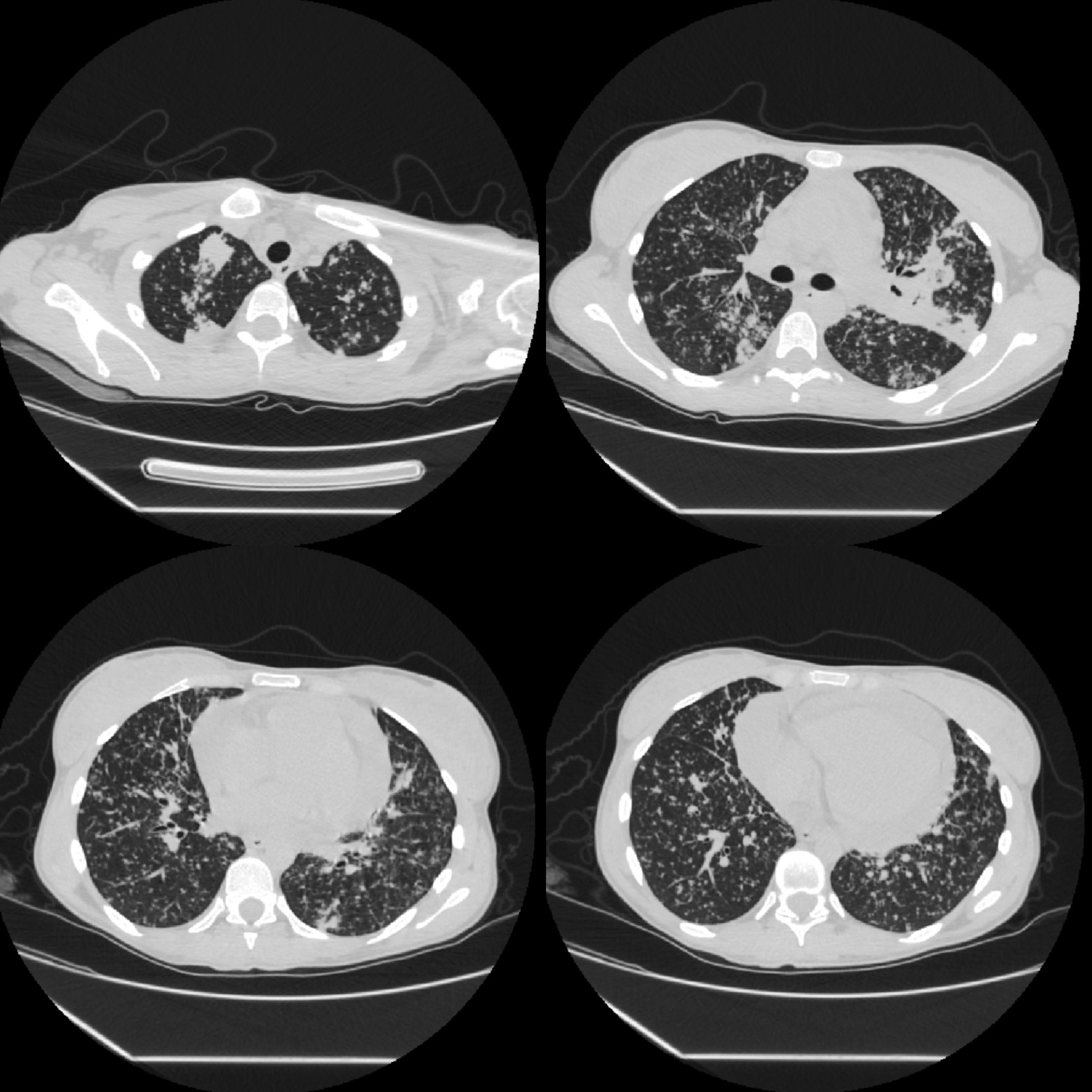
Figure 2. Non-contrast chest CT scan, lung window (description above in the text).
