Abstract
Background: The aim of this study was to consider organism patterns and antimicrobial sensitivity over a 16-year period using brain-heart infusion broth, from a district general hospital in the south-west of the UK.
Methods: This was a retrospective review of microbiological data of all cornea scrapes performed at the Royal United Hospitals, Bath, over a period from 1st January 2006 to 31st December 2022. Both direct and enrichment cultures were used. Monotherapy with a fluoroquinolone antibiotic was used as first line throughout the study period.
Results: A total of 611 cornea scrapes (323/611, 52.9% culture positive) from 524 patients, were performed in the 16-year period. Organisms cultured were bacteria in 94% (68% gram-positive, 32% gram-negative), fungi in 4.4% and Acanthamoeba in 1.6% of cases. The most identified organism was Pseudomonas aeruginosa (n=70). There was a statistically significant rise in cases of Pseudomonas aeruginosa in 2022 compared to 2019 (13/37 vs 5/41 p-value 0.01). All gram-negative bacteria were sensitive to fluoroquinolone antibiotics. 96% of gram-positive bacteria were sensitive to fluoroquinolone antibiotics, the 4% that were resistant to this, were sensitive to vancomycin.
Conclusions: A fluoroquinolone antibiotic first line appears an appropriate agent in cases of gram-negative keratitis. It is vital, however, to consider antimicrobial sensitivities especially where a gram-positive organism is identified, to allow early switching to a second line agents in resistant cases. The rise in cases of Pseudomonas aeruginosa in recent years may warrant local public health measures to help increase local community awareness on contact lens hygiene.
Keywords
Microbial keratitis, Antimicrobial sensitivity, Pseudomonas aeruginosa
Introduction
Microbial keratitis is a common potentially sight-threatening acute corneal condition, most commonly occurring in contact lens wearers, especially those that may swim or shower with their contact lenses in place. Furthermore, patients with ocular surface diseases, such as keratoconjunctivitis sicca, neurotrophic keratopathy, surface disease and extended exposure to topical steroids, following anterior segment surgical procedures, for example, corneal transplantation, are at risk of this condition. Microbial keratitis remains a significant cause of visual loss worldwide [1]. Prompt diagnosis and treatment with topical antimicrobial therapy is vital to allow better recovery and visual prognosis [2]. As part of the investigations, a cornea scrape is performed to identify the responsible organism. This helps to tailor appropriate antimicrobial treatments to the responsible organism.
There have been several studies over recent years, within the UK which have considered trends in microbial keratitis and organisms involved [3–7]. These studies included the use of culture plates (blood agar plate, chocolate agar plate, Sabouraud’s agar plate and non-nutrient agar seeded with Escherichia coli for Acanthamoeba) [4–6] as well as brain–heart infusion (BHI) broth [3]. Brain–heart infusion (BHI) broth provides a pragmatic and equally efficacious method to perform corneal scrapes [8]. Part of the results of this current study have been included in the previous study looking at trends across an 11-year period within the south-west of the UK, where the results from this unit were combined with the results from the local tertiary referral unit Bristol Eye Hospital. However, given the demographics may vary between these two cities, as well as the potential differences in patients presenting at a tertiary referral unit and a district general hospital, we were interested to consider the organism pattern and microbial sensitivities solely within the Royal United Hospitals, Bath, over a 16-year period, to ensure we were offering appropriate first line treatment to our patients at this district general hospital.
The purpose of this study was thus to analyze the microbiological data from all corneal scrapes performed over a 16-year period from the Royal United Hospitals, Bath. The aims were to identify the organism patterns, trends over time and antimicrobial sensitivities.
Methods
This was a retrospective review of the microbiological data of all the cornea scrapes performed at the Royal United Hospitals, Bath in the UK, between the 1st of January 2006 to the 31st of December 2022, that were sent for microbiological analysis. Data collection included the age and gender of the patient, the date of the scrape, the organism identified from either direct or enrichment culture, and the antimicrobial sensitivities. Retrospective data collection was performed using data available on a microbiology database.
