Abstract
Background: Breast cancer demonstrates substantial molecular heterogeneity that influences prognosis and therapeutic decisions. Immunohistochemistry (IHC) provides an accessible surrogate method for molecular classification in low-resource settings. This study evaluated the distribution and clinicopathological significance of IHC-defined molecular subtypes among breast cancer cases in a Nigerian population.
Methods: A retrospective cross-sectional study was conducted on 273 histologically confirmed breast cancers diagnosed between 2019 and 2025. Formalin-fixed, paraffin-embedded tissues were stained for estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), and Ki-67. Tumors were categorized into Luminal A, Luminal B (HER2−), Luminal B (HER2+), HER2-enriched, and Triple-Negative Breast Cancer (TNBC). Statistical analysis was performed using SPSS version 27. Associations between molecular subtype and clinicopathological parameters were evaluated using Chi-square tests, with p<0.05 considered significant.
Results: TNBC was the most prevalent subtype (33.7%), followed by Luminal A (20.5%), Luminal B (HER2+) (16.1%), HER2-enriched (16.1%), and Luminal B (HER2−) (13.6%). Infiltrating duct carcinoma (NOS) accounted for 85.3% of cases. A significant association was observed between age group and molecular subtype (χ² = 14.36, df = 8, p = 0.023). No significant relationship was found between molecular subtype and histologic grade (p = 0.149). The Ki-67 index was high (≥20%) in 41% of tumors.
Conclusion: The predominance of aggressive subtypes, particularly TNBC, highlights an urgent need to integrate routine molecular classification into breast cancer diagnostics in Nigeria. IHC-based profiling is a practical tool for improving risk stratification and guiding individualized therapy in resource-limited regions.
Keywords
Breast cancer, Molecular subtypes, Immunohistochemistry, Estrogen receptor, HER2, Triplenegative breast cancer, Ki-67, Nigeria
Background
Breast cancer remains the most frequently diagnosed malignancy among women worldwide and a leading cause of cancer-related mortality [1]. Its burden continues to rise in low- and middle-income countries [2,3], where late presentation, limited diagnostic capacity, and uneven access to targeted therapies contribute substantially to poor outcomes [4]. Although traditionally classified using histomorphology, breast cancer is now recognized as a biologically diverse disease composed of distinct molecular entities, each characterized by unique patterns of growth, behavior, and therapeutic response [5].
The introduction of gene-expression profiling revolutionized breast cancer taxonomy by identifying intrinsic subtypes, Luminal A, Luminal B, HER2-enriched, and basal-like, each associated with distinct prognostic profiles [6]. However, genomic technologies remain largely inaccessible in many resource-constrained settings. Immunohistochemistry (IHC) has therefore emerged as a practical surrogate method for molecular classification [7]. Assessment of ER, PR, HER2, and Ki-67 permits reasonably accurate assignment of tumors to clinically meaningful surrogate molecular groups [8,9]
Despite increasing reliance on IHC for clinical decision-making, data on the molecular landscape of breast cancer in African populations remain limited. Several studies suggest a disproportionate burden of biologically aggressive tumors, particularly triple-negative breast cancer (TNBC), among women of African ancestry [10–12]. Studies from West, East, and Southern Africa consistently report higher frequencies of TNBC than in European or Asian cohorts [13–15]. Yet many of these regional reports are constrained by small sample sizes, outdated classification systems, or incomplete biomarker profiling. Consequently, there is a critical need for robust, contemporary, population-level data to better understand disease biology and guide treatment planning.
This study addresses this gap by evaluating the distribution of IHC-defined molecular subtypes and their clinicopathological correlates in a large cohort of Nigerian breast cancer patients. By characterizing the molecular profile of tumors within this population, the study aims to support context-specific strategies for diagnosis, prognostication, and individualized therapy.
Methods
Study design and setting
This study employed a retrospective cross-sectional design involving breast cancer cases diagnosed at the Anatomic Pathology Unit of SynLab Nigeria, Lagos. The study period spanned January 2019 to January 2025, during which all eligible cases received routine histopathologic evaluation and immunohistochemical profiling. SynLab is a high-volume diagnostic center serving a broad population that includes both referred tertiary hospital patients and individuals presenting directly for diagnostic evaluation. This setting provides a diverse and representative sample of breast cancer diagnoses in the region.
Study population
The study included 273 women of age ranged from 22 to 86 years, with histologically confirmed primary breast cancer. Each case underwent detailed morphologic examination followed by immunohistochemical analysis as part of diagnostic workup. All available patient records and tumor blocks meeting the inclusion criteria were reviewed consecutively to minimize selection bias.
Inclusion and exclusion criteria
Inclusion criteria
- Histologically confirmed malignant breast tumors.