|
Smear microscopy |
Xpert MTB/RIF assay |
Culture |
||||
|
Date |
Material |
Ziehl-Neelsen |
Fluorescence microscopy |
Xpert MTB/RIF assay |
Liquid media |
Solid media |
|
28.05.2025 |
Sputum |
Negative |
2 AFB |
Positive for MTB DNA (low concentration), without RIF resistence |
Positive |
Positive (15 colonies) |
|
28.05.2025 |
Pericardial fluid |
Negative |
Negative |
'Trace', RIF resistance cannot be determined |
Negative |
Positive (4 colonies) |
|
28.05.2025 |
Urine |
— |
Negative |
Negative |
Negative |
Negative |
|
28.05.2025 |
Blood |
— |
— |
— |
Negative |
— |
|
29.05.2025 |
Feces |
— |
Negative |
Positive for MTB DNA (low concentration), without RIF resistance |
Positive |
Positive (2 colonies) |
|
29.05.2025 |
Sputum |
— |
Negative |
— |
— |
Positive (5 colonies) |
|
11.06.2025 |
Sputum |
Negative |
Negative |
— |
— |
— |
|
12.06.2025 |
Sputum |
— |
Negative |
— |
— |
— |
|
13.06.2025 |
Sputum |
— |
Negative |
— |
— |
— |
|
MTB: Mycobacterium tuberculosis; RIF: Rifampicin; AFB: Acid-Fast Bacilli; — = not tested, 'Trace' — the lowest detectable amount of MTB DNA. |
||||||
The patient was transferred to a specialist tuberculosis unit, where she was placed in isolation. Standard first-line antituberculous therapy was initiated with isoniazid, rifampicin, pyrazinamide, and ethambutol for 2 months, followed by isoniazid and rifampicin for a planned 4 months (2HRZE/4HR). Adalimumab therapy was discontinued upon diagnosis. To mitigate the risk of progression to constrictive tuberculous pericarditis, adjunctive corticosteroid therapy was initiated. Oral prednisolone was started at 60 mg daily, followed by a structured taper of 10 mg per week. Concurrent screening for HIV and viral hepatitis B and C was negative.
No further drainage output was observed after several days. Repeat transthoracic echocardiography performed approximately one week later demonstrated well-defined pericardial layers and complete resolution of the effusion (Figure 4), with otherwise normal cardiac findings, and the drain was removed.
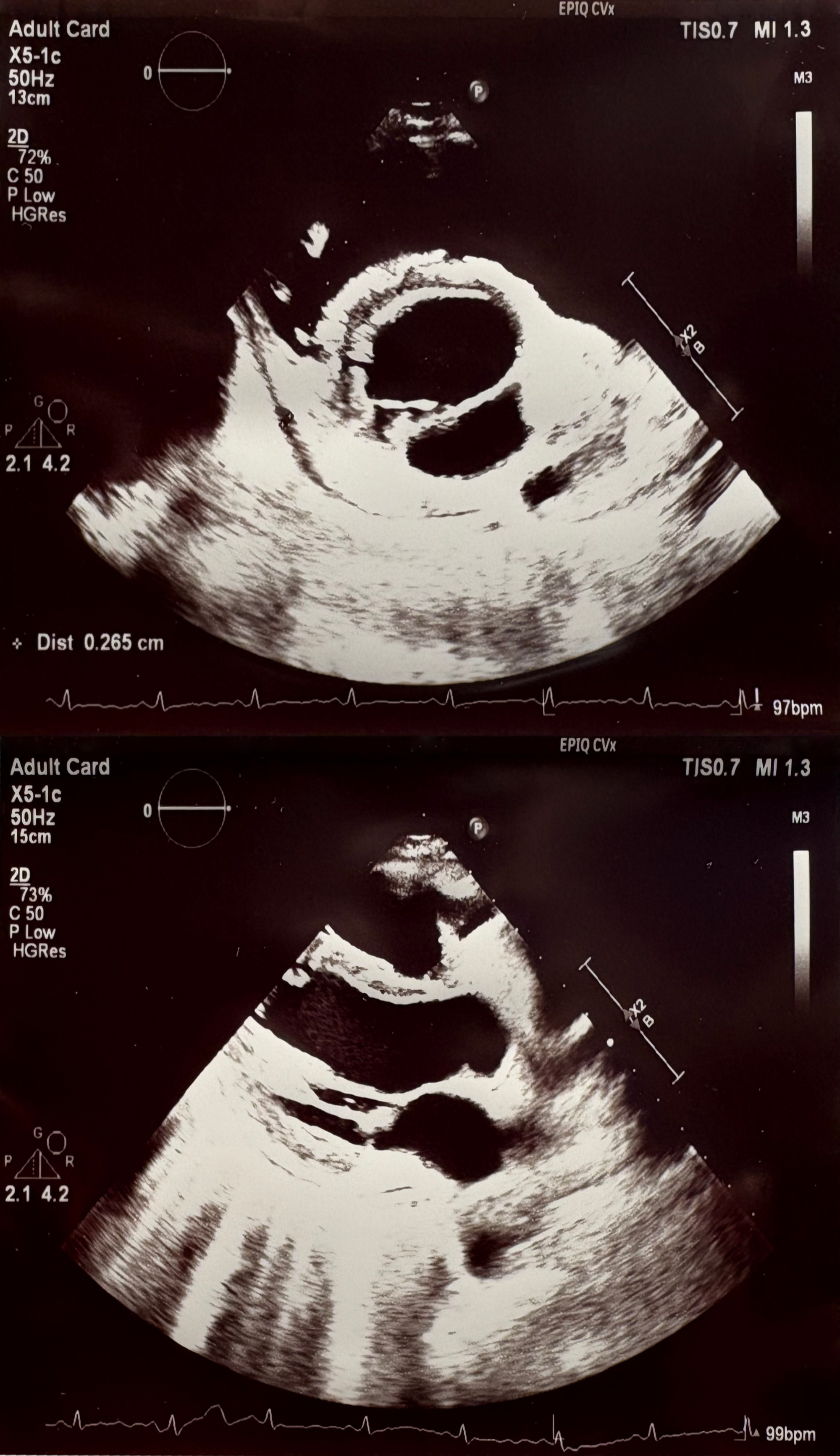
At the two-week follow-up, three consecutive sputum microscopy examinations were negative for AFB. The patient was subsequently discharged with instructions to maintain the prescribed antituberculous regimen, supplemented by outpatient clinical surveillance and a structured corticosteroid taper. Adalimumab therapy was reinitiated after two months of antituberculous treatment.