In this unit, corneal scraping was performed at the discretion of the reviewing clinician, this was typically done during the study period, where the corneal ulcer and infiltrate were either larger than 0.5 mm or atypical in appearance and not improving on treatment. This was either at presentation in the eye casualty clinic or in the corneal clinic as per the judgement of the reviewing ophthalmologist. Corneal samples were taken using a sterile scalpel blade or needle. The samples were then placed on slides for Gram staining and further samples were taken using a new scalpel blade or needle and immediately placed in 1 mL BHI broth in a sealed glass bottle, as described previously [3,8]. Microbiological analysis of the samples was done in routine manner as already described [3]. Results included both direct and enrichment cultures. Direct culture was for organisms that were grown on the initial plates used for analysis by microbiology. Enrichment culture included organisms that were grown once the BHI was incubated for a further 5 days. In vitro antibiotic sensitivity was performed by microbiology using either the disc diffusion method or the automated VITEK2 system (BioMeriux) method of testing. Where there was a clinical suspicion of acanthamoeba keratitis, corneal scrape samples were additionally sent separately for PCR to a reference laboratory in Glasgow.
To assess trends over time, the data set was separated into two-time frames, 1st January 2006 to 31st December 2013 and 1st January 2014 to 31st December 2022. Additionally, cases in the year 2019 (taken as pre-pandemic year) were compared to cases in the year 2022 (taken as post-pandemic year), to provide a further consideration for changes over time. Statistical significance between these groups was assessed using the Chi-squared test. A p value of <0.01 was taken as statistically significant.