- Adequate and well-preserved formalin-fixed, paraffin-embedded (FFPE) tissue blocks suitable for IHC staining.
- Complete pathology records, including demographic and clinical data.
Exclusion criteria
- Severely autolyzed, crushed, or poorly preserved FFPE blocks unsuitable for IHC evaluation.
- Specimens from patients previously treated with neoadjuvant chemotherapy, as therapy may alter biomarker expression.
- Tumors with missing or incomplete immunohistochemical data.
Histopathologic evaluation
Archived FFPE blocks were retrieved, sectioned at 4 μm thickness, and stained with hematoxylin and eosin (H&E). Tumors were re-evaluated by a consultant pathologist, blinded to initial reports, to ensure diagnostic accuracy and uniform application of the 2019 WHO Classification of Breast Tumors [16].
Tumor characteristics assessed included
Histologic type
Histologic grade using the Nottingham grading system (I–III) [17]
Presence of lymphovascular invasion (LVI)
Tumor margins and architectural patterns
Any discrepancies between the original report and the review were resolved by consensus.
Immunohistochemistry (IHC)
IHC staining was performed using SynLab’s standardized automated platform.
The stains are highly specific with high purity, staining what was expected to stain and the type of stain is Indirect Staining: This is a two-step (or multi-step) method involving an unlabeled primary antibody binds to the target antigen, and then a labelled secondary antibody, which recognizes the primary antibody and significantly amplifying the signal and increasing sensitivity.
The following primary antibodies were used:
Estrogen receptor (ER), Progesterone receptor (PR), HER2/neu (c-erbB-2), Ki-67 proliferative index.
Positive and negative controls were included for each run to ensure assay reliability.
Interpretation criteria
ER and PR
A tumor was considered positive if ≥1% of tumor nuclei demonstrated immunoreactivity.
HER2
Scored according to cri/CAP guidelines (2018) [18]:
0 or 1+ = negative
2+ = equivocal; reflex testing recommended
3+ = positive
Due to resource constraints, HER2 2+ cases without confirmatory FISH were categorized as negative, as is standard in many LMIC diagnostic settings.
Ki-67
Dichotomized as low (<20%) and high (≥20%), consistent with contemporary recommendations for IHC-based surrogate classification. The 20% Ki-67 cut-off was selected primarily because studies showed it best stratifies breast cancer patients into distinct prognostic groups (low vs. high risk of recurrence and death), with the high Ki-67 group showing significantly poorer outcomes. This cut-off also showed good concordance with molecular tests and effectively identified patients who might benefit from more aggressive therapies like chemotherapy [19,20].
Molecular subtype classification
Tumors were assigned to one of five IHC-defined molecular subtypes:
Luminal A: ER+ and/or PR+, HER2–, Ki-67 <20%
Luminal B (HER2–): ER+ and/or PR+, HER2–, Ki-67 ≥20%
Luminal B (HER2+): ER+ and/or PR+, HER2+
HER2-Enriched: ER–, PR–, HER2+
Triple-Negative Breast Cancer (TNBC): ER–, PR–, HER2–
This classification mirrors surrogate systems widely used in clinical practice and research settings where genomic profiling is unavailable.
Data extraction
Demographic and clinicopathologic data were extracted from SynLab’s Laboratory Information System (LIS). Variables collected included:
- Age at diagnosis
- Histologic diagnosis
- Histologic grade
- IHC biomarker status (ER, PR, HER2, Ki-67)
- Derived molecular subtype
All data were anonymized to protect patient confidentiality.
Statistical analysis
Data analysis was performed using IBM SPSS Statistics version 27.
Descriptive statistics
Categorical variables were summarized using frequencies and percentages. Continuous variables (e.g., age) were expressed as means ± standard deviations where appropriate.
Inferential statistics
Associations between molecular subtypes and clinicopathological variables (age group, grade, histologic type, Ki-67) were tested using the Chi-square test (χ²).
When expected cell counts were <5, Fisher's exact test was applied. Statistical significance was set at p < 0.05, while Cramer's V was calculated for effect size to aid interpretation of significant relationships.
Ethical considerations
Ethical approval for this study was granted by the appropriate institutional review board. All data were handled in accordance with the principles of the Declaration of Helsinki. As a retrospective review of anonymized records, the requirement for informed consent was waived.