Discussion
This case underscores the risk of tuberculosis reactivation or dissemination during TNF-α inhibitor therapy, even in regions with low tuberculosis incidence, where clinicians may be inclined to underestimate this risk.
The association between TNF-α inhibitor therapy and tuberculosis reactivation is well established worldwide and is strongly influenced by the background tuberculosis burden. Incidence rates are higher in high-burden regions such as South America and Asia. However, cases continue to occur in low-incidence settings, including Europe [4], highlighting the limitations of current diagnostic tools and the need for continued vigilance, even in these regions.
In the presented clinical case, despite negative chest imaging and a negative IGRA prior to the initiation of biologic therapy, the patient developed miliary tuberculosis with tuberculous pericarditis, complicated by life-threatening cardiac tamponade, within one year. This highlights that immunosuppression with TNF-α inhibitors can unmask latent tuberculosis infection or promote rapid disease progression.
Although guidelines recommend baseline screening for tuberculosis infection and disease before initiating TNF-α inhibitor therapy, recommendations regarding follow-up screening vary, and routine annual testing is not universally endorsed [2]. Moreover, IGRA has important limitations in immunocompromised hosts, including the possibility of false-negative results [1].
Pericardial tuberculosis is a rare condition with a mortality rate of 17–40% within 6 months after diagnosis [5]. It is often difficult to diagnose in the absence of pericardial fluid analysis. In this case, however, the diagnosis was confirmed by microbiological evidence obtained from the pericardial fluid, while additional positive sputum and stool samples further supported the diagnosis of miliary tuberculosis. Importantly, cardiac tamponade represents a medical emergency with the potential for rapid hemodynamic deterioration and sudden death if not promptly recognized and treated. Clinical presentation may be subtle, atypical, or even initially asymptomatic, and classical signs of tamponade are not always present, making early diagnosis particularly challenging. Delayed recognition and intervention in cardiac tamponade are associated with increased morbidity and mortality, highlighting the importance of early risk stratification and prompt pericardial drainage in suspected cases [6].