Results
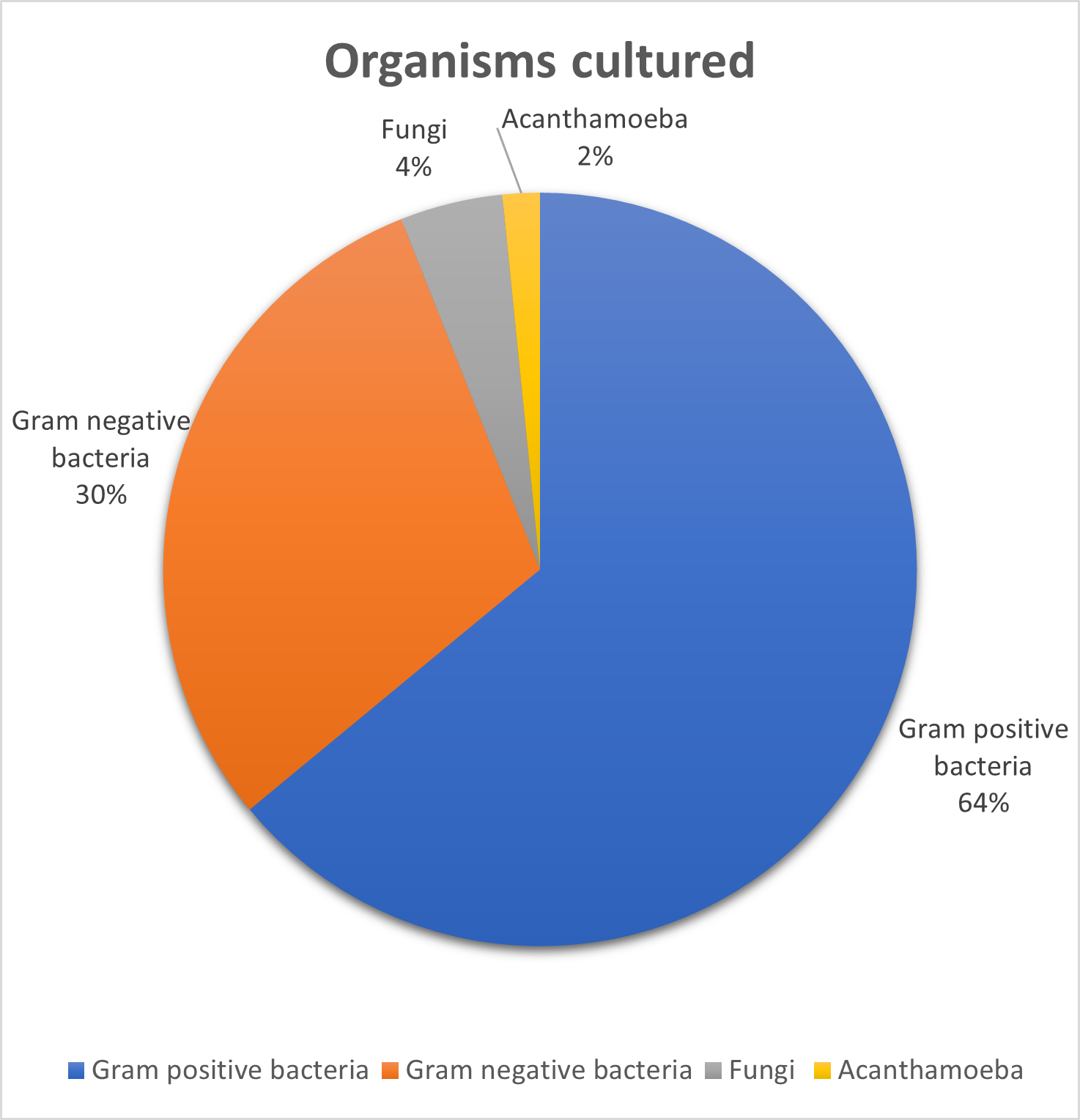
A total of 611 corneal scrapes, from 524 patients, were performed in the 16-year period. The mean age of the patients was 50.6 years, 57% were female and 43% were male. Of the 611 corneal scrapes, an organism was cultured in a total of 323, giving a culture positive rate of 52.9%. Organism groups cultured were bacteria in 304 cases, 94% (of which 207 cases, 68% were gram-positive, 97 cases, 32% were gram-negative), fungi in 14 cases, 4.4% and acanthamoeba in 5 cases, 1.6% of cases (Figure 1). Table 1 shows a summary of the organisms cultured. The most frequently identified organism during this period was Pseudomonas aeruginosa (n=70).
|
Organism |
Numbers |
|
Streptococci |
18 |
|
Staphylococcus aureus |
21 |
|
Staphylococcus epidermidis |
41 |
|
Coagulase negative staphylococci |
58 |
|
Other G+ve |
69 |
|
Pseudomonas |
70 |
|
Moraxella |
14 |
|
Other G-ve |
13 |
|
Fungi |
14 |
|
Acanthamoeba |
5 |
|
Total culture positive |
323 |
Figure 1. Summary of the organism groups cultured over the 16-year study period in Bath. Gram-positive bacteria were the most frequently cultured group of organisms, followed by gram-negative bacteria, fungi and Acanthamoeba.
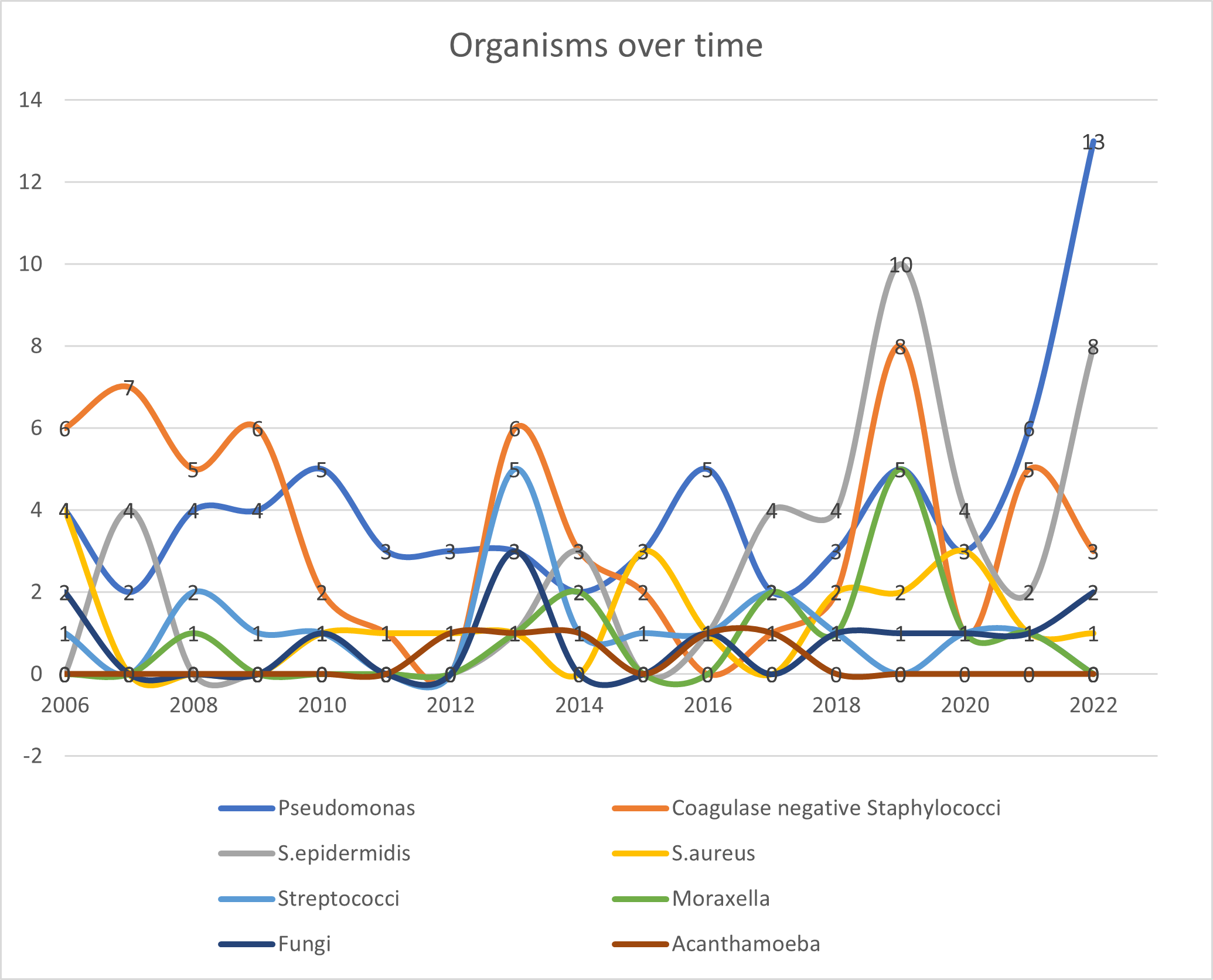
When considering trends over time, there was a statistically significant rise in cases of Pseudomonas aeruginosa in 2022 compared to 2019 (13/37 vs 5/41 p-value 0.01) (Figure 2). There were no further statistically significant differences in the numbers of individual organisms cultured between 2019 and 2022. When considering differences over the first half of the study period (2006–2013) and