Results
Patient demographics
A total of 273 breast cancer cases met the inclusion criteria with a mean age of 51.8 ± 12.6 years (Table 1). More than half of the cohort (54.6%) fell within the 41–60 years age group, while 19.8% were below 40 years and 25.6% were above 60 years. Thus, the disease predominantly affected middle-aged women, but a substantial subset of younger patients was also represented [15,21].
|
Variable |
Category |
n |
% |
|
Age (years) |
< 40 41–60 > 60 |
54 149 70 |
19.8 54.6 25.6 |
|
Histologic grade |
I II III |
32 166 75 |
11.7 60.8 27.5 |
|
ER status |
Positive Negative |
166 107 |
60.8 39.2 |
|
PR status |
Positive Negative |
135 138 |
49.5 50.5 |
|
HER2 status |
Positive (3+) Negative (0–1+) |
54 219 |
19.8 80.2 |
|
Ki-67 index |
High (≥ 20 %) Low (< 20 %) |
112 161 |
41 59 |
Histologic characteristics
The vast majority of tumors were infiltrating ductal carcinoma (NOS), accounting for 85.3% (n = 233) of cases (Table 1). Less frequent diagnoses included lobular carcinoma (5.1%), high-grade ductal carcinoma in situ with microinvasion (2.2%), inflammatory carcinoma (1.8%), neuroendocrine carcinoma (1.5%), and other rare subtypes (4.0%). This distribution reinforces the well-documented predominance of ductal carcinoma in global breast cancer epidemiology [22].
Histologic grading showed a predominance of grade II tumors, comprising 60.8% of cases, followed by grade III (27.5%) and grade I (11.7%). The predominance of intermediate-grade tumors suggests a moderately differentiated overall tumor profile in this cohort.
Hormone receptor and proliferative marker expression
Overall, 60.8% of tumors were ER-positive, while 39.2% lacked ER expression. PR expression demonstrated a balanced distribution, with 49.5% positivity and 50.5% negativity.
HER2 overexpression (3+) was identified in 19.8% of cases, while the majority (80.2%) were HER2-negative (0–1+ or 2+ without confirmatory testing) (Table 1).
The Ki-67 proliferation index was high (≥20%) in 41% of tumors, reflecting a substantial subset of highly proliferative malignancies. The remaining 59% demonstrated a low Ki-67 index (<20%) [20] (Table 1). Figure 1 shows representative immunohistochemical expression patterns of breast cancer biomarkers.
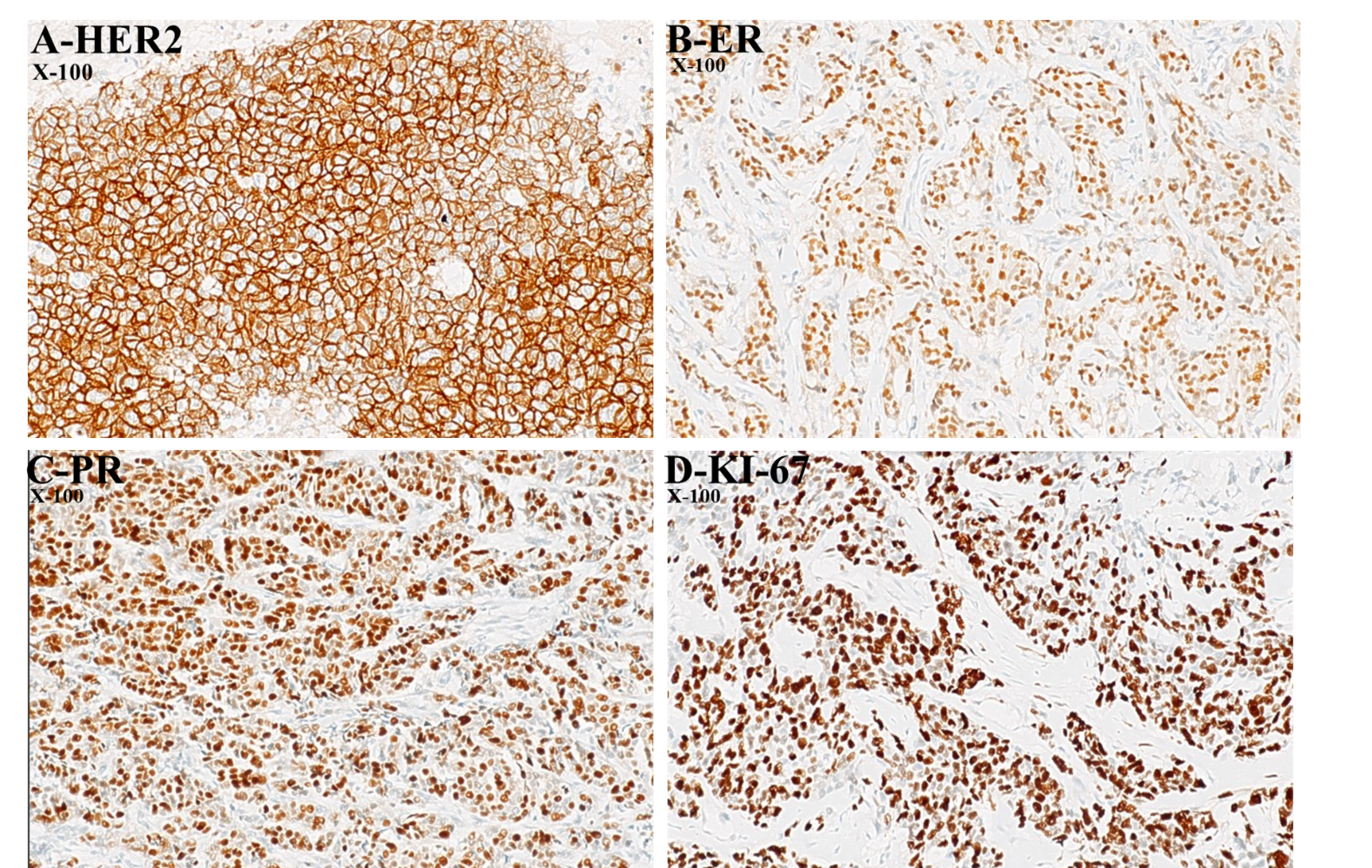
Figure 1. Representative immunohistochemical expression patterns of breast cancer biomarkers. (A) HER2 showing strong, complete membranous staining of tumour cells (score 3+), indicating HER2 overexpression (×100), (B) Estrogen receptor (ER) showing distinct brown nuclear positivity in a subset of tumour cells (×100), (C) Progesterone receptor (PR) demonstrating diffuse brown nuclear staining in tumour cells (×100) and (D) Ki-67 showing strong brown nuclear immunoreactivity in a high proportion of tumor cells, indicating a high proliferative index (×100).