In addition to microbiological confirmation by nucleic acid amplification testing and culture, several adjunctive modalities may aid in the diagnosis of tuberculous pericarditis. In the present case, pericardial fluid was analyzed using smear microscopy (Ziehl–Neelsen and fluorescence staining), the Xpert MTB/RIF assay, and mycobacterial culture on both liquid and solid media, enabling direct microbiological confirmation of Mycobacterium tuberculosis. A recent network meta-analysis identified pericardial fluid adenosine deaminase (ADA) as a supportive diagnostic marker, although its performance varies compared with molecular assays and culture methods [7]. Additionally, pericardial biopsy with histopathological evaluation may enhance diagnostic yield when fluid-based investigations are negative [3]. Although neither ADA testing nor pericardial biopsy was required in this patient due to definitive microbiological evidence, these approaches remain important considerations in diagnostically challenging cases.
According to the World Health Organization guidelines for tuberculosis, adjuvant corticosteroids may be considered in cases of tuberculous pericarditis. However, this is a conditional recommendation supported by very low-certainty evidence [8]. The literature is mixed, with some trials showing benefit and others not, and corticosteroids are generally reserved for patients with a high risk of inflammatory complications such as constrictive pericarditis [9]. In this patient, the decision to initiate prednisolone alongside antituberculous therapy was justified by the high risk of developing constrictive pericarditis. She subsequently demonstrated gradual clinical and radiological improvement, with resolution of the pericardial effusion.
Long-term outcomes of tuberculous pericarditis remain clinically significant. Among patients with tuberculous pericarditis, approximately 10% experience transient constrictive physiology, whereas 20%–50% progress to established chronic constrictive pericarditis despite appropriate antituberculous treatment [10]. Accordingly, prolonged follow-up is warranted, as some patients may ultimately require surgical pericardiectomy [11]. Although relapse is uncommon with adequate treatment adherence, it should be considered in immunocompromised individuals [12].
Reintroduction of TNF-α inhibitor therapy requires careful multidisciplinary evaluation, balancing the risk of tuberculosis reactivation against the need for adequate immunosuppressive control of the underlying disease. The optimal timing for resuming TNF antagonist therapy in patients undergoing treatment for active tuberculosis remains uncertain. Current expert opinion recommends reinitiating TNF-α inhibitor therapy only after completion of a full course of anti-tuberculous treatment in accordance with international guidelines. However, the supporting clinical data are limited [13].
This case also highlights important practical considerations for follow-up monitoring during TNF-α inhibitor therapy. Although routine periodic IGRA testing is not universally recommended in low-incidence settings, robust data are lacking in defining the optimal frequency of re-screening. Therefore, periodic reassessment may be considered, particularly in patients with existing or newly emerging risk factors [14].
Patient education represents a key component of risk mitigation in individuals receiving biologic therapy. Patients should be informed about the risk of tuberculosis and advised to promptly report any new or unexplained symptoms, including persistent fever, night sweats, weight loss, cough, or dyspnea. Early recognition may reduce diagnostic delays, limit disease dissemination, and decrease the risk of severe complications and mortality.
Conclusions
The diagnosis of tuberculous pericarditis remains challenging due to its nonspecific clinical presentation and its rarity in low-incidence regions such as Europe. Although comprehensive screening for latent tuberculosis infection and active disease is mandatory prior to initiating biologic therapy, negative baseline results do not eliminate the risk of subsequent tuberculosis. Once immunosuppressive treatment is initiated, sustained clinical vigilance is imperative, and patients should be educated about the signs and symptoms of tuberculosis to facilitate early recognition and timely evaluation.
This case reinforces a critical clinical lesson: severe extrapulmonary tuberculosis, including life-threatening cardiac tamponade, may progress rapidly during TNF-α inhibitor therapy despite appropriate pre-treatment evaluation. Physicians managing patients receiving biologic agents must maintain a high index of suspicion for tuberculosis whenever unexplained systemic or respiratory symptoms arise, irrespective of prior screening results.