the second half of the study period (2014–2022), there was a significant rise in Staphylococcal epidermidis (5/115 vs 36/208 p=0.0008) and a significant decline in Coagulase negative staphylococci (33/115 vs 22/208 p= 0.000034) (Figure 3). No further statistical significance in organism trends was identified between these two study periods. When considering antimicrobial sensitivities, all gram-negative bacteria showed in vitro sensitivity to ciprofloxacin which was the fluoroquinolone used to assess in-vitro sensitivity. Of the gram-positive bacteria, 96% showed in vitro sensitivity to ciprofloxacin. Four percent (8 cases) of gram-positive bacteria that were resistant to ciprofloxacin. The eight ciprofloxacin resistant organisms were identified as Staphylococcal epidermidis in five cases (12% of all the Staphylococcal epidermidis cases were thus identified as showing in vitro resistance to the fluoroquinolone ciprofloxacin), one case of Corynebacterium amycolatum, one case of Kocuria rhizophilia and one case of Actinomyces sp. Seven of these eight cases showed in vitro sensitivity to vancomycin, in the additional case (Kocuria rhizophilia) vancomycin sensitivity was not assessed or reported.
Figure 2. Pseudomonas aeruginosa cases over time. There was a statistically significant greater number of cases in 2022 compared to 2019.
Figure 3. The numbers of organisms identified over time. There was a rise in Pseudomonas aeruginosa in 2019 vs 2022 (5/41 vs 13/37 p-value 0.01). When comparing 2006–2013 to 2014–2022, there was a statistically significant rise in cases of S. epidermidis (5/115 vs 36/208 p=0.0008), and there was a statistically significant decline in cases of Coagulase negative staphylococci (33/115 vs 22/208 p=0.000034).
