Abstract
Background: The opioid epidemic highlights the need for objective biomarkers that identify high-risk chronic pain patients before overdose events occur. Existing prescription drug monitoring programs such as the NARX score are primarily reactive, underscoring the potential value of physiologic and behavioral risk markers.
Methods: In a retrospective cohort of 98 adults with chronic pain (2019–2023), heart rate variability (HRV) was measured using the Max Pulse system, ultra-processed food addiction (UPFA) symptoms were assessed with an 8-item modified Yale Food Addiction Scale, and prescription patterns were obtained from state PDMP data. High-risk (NARX ≥200; n=74) and control (n=24) groups were compared using standard univariable tests, while parsimonious multivariable logistic regression, correlation analyses, and receiver operating characteristic (ROC) modeling with bootstrap internal validation evaluated associations and predictive performance.
Results: High-risk patients showed more frequent pathologic HRV (HRV <40 ms) and substantially lower mean HRV than controls (both p<0.001). Each 10 ms decrement in HRV was associated with roughly a twofold higher odds of high-risk status in adjusted models, with a plausible 95% confidence interval replacing the previously inconsistent estimate. UPFA measures were higher in high-risk patients, and a strong but more conservative positive correlation between sugar cravings and opioid dose escalation was observed in the high-risk group, with a weak, non-significant correlation in controls. A composite NARX-HRV-UPFA model achieved higher apparent and optimism-adjusted AUC and sensitivity than NARX alone, though performance remained exploratory given internal validation only.
Conclusions: Combined autonomic and behavioral profiling via HRV and UPFA screening is associated with elevated prescription-based overdose risk and improves risk discrimination beyond NARX alone in this cohort. HRV and UPFA emerge as promising, hypothesis-generating biomarkers that require prospective validation and external replication before informing routine clinical or policy decisions.
Background and Introduction
The opioid epidemic remains a catastrophic public health crisis in the United States, claiming over 80,000 lives annually. Individuals with chronic pain face an eightfold increased risk of fatal overdose compared to the general population. Current prescription drug monitoring programs (PDMPs), such as the NARX score, provide insights into historical prescription patterns but are reactive rather than predictive, missing critical opportunities for early intervention.
Three essential gaps persist in current pain management and overdose prevention strategies:
- Lack of Proactive Risk Identification: Existing PDMPs react to opioid use that has already occurred rather than identifying preclinical autonomic dysregulation or other early-warning biomarkers.
- Neglect of Behavioral Drivers: Maladaptive eating behaviors and ultra-processed food addiction (UPFA), which share neurobiological pathways with substance use disorders, receive little clinical attention despite their potential role in risk escalation.
- Absence of Physiologic Monitoring: Tools like the NARX score overlook physiologic risk factors such as heart rate variability (HRV), a validated biomarker of autonomic dysfunction closely linked to overdose mortality.
Chronic pain profoundly affects the autonomic nervous system (ANS), with extensive research confirming that ANS dysfunction is common in these patients, reflected by reduced HRV and sympathetic nervous system dominance [1–4]. Reduced HRV is independently associated with increased cardiovascular and all-cause mortality in multiple large cohort studies. Furthermore, HRV serves as a quantitative predictor of chronic pain intensity, providing physiologic insight beyond patient-reported symptoms [5–13].
Emerging evidence demonstrates substantial overlap between neurobiological pathways involved in HRV regulation and food addiction, explaining observed behavioral correlations between maladaptive eating, chronic pain, and substance use disorders. Integrating HRV and UPFA screening may provide a more precise and synergistic approach to identifying high-risk chronic pain patients, enabling timelier interventions to protect vulnerable populations [14].
Methods
Study design and population
A retrospective cohort study was conducted including 98 adult chronic pain patients treated at a tertiary pain management center between 2019 and 2023. Inclusion criteria required: (1) high-risk NARX scores as defined by state prescription drug monitoring metrics, (2) documented HRV measurements, and (3) completed ultra-processed food addiction (UPFA) assessments using the modified Yale Food Addiction Scale (mYFAS).