These findings underscore the limitations of baseline testing alone and support the need for ongoing risk assessment and individualized monitoring strategies during TNF-α inhibitor therapy. Early recognition and prompt intervention are essential to mitigate the risk of catastrophic complications and death.
The role of adjunctive corticosteroid therapy in tuberculous pericarditis remains debated. However, it may be justified in carefully selected high-risk patients, particularly those with features suggestive of impending constrictive pericarditis.
Conflicts of Interest
The authors declare that there are no competing interests.
Patient Consent
The patient has given informed consent.
References
2. Maeda T, Connolly M, Thevenet-Morrison K, Levy P, Utell M, Munsiff S, et al. Tuberculosis screening for patients on biologic Medications: A Single-Center experience and Society guideline Review, Monroe County, New York, 2018-2021. J Clin Tuberc Other Mycobact Dis. 2024 Jun 22;36:100460.
3. Adler Y, Charron P, Imazio M, Badano L, Barón-Esquivias G, Bogaert J, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015 Nov 7;36(42):2921–64.
4. Sartori NS, de Andrade NPB, da Silva Chakr RM. Incidence of tuberculosis in patients receiving anti-TNF therapy for rheumatic diseases: a systematic review. Clin Rheumatol. 2020 May;39(5):1439–47.
5. Mayosi BM, Wiysonge CS, Ntsekhe M, Gumedze F, Volmink JA, Maartens G, et al. Mortality in patients treated for tuberculous pericarditis in sub-Saharan Africa. S Afr Med J. 2008 Jan;98(1):36–40.
6. European Society of Cardiology. Cardiac tamponade: a clinical challenge [Internet]. Sophia Antipolis (FR): European Society of Cardiology; 2021 [cited 2026 Feb 12]. Available from: https://www.escardio.org/communities/councils/cardiology-practice/scientific-documents-and-publications/ejournal/volume-15/Cardiac-tamponade-a-clinical-challenge/.
7. Pervez A, Hasan SU, Hamza M, Asghar S, Qaiser MH, Zaidi S, et al. Diagnostic accuracy of tests for tuberculous pericarditis: A network meta-analysis. Indian J Tuberc. 2024 Apr;71(2):185–94.
8. World Health Organization. Consolidated guidelines on tuberculosis. Module 4: treatment – treatment and care. Chapter 1: drug-susceptible TB treatment. Recommendation 4: the use of adjuvant steroids in the treatment of TB meningitis and pericarditis [Internet]. Geneva: World Health Organization; 2022. Available from:https://tbksp.who.int/en/node/2965.
9. Chaisson RE, Post WS. Immunotherapy for tuberculous pericarditis. N Engl J Med. 2014 Sep 18;371(12):1155–7.
10. López-López JP, Posada-Martínez EL, Saldarriaga C, Wyss F, Ponte-Negretti CI, Alexander B, et al. Tuberculosis and the Heart. J Am Heart Assoc. 2021 Apr 6;10(7):e019435.
11. Krzelj K, Bakaeen M, Wang TKM, Klein A, Vega SLP, Jellis C, et al. Pericardiectomy for Constrictive and Recurrent Pericarditis: State of the Art Update. Curr Cardiol Rep. 2026 Jan 22;28(1):14.
12. Chaisson RE, Churchyard GJ. Recurrent tuberculosis: relapse, reinfection, and HIV. J Infect Dis. 2010 Mar;201(5):653–5.
13. Solovic I, Sester M, Gomez-Reino JJ, Rieder HL, Ehlers S, Milburn HJ, et al. The risk of tuberculosis related to tumour necrosis factor antagonist therapies: a TBNET consensus statement. Eur Respir J. 2010 Nov;36(5):1185–206.
14. Cantini F, Niccoli L, Capone A, Petrone L, Goletti D. Risk of tuberculosis reactivation associated with traditional disease modifying anti-rheumatic drugs and non-anti-tumor necrosis factor biologics in patients with rheumatic disorders and suggestion for clinical practice. Expert Opin Drug Saf. 2019 May;18(5):415–25.