Discussion
This is the first large scale study analyzing microbiology and trends in corneal scrapes performed over a 16-year period at the Royal United Hospitals Bath. The overall proportion of culture positive samples over the 16-years analyzed in this study was 52.9%, which although higher than other recent reports, remains comparable to that from previous reports in the UK (32.6% [6], 33% [8], 38.1% [3], 44.5% [5]). In this unit, over the study period BHI was used as the collection method for corneal scrapes and this method provides an efficient and efficacious way for corneal scrape analysis [3,8]. Reports suggest that microbiological analysis of microbial keratitis using impression cytology may yield an even higher culture positivity rate, which has been reported as 65.2% [9]. Of note, a previous study considered part of this data set from 4th April 2006 to 31st October 2017 but combined it with the data from the local tertiary referral unit Bristol Eye Hospital [3]. Our current study considers a larger time scale of 16-years (from 1st January 2006 to 31st December 2022) and given the demographics varies between these two cities, as well as the likely differences that exist in the patient cohort between the tertiary referral unit and district general hospital, this present study was vital to provide additional data to consider local organisms, patterns and trends in microbial keratitis cases.
In this study, gram-positive bacteria were the most frequently cultured group of organisms (Figure 1). However, when considering the most frequently cultured organism overall, this was found to be the gram-negative organism Pseudomonas aeruginosa (Table 1). As seen in Figure 2, there was a rise in cases of Pseudomonas aeruginosa in the year 2022, and this showed statistical significance when compared to the year 2019. Although the cause of this is unclear, these years may signify post pandemic and lockdowns vs pre pandemic in the UK. Given that cases of microbial keratitis secondary to the organism Pseudomonas aeruginosa occur more frequently in contact lens wearers, it is possible that patterns of local contact lens hygiene varied post pandemic. Altered healthcare access, or health-seeking behavior following the pandemic may also be a potential contributing factor. However, since this study did not consider clinical data or specific patient risk factors, one can only speculate the clinical cause for this rise. No further trends or differences were noted in the pattern of organisms when considering 2022 vs 2019. Interestingly, a recent study from the Manchester group did not identify any differences in incidence or characteristics of microbial keratitis in the year after lockdown [10]. In view of the apparent rise in cases of Pseudomonas aeruginosa keratitis seen in our study in 2022, public health measures were taken in Bath in the summer of 2023, to help increase local community awareness on contact lens hygiene. This included a radio interview with BBC somerset, a media release from the Royal United Hospitals, an article in the local paper and a social media video released on the hospital Instagram page (see supplementary material). Preliminary data and clinical observations from the remainder of 2023 and the early parts of 2024, revealed no further cases of Pseudomonas aeruginosa picked up on corneal scraping locally, however, further data analysis and future studies will be required to assess if these measures provided any significant impact. Nonetheless, this suggests that local public health measures may be useful to help raise public awareness on contact lens hygiene and risk factors for microbial keratitis.
When further considering trends over time, as shown in Figure 3, comparisons of the first half of the study period (2006–2013) and the second half of the study period (2014–2022), identified a significant rise in Staphylococcal epidermidis (5/115 vs 36/208 p=0.0008) and a significant decline in Coagulase negative staphylococci (33/115 vs 22/208 p= 0.000034). No further statistical significance in organism trends was identified between these two study periods. When comparing our data to that from other studies in the UK, gram-positive bacteria have also been reported as the most common isolates in other studies from the UK [3–8]. As per our study, Pseudomonas species were found to be the most frequently occurring organism overall in a previous report from the East of England [4], as well as in Taiwan [11], China [12]; South Florida [13]. This contrasts with several studies in the UK, that have found coagulase negative staphylococci to be the most frequently identified organism [3,5,6,7]. This has been with an increasing trend in the Sunderland and Oxford series [5,7] and a decreasing trend in the Manchester series [6], and with no trend in the previous South West of the UK series [3]. Coagulase negative staphylococci were also identified as the most common isolates in studies from India [14] and Saudi Arabia [15]. These findings further support the notion that variations do exist geographically [16], hence the need to identify local patterns of microbial keratitis.