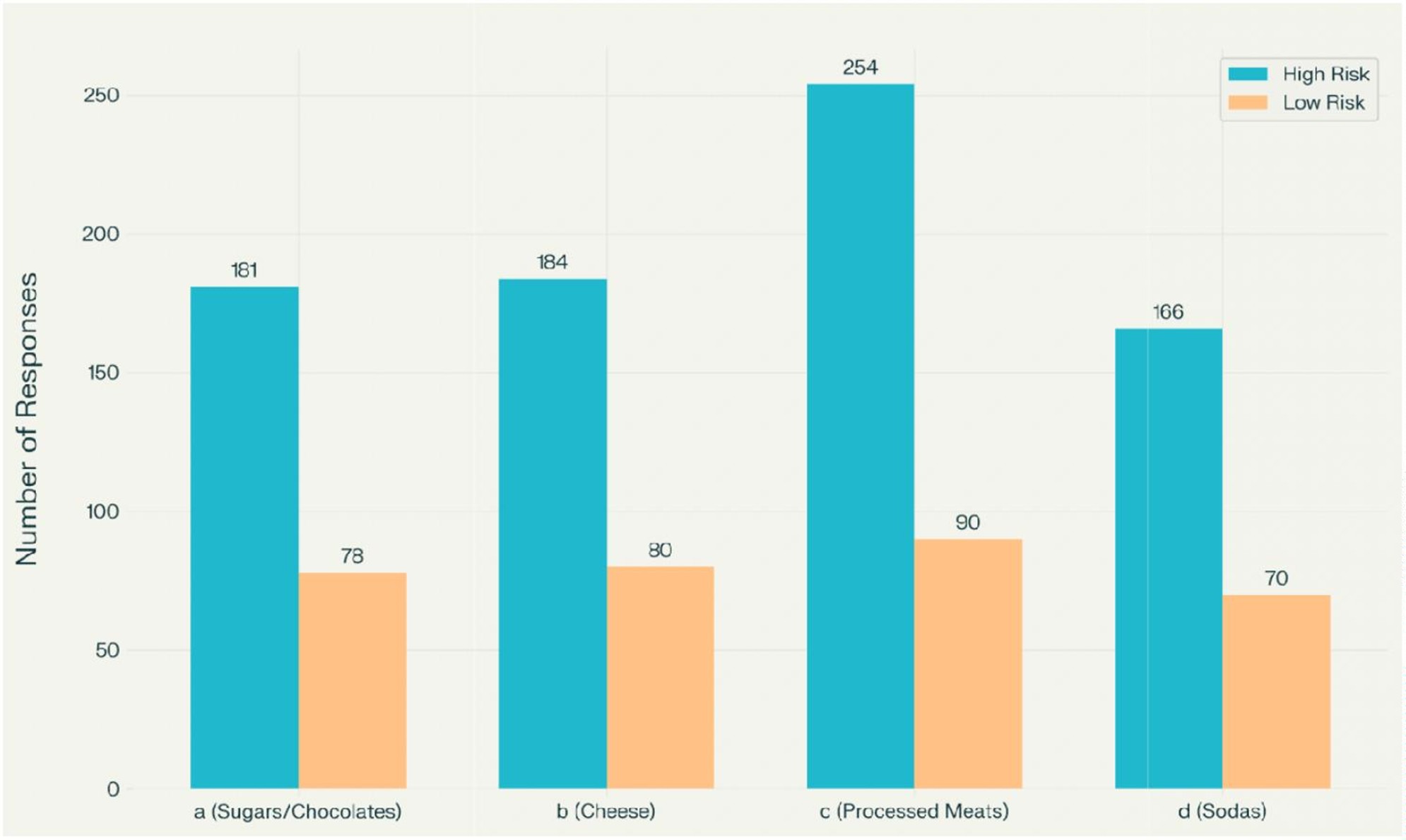
Figure 1. Distribution of UPFA response categories among high- and low-risk patients.