Molecular subtype distribution
Based on combined expression of ER, PR, HER2, and Ki-67, as shown in Table 2, the IHC-defined molecular subtypes were distributed as follows:
- Triple-Negative Breast Cancer (TNBC): 33.7% (n = 92)
- Luminal A: 20.5% (n = 56)
- Luminal B (HER2–): 13.6% (n = 37)
- Luminal B (HER2+): 16.1% (n = 44)
- HER2-Enriched: 16.1% (n = 44)
TNBC emerged as the single most common molecular subtype, accounting for one-third of all cases. Together, Luminal A and Luminal B (HER2–) tumors represented approximately 34% of the cohort, consistent with the predominance of hormone receptor–positive disease across many populations but highlighting a greater burden of aggressive subtypes in this setting.
|
Subtype |
Definition |
n |
% |
|
Luminal A |
ER+/PR+, HER2−, Ki-67 < 20 % |
87 |
31.9 |
|
Luminal B |
ER+/PR+/−, HER2+/−, Ki-67 ≥ 20 % |
67 |
24.5 |
|
HER2-enriched |
ER−/PR−/HER2+ |
27 |
9.9 |
|
Triple-negative |
ER−/PR−/HER2− |
92 |
33.7 |
Relationship between molecular subtypes and clinicopathological features
Age and molecular subtype
A statistically significant association was observed between age group and molecular subtype (χ² = 7.56, df = 3, p = 0.023) (Table 3).
TNBC predominated among women under 40 years and those 41–60 years, whereas luminal tumors, especially Luminal A, were more common among patients above 60 years. This suggests a potential age-related shift in tumor biology, with younger women demonstrating more aggressive phenotypes (Table 3).
|
IHC classification of molecular subtypes |
||||||
|
Age group (years) |
HER2- enriched |
Luminal A |
Luminal B (HER2- ) |
Luminal B (HER2+) |
Triple negative |
P-value |
|
20-24 |
0 |
1 |
0 |
0 |
0 |
df = 3, |
|
25-29 |
0 |
0 |
0 |
3 |
5 |
|
|
30-34 |
5 |
4 |
4 |
1 |
5 |
|
|
35-39 |
2 |
4 |
8 |
7 |
9 |
|
|
40-44 |
6 |
18 |
4 |
6 |
12 |
|
|
45-49 |
8 |
7 |
3 |
10 |
18 |
|
|
50-54 |
7 |
4 |
5 |
4 |
11 |
|
|
55-59 |
10 |
4 |
2 |
6 |
9 |
|
|
60 and above |
6 |
14 |
11 |
7 |
23 |
|
Histologic grade and molecular subtype
No statistically significant association was found between histologic grade and molecular subtype (p = 0.149). Although high-grade morphology was more frequently observed in TNBC and HER2-enriched tumors, the overlap across subtypes diluted statistical significance. This finding reinforces the well-known heterogeneity of grade within luminal and non-luminal tumors.
Histologic type and molecular subtype
Infiltrating ductal carcinoma exhibited the full range of molecular subtypes, with TNBC and luminal groups represented prominently. Lobular carcinoma cases predominantly fell within the luminal categories, reflecting their known steroid receptor–driven biology.
Conversely, inflammatory carcinoma