In vitro sensitivity testing confirmed monotherapy with a fluoroquinolone antibiotic, to be a reasonable first line agent, with excellent cover against gram-negative organisms (100% sensitivity). When considering gram-positive organism cover, in vitro sensitivity suggested fluroquinolones to be sensitive in 96% of cases. Thus, our first line agents seem highly reasonable, especially against gram-negative organisms. This is consistent with other reports from the UK where monotherapy with a fluoroquinolone is considered a reasonable first line choice [3,5,6]. It is important to emphasize that although in vitro sensitivity is considered a useful guide to its clinical response [17], the high intensity at which antibiotics are given topically in clinical practice (initially hourly day and night for 48-hours) may achieve a sufficiently high concentration to yield a bactericidal effect and thus result in a higher rate of clinical sensitivity compared to that seen with in vitro testing. Furthermore, these parameters are based on systemic breakpoint criteria rather than ophthalmic breakpoints [18], the response seen in the serum is likely different to that achieved in the cornea and aqueous humor following topical application [19]. Of note, in vitro sensitivity was not tested or reported for all antimicrobial agents. When considering fluoroquinolones, ciprofloxacin sensitivity was assessed by the microbiology laboratory as a class surrogate for the fluoroquinolones used in this unit. Thus, although clinically ciprofloxacin was not used in this unit, levofloxacin 0.5% or ofloxacin 0.3% drops were typically used first line, the reported in vitro sensitivity to ciprofloxacin was considered the same as that for the fluoroquinolone drops used in the unit. A limitation may be in varied clinical sensitivity to the individual fluoroquinolones. Furthermore, minimum inhibitory concentration values were not provided routinely in microbiological reports.
In this unit, second line agents may include dual therapy with cefuroxime 5% and gentamicin 1.5%. Unfortunately, data on in vitro sensitivity to cefuroxime was not available for our resistant cases, as this was not tested in the laboratory for all the cases. A further option in such cases, may be topical vancomycin which showed in vitro sensitivity to seven of the eight ciprofloxacin resistant organisms (in the case of the Kocuria rhizophilia keratitis, in vitro sensitivity to vancomycin was not reported). Interestingly, five of the ciprofloxacin resistant cases were those of Staphylococcal epidermidis, meaning that 12% of all the Staphylococcal epidermidis cases had in vitro resistance to the fluoroquinolone ciprofloxacin. Thus, early recognition of poor clinical efficacy is crucial when considering microbial keratitis, secondary to gram-positive organisms and in particular Staphylococcal epidermidis, with a low threshold and early request to add a second line agent. This is of particular importance in the setting of a district general hospital, where not all topical antibiotic medications are readily available at the in-house pharmacy, and ordering these may take several days.
The main limitation of our study is the lack of clinical data. In clinical practice, a great deal of information is provided by clinical history and examination, with certain risk factors, such as contact lens use and poor contact lens hygiene, making certain organisms more likely. Furthermore, during the study period in cases where there was a clinical suspicion for Acanthamoeba keratitis, PCR testing was also carried out, as well as requesting confocal microscopy from either Moorfields Eye Hospital, or more recently from Bristol Eye Hospital. The results of our study, however, only include the numbers of Acanthamoeba keratitis cases isolated by culture alone, and thus the total number of Acanthamoeba cases seen during the study period are likely to have been underestimated. Due to the nature of the study, the results include only ulcers that were scraped, rather than all corneal ulcers that presented to this unit. Our sample sizes were also relatively low, when considering the apparent rise in Pseudomonas aeruginosa cases in 2022 compared to 2019, thus limiting the robustness of our statistical analysis, meaning this apparent rise should be interpreted cautiously.
Conclusion
In conclusion, our study provides a 16-year review of all cases of corneal scrapes performed within a single-unit district general hospital setting and supports the notion that identifying local geographical trends in organism pattern can be helpful. Despite the discussed limitations, in particular the retrospective nature of the study and the relatively small sample size, the information identified is helpful locally. A fluoroquinolone antibiotic first line remains an appropriate agent in cases of gram-negative keratitis. It is vital to consider antimicrobial sensitivities especially where a gram-positive organism is identified, to allow early switching to a second line agent in resistant cases. The rise in cases of pseudomonal keratitis in the final year of the study period warranted local public health measures to help educate the local community and raise awareness on contact lens hygiene.
Acknowledgments
Sabrina Fudge and the Microbiology department at Royal United Hospitals, NHS trust.
Paul North for providing the data. Fardad T Afshari for statistical analysis. Scottish Parasite Diagnostic Reference Section, Scottish Microbiology Reference Laboratories, Glasgow for Acanthamoeba PCR. Moorfields Eye Hospital and Bristol Eye Hospital for confocal microscopy of Acanthamoeba suspects.
References
2. Allan BD, Dart JK. Strategies for the management of microbial keratitis. Br J Ophthalmol. 1995 Aug;79(8):777–86.
3. Tavassoli S, Nayar G, Darcy K, Grzeda M, Luck J, Williams OM, et al. An 11-year analysis of microbial keratitis in the South West of England using brain-heart infusion broth. Eye (Lond). 2019 Oct;33(10):1619–25.