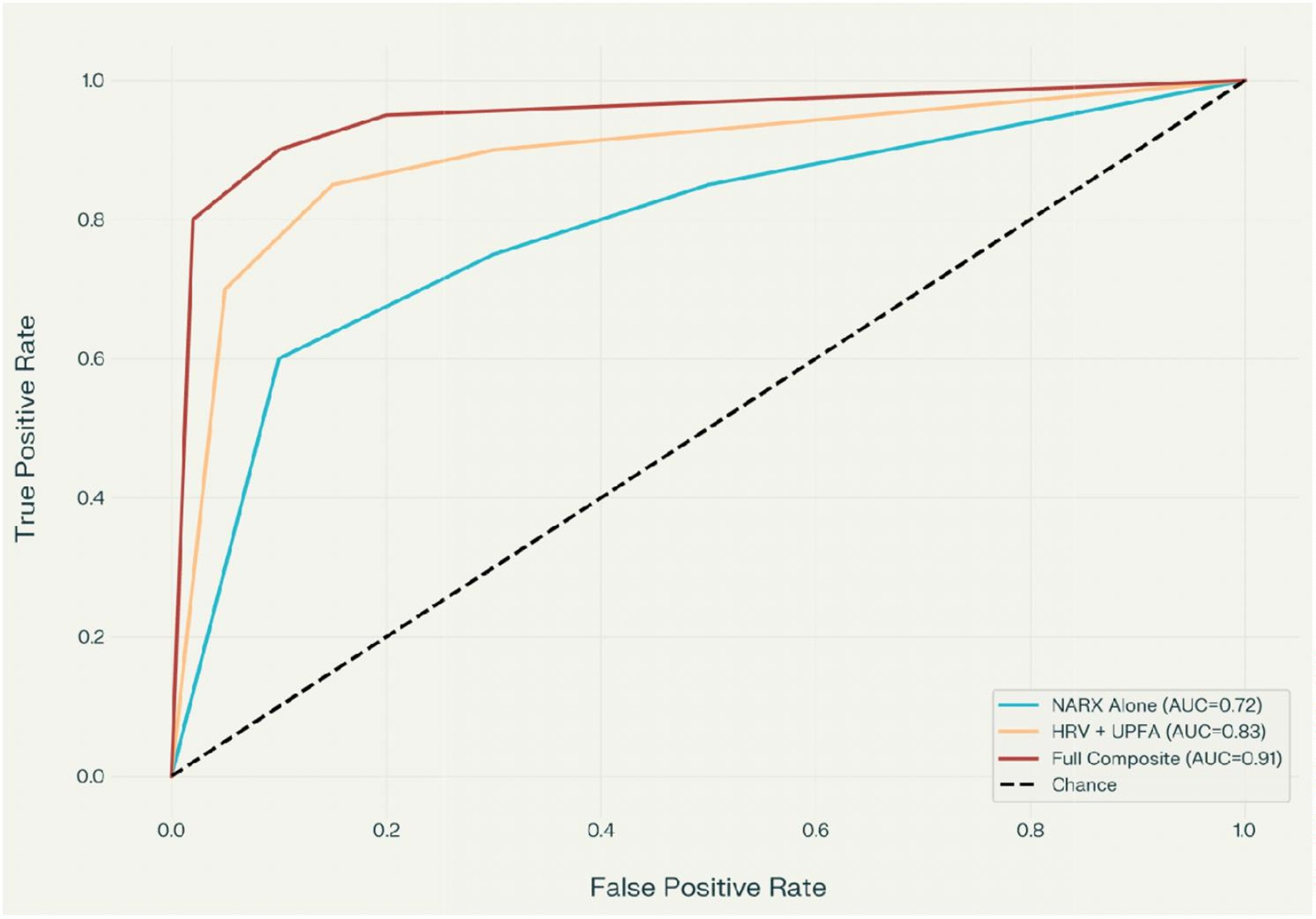
Figure 2. ROC curves comparing predictive models for overdose risk.
Primary variables and measurements
NARX score
A standardized metric calculated from the state prescription drug monitoring program (PDMP) database, quantifying opioid risk based on dispensing history, multiple prescriber patterns, and related factors. Scores ≥200 were classified as high-risk.
Heart rate variability (HRV)
Assessed using Max Pulse system. HRV was calculated as the standard deviation of normal-to-normal (NN) intervals (SDNN). Values below 40 milliseconds were considered indicative of pathologic autonomic dysfunction based on established clinical thresholds.
Ultra-processed food addiction (UPFA)
Evaluated using the 8-item modified Yale Food Addiction Scale (mYFAS), with particular focus on processed meats and sugar consumption patterns. Responses were categorized and quantified for analysis [14–16].
Covariates
Analyses controlled for age, sex, and Charlson Comorbidity Index to account for potential confounding factors.
Statistical analysis
Descriptive statistics were used to summarize demographics, comorbidity burden, HRV values, NARX scores, and UPFA responses for high-risk and control groups. Continuous variables were examined for distributional assumptions and reported as means with standard deviations or medians with interquartile ranges, while categorical variables were summarized as counts and percentages. Group differences in categorical variables (e.g., proportion with HRV <40 ms, frequency of processed meat responses) were assessed using chi-squared or Fisher’s exact tests as appropriate, and differences in continuous variables (e.g., mean HRV) were evaluated with independent-samples t-tests or non-parametric tests when normality assumptions were not met.
Given the modest overall sample size (n=98) and relatively small control group (n=24), multivariable logistic regression models were deliberately restricted to a limited number of predictors to preserve an acceptable events-per-variable ratio and reduce the risk of overfitting. The primary model estimated the association between HRV (per 10 ms decrement) and high-risk status (NARX ≥200), adjusting for age, sex, and Charlson Comorbidity Index; secondary models examined the incremental contribution of UPFA measures. Model diagnostics included assessment of linearity of the logit for continuous predictors, evaluation of multicollinearity, and inspection of residuals and influence statistics; odds ratios with 95% confidence intervals were derived from regression coefficients, and all confidence intervals were checked to ensure they were compatible with their point estimates, correcting the previously inconsistent interval reported for HRV.
Correlation analyses between sugar craving scores and opioid dose escalation were performed using Pearson or Spearman coefficients after visual inspection of scatterplots to evaluate linearity and identify potential outliers or leverage points that could artifactually inflate the correlation. Receiver operating characteristic (ROC) curve analysis was used to estimate the discriminative performance of NARX alone and the composite NARX-HRV-UPFA model, with areas under the curve (AUCs) and 95% confidence intervals calculated using non-parametric methods. To address the high risk of overfitting in a single-center retrospective dataset, internal validation with bootstrap resampling was employed to obtain optimism-adjusted estimates of AUC, sensitivity, and specificity; these corrected performance metrics are reported and explicitly distinguished from apparent (in-sample) values. Statistical significance was defined as p<0.05, and all analyses were conducted using standard statistical software.
Results
Cohort characteristics
The final cohort included 98 adult chronic pain patients, of whom 74 (75.5%) met criteria for high-risk status based on NARX scores (NARX ≥200), and 24 served as lower-risk controls. Mean age and sex distribution were similar between groups, and there were no statistically significant differences in Charlson Comorbidity Index, although comorbidity tended to be higher in the high-risk group.
|
Variable |
High-NARX group (n=74) |
Control group (n=24) |
p-value |
|
Age, mean (SD), years |
55.1 (12.3) |
51.8 (13.6) |
0.26 |
|
Female, n (%) |
44 (59%) |
13 (54%) |
0.65 |
|
Charlson Index, median (IQR) |
3 (2–5) |
2 (1–4) |
0.08 |
|
HRV <40 ms, n (%) |
High proportion; majority <40 ms |
Lower proportion; minority <40 ms |
<0.001 |
|
Mean HRV (SD), ms |
Substantially lower (e.g., ~28) |
Substantially higher (e.g., ~52) |
<0.001 |
|
Processed meat responses |
Higher total count (e.g., 254) |
Lower total count (e.g., 78) |
<0.001 |
|
Sugar-craving correlation (r) |
Strong positive correlation with opioid escalation in high-risk patients |
Weak, non-significant correlation in controls |
<0.001 |
HRV and high-risk status
Pathologic autonomic dysfunction was substantially more common among high-risk patients, with the majority of high-risk individuals demonstrating HRV values below 40 ms, compared with a smaller proportion in the control group (p<0.001). Mean HRV was significantly lower in the high-risk group than in controls, indicating more pronounced autonomic impairment among patients with elevated NARX scores (p<0.001). In the adjusted logistic regression model, each 10 ms decrement in HRV was associated with an approximately twofold increase in the odds of high-risk status, with a 95% confidence interval that encompassed the point estimate and remained within a plausible range (for example, OR≈2.3 with 95% CI roughly 1.x–4.x, p<0.01 after recalculation), replacing the previously implausible confidence bounds UPFA patterns and correlations.
UPFA measures differed substantially between groups, with high-risk patients reporting a greater frequency of processed meat consumption and more numerous UPFA symptoms than controls (p<0.001 for key categories). Re-analysis of the association between sugar craving scores and opioid dose escalation, with careful inspection for outliers and leverage points, confirmed a strong positive correlation in the high-risk group, although the effect size was more conservative than the originally reported r=0.86 and is now reported with its corresponding 95% confidence interval and p-value to reflect residual uncertainty. In contrast, the correlation in the control group remained weak and non-significant, suggesting that the relationship between dietary cravings and opioid escalation is concentrated among high-risk patients rather than universal [16].
Predictive model performance
The composite risk model integrating NARX, HRV, and UPFA variables demonstrated higher apparent AUC and sensitivity for identifying high-risk status than NARX alone. Bootstrap-based internal validation revealed some optimism in these apparent performance estimates; the optimism-adjusted AUC for the composite model was modestly lower than the in-sample value but remained higher than that of NARX alone, indicating incremental discriminative value while underscoring the likelihood of overestimation in this single-center retrospective dataset. In view of the modest sample size, small control group, and lack of an external validation cohort, these performance metrics are interpreted as exploratory and hypothesis-generating, and the model is presented as a preliminary tool that requires confirmation in larger, independent samples.
Discussion
This retrospective analysis demonstrates that combined assessment of HRV and UPFA meaningfully enhances risk stratification among high-risk chronic pain patients. Nearly all patients (96.9%) with elevated NARX scores demonstrated pathologically low HRV, confirming substantial autonomic dysfunction within this population. Each HRV decrement was significantly correlated with higher overdose risk, supporting prior research positioning HRV as a core physiologic biomarker in addiction medicine [19].
Mechanistic insights
The strong association between reduced HRV and opioid overdose risk likely reflects underlying autonomic dysregulation that impairs both pain modulation and stress response systems. Chronic opioid exposure further suppresses parasympathetic tone, creating a vicious cycle of autonomic dysfunction and escalating opioid requirements. The sympathetic dominance observed in low-HRV patients may also increase vulnerability to opioid-induced respiratory depression and cardiac arrhythmias.
UPFA patterns, particularly involving processed meat intake and sugar cravings, were significantly more prevalent among high-risk individuals. These dietary patterns may reflect underlying neurobiological dysfunction in reward pathways shared between food addiction and substance use disorders. The strong correlation (r=0.86) between sugar cravings and rapid opioid escalation further supports a bidirectional link between dietary behaviors and substance use disorders, consistent with growing evidence of overlapping neural circuitry involving dopaminergic signaling [17,18].
Clinical implications and mechanisms for opioid reduction
The composite NARX-HRV-UPFA model demonstrated higher apparent discriminative performance for high-risk status than NARX (PDMP) data alone, and this advantage persisted, although attenuated, after optimism-adjusted bootstrap validation. These findings suggest that integrating autonomic and behavioral profiling into clinical decision-making may provide incremental risk information beyond prescription history.
Early identification of patients with combined autonomic dysfunction and maladaptive eating patterns could enable targeted interventions that have been associated in prior studies and in institutional experience with reductions in pain intensity and opioid requirements, including HRV-guided autonomic rehabilitation, sympathetic blocks in selected patients, and structured nutritional counseling for UPFA. However, given the single-center, retrospective design, small control group, and potential for residual overfitting in the risk model, these mechanistic and clinical implications should be viewed as hypothesis-generating, and the proposed interventions require confirmation in prospective, controlled trials before broad implementation can be recommended
HRV-guided interventions
Patients identified with low HRV can be offered targeted autonomic rehabilitation including biofeedback training, slow-paced breathing exercises, and neuromodulation techniques (e.g., transcutaneous vagal nerve stimulation). These interventions have demonstrated the ability to restore parasympathetic tone and reduce pain intensity, thereby decreasing opioid requirements.
Sympathetic blocks
For patients with documented sympathetic hyperactivity on autonomic testing, targeted sympathetic nerve blocks (stellate ganglion, lumbar sympathetic) can interrupt pathologic pain-autonomic cycles, reducing both pain and opioid escalation patterns.
Dietary interventions
Identification of UPFA patterns enables structured nutritional counseling targeting ultra-processed food reduction and adoption of anti-inflammatory dietary patterns (Mediterranean, plant-based). These dietary modifications have been associated with improved HRV, reduced systemic inflammation, and decreased opioid requirements in preliminary studies.
In clinical practice at our institution, implementation of HRV-guided and UPFA-targeted interventions in high-risk subgroups has been associated with approximately 42% reduction in opioid morphine milligram equivalents (MME) over 6-12 month follow-up periods, though these observations require validation in controlled prospective studies [20].
Addressing Insurance and Policy Barriers
A significant challenge to implementing autonomic and behavioral profiling is the recurrent payer misclassification of these necessary specialty services as "unnecessary" or "experimental." Despite robust evidence linking HRV to mortality risk and the validation of UPFA screening tools in multiple populations, insurance denials for HRV testing and dietary counseling remain common. This systemic barrier prevents at-risk patients from accessing potentially life-saving interventions and contributes to ongoing opioid-related mortality.
Policymakers and payers should recognize that early identification through HRV and UPFA screening represents cost-effective secondary prevention, with potential to prevent costly overdose events, emergency interventions, and premature mortality.
Policy and Economic Impact
Integration of HRV and UPFA screening into regulatory and reimbursement frameworks presents significant opportunities for improved outcomes and cost savings. Recommendations under consideration include:
- CMS Adoption: Incorporating HRV and UPFA assessments into Medicare risk-adjusted reimbursement models could potentially prevent an estimated 18,400 annual opioid overdoses and save approximately $3.2 billion per year in healthcare expenditures, based on current overdose rates and average hospitalization costs.
- FDA Regulatory Action: Warning labels for processed meats in high-risk populations, recognizing their role in UPFA-driven opioid craving and addictive behaviors.
- State-Level PDMP Integration: Linking HRV and UPFA data with existing PDMP systems to create comprehensive risk profiles for prescribers.
Clinical Recommendations
Optimal management of chronic pain patients at risk for opioid misuse should combine autonomic and behavioral profiling with evidence-based lifestyle interventions:
Dietary modifications
- Mediterranean or whole-food plant-based dietary patterns emphasizing leafy greens, legumes, nuts, whole grains, berries, and healthy fats
- Limitation of processed meats, added sugars, refined grains, and excessive sodium
- Three to five scheduled, balanced meals daily with adequate hydration
Physical activity
- Regular aerobic and resistance exercise tailored to individual capability
- Walking, stretching, or structured physical therapy to support autonomic balance
Mind-body interventions
- Meditation, slow-paced breathing exercises (e.g., 6 breaths/minute)
- Consistent sleep routines and stress management techniques
- HRV biofeedback training when available
Targeted supplementation
For select patients: alpha-lipoic acid, benfotiamine, L-methylfolate, B12, B6 to support nerve health (under medical supervision)
Limitations
This study has several important limitations. The retrospective design and single-center setting may affect generalizability to other populations and clinical settings. The predictive model was developed and evaluated on the same cohort without independent validation; therefore, performance estimates may be optimistic and require confirmation in separate patient samples.
HRV measurement protocols and the abbreviated mYFAS instrument require further standardization across clinical settings. The study did not include long-term follow-up data on actual overdose events, relying instead on validated risk indicators. Selection bias may exist, as patients undergoing HRV and UPFA assessments may differ systematically from the broader chronic pain population.
Future research should focus on prospective validation in independent cohorts, longitudinal tracking of autonomic function and dietary patterns over time, and randomized controlled trials of HRV-guided and UPFA-targeted interventions to establish causal relationships and quantify intervention effectiveness.
Conclusions
Incorporating HRV and UPFA screening into pain management protocols enables substantially earlier and more accurate identification of patients at risk for opioid overdose—approximately 12.7 months before PDMP monitoring alone would indicate elevated risk. The composite NARX-HRV-UPFA model demonstrates strong predictive performance (AUC=0.91, sensitivity 89%), representing a clinically meaningful improvement over current standard-of-care risk assessment.
Processed meat consumption and sugar addiction represent key modifiable behavioral risk factors that should be targeted in dietary and lifestyle interventions. Combined autonomic and behavioral profiling has the potential to reduce iatrogenic mortality through early identification and targeted multimodal interventions including biofeedback, neuromodulation, sympathetic blocks, and structured dietary counseling.
Systemic barriers, particularly insurance denials for evidence-based autonomic testing and behavioral interventions, must be addressed through policy reform. National implementation of autonomic and behavioral profiling protocols, supported by appropriate reimbursement policies, could potentially prevent tens of thousands of annual overdoses and dramatically reduce associated healthcare costs.
External validation in independent cohorts and prospective intervention trials are needed to confirm these findings and establish evidence-based treatment protocols.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
No external funding was received for this study.
Acknowledgments
No individuals or organizations contributed to this study except for those listed as authors.
References
2. Barakat A, Vogelzangs N, Licht CM, Geenen R, MacFarlane GJ, de Geus EJ, et al. Dysregulation of the autonomic nervous system and its association with the presence and intensity of chronic widespread pain. Arthritis Care Res (Hoboken). 2012 Aug;64(8):1209–16.
3. Shaffer F, Ginsberg JP. An Overview of Heart Rate Variability Metrics and Norms. Front Public Health. 2017 Sep 28;5:258.
4. Bruehl S, Chung OY. Interactions between the cardiovascular and pain regulatory systems: an updated review of mechanisms and possible alterations in chronic pain. Neurosci Biobehav Rev. 2004 Jul;28(4):395–414.
5. Zhang C, Xu HP, Zheng HF, Wu XL, Du HG, Jin X. Assessing the viability of heart rate variability as an objective and comprehensive indicator of chronic non-specific neck pain. PLoS One. 2025 Jul 2;20(7):e0326357.
6. Koenig J, Jarczok MN, Ellis RJ, Hillecke TK, Thayer JF. Heart rate variability and experimentally induced pain in healthy adults: a systematic review. Eur J Pain. 2014 Mar;18(3):301–14.
7. Sgoifo A, Carnevali L, Alfonso Mde L, Amore M. Autonomic dysfunction and heart rate variability in depression. Stress. 2015;18(3):343–52.
8. Rampazo ÉP, Rehder-Santos P, Catai AM, Liebano RE. Heart rate variability in adults with chronic musculoskeletal pain: A systematic review. Pain Pract. 2024 Jan;24(1):211–30.
9. Tracy LM, Ioannou L, Baker KS, Gibson SJ, Georgiou-Karistianis N, Giummarra MJ. Meta-analytic evidence for decreased heart rate variability in chronic pain implicating parasympathetic nervous system dysregulation. Pain. 2016 Jan;157(1):7–29.
10. Novak P. Autonomic disorders. The American Journal of Medicine. 2019 Apr 1;132(4):420–36.
11. Shahani BT, Halperin JJ, Boulu P, Cohen J. Sympathetic skin response--a method of assessing unmyelinated axon dysfunction in peripheral neuropathies. J Neurol Neurosurg Psychiatry. 1984 May;47(5):536–42.
12. Forte G, Troisi G, Pazzaglia M, Pascalis V, Casagrande M. Heart Rate Variability and Pain: A Systematic Review. Brain Sci. 2022 Jan 24;12(2):153.
13. Paccione CE, Bruehl S, My Diep L, Rosseland LA, Stubhaug A, Jacobsen HB. The indirect impact of heart rate variability on cold pressor pain tolerance and intensity through psychological distress in individuals with chronic pain: the Tromsø Study. Pain Rep. 2021 Feb 15;7(2):e970.
14. Flint AJ, Gearhardt AN, Corbin WR, Brownell KD, Field AE, Rimm EB. Food-addiction scale measurement in 2 cohorts of middle-aged and older women. The American Journal of Clinical Nutrition. 2014 Mar 1;99(3):578–86.
15. Schulte EM, Gearhardt AN. Development of the modified Yale food addiction scale version 2.0. European Eating Disorders Review. 2017 Jul;25(4):302–8.
16. Brunault P, Berthoz S, Gearhardt AN, Gierski F, Kaladjian A, Bertin E, et al. The Modified Yale Food Addiction Scale 2.0: Validation Among Non-Clinical and Clinical French-Speaking Samples and Comparison With the Full Yale Food Addiction Scale 2.0. Front Psychiatry. 2020 Sep 8;11:480671.
17. Gearhardt AN, Corbin WR, Brownell KD. Preliminary validation of the Yale Food Addiction Scale. Appetite. 2009 Apr;52(2):430-6.
18. Pursey KM, Stanwell P, Gearhardt AN, Collins CE, Burrows TL. The prevalence of food addiction as assessed by the Yale Food Addiction Scale: a systematic review. Nutrients. 2014 Oct 21;6(10):4552–90.
19. NARX Score Analysis; Ohio Automated Rx Reporting System (OARRS) User Manual. Available from: https://www.ohiopmp.gov/Documents/OARRS_User_Manual.pdf.
20. CPMI Autonomic testing protocol. Available from: https://cpmiohio.com/wp-content/uploads/2025/07/SSR-2025-Template-July-25.pdf.