4. Moledina M, Roberts HW, Mukherjee A, Spokes D, Pimenides D, Stephenson C, et al. Analysis of microbial keratitis incidence, isolates and in-vitro antimicrobial susceptibility in the East of England: a 6-year study. Eye (Lond). 2023 Sep;37(13):2716–22.
5. Ting DSJ, Settle C, Morgan SJ, Baylis O, Ghosh S. A 10-year analysis of microbiological profiles of microbial keratitis: the North East England Study. Eye (Lond). 2018 Aug;32(8):1416–7.
6. Tan SZ, Walkden A, Au L, Fullwood C, Hamilton A, Qamruddin A, et al. Twelve-year analysis of microbial keratitis trends at a UK tertiary hospital. Eye (Lond). 2017 Aug;31(8):1229–36.
7. Orlans HO, Hornby SJ, Bowler IC. In vitro antibiotic susceptibility patterns of bacterial keratitis isolates in Oxford, UK: a 10-year review. Eye (Lond). 2011 Apr;25(4):489–93.
8. Kaye SB, Rao PG, Smith G, Scott JA, Hoyles S, Morton CE, Willoughby C, Batterbury M, Harvey G. Simplifying collection of corneal specimens in cases of suspected bacterial keratitis. J Clin Microbiol. 2003 Jul;41(7):3192–7.
9. Somerville TF, Herbert R, Neal T, Horsburgh M, Kaye SB. An Evaluation of a Simplified Impression Membrane Sampling Method for the Diagnosis of Microbial Keratitis. J Clin Med. 2021 Nov 30;10(23):5671.
10. Michaels L, Richardson J, Walkden A, Carley F. Impact of the COVID-19 Pandemic on the Incidence and Characteristics of Culture-Positive Microbial Keratitis at a Tertiary Eye Hospital in the UK. Clin Ophthalmol. 2022 Aug 10;16:2513–9.
11. Hsiao CH, Sun CC, Yeh LK, Ma DH, Chen PY, Lin HC, Tan HY, Chen HC, Chen SY, Huang YC. Shifting Trends in Bacterial Keratitis in Taiwan: A 10-Year Review in a Tertiary-Care Hospital. Cornea. 2016 Mar;35(3):313–7.
12. Zhang C, Liang Y, Deng S, Wang Z, Li R, Sun X. Distribution of bacterial keratitis and emerging resistance to antibiotics in China from 2001 to 2004. Clin Ophthalmol. 2008 Sep;2(3):575–9.
13. Alexandrakis G, Alfonso EC, Miller D. Shifting trends in bacterial keratitis in south Florida and emerging resistance to fluoroquinolones. Ophthalmology. 2000 Aug;107(8):1497–502.
14. Gopinathan U, Sharma S, Garg P, Rao GN. Review of epidemiological features, microbiological diagnosis and treatment outcome of microbial keratitis: experience of over a decade. Indian J Ophthalmol. 2009 Jul-Aug;57(4):273–9.
15. Al-Dhaheri HS, Al-Tamimi MD, Khandekar RB, Khan M, Stone DU. Ocular Pathogens and Antibiotic Sensitivity in Bacterial Keratitis Isolates at King Khaled Eye Specialist Hospital, 2011 to 2014. Cornea. 2016 Jun;35(6):789–94.
16. Shah A, Sachdev A, Coggon D, Hossain P. Geographic variations in microbial keratitis: an analysis of the peer-reviewed literature. Br J Ophthalmol. 2011 Jun;95(6):762–7.
17. Wilhelmus KR, Abshire RL, Schlech BA. Influence of fluoroquinolone susceptibility on the therapeutic response of fluoroquinolone-treated bacterial keratitis. Arch Ophthalmol. 2003 Sep;121(9):1229–33.
18. Kaye S, Tuft S, Neal T, Tole D, Leeming J, Figueiredo F, Armstrong M, McDonnell P, Tullo A, Parry C. Bacterial susceptibility to topical antimicrobials and clinical outcome in bacterial keratitis. Invest Ophthalmol Vis Sci. 2010 Jan;51(1):362–8.
19. Kaye S. Microbial keratitis and the selection of topical antimicrobials. BMJ Open Ophthalmol. 2017 May 24;1(1):e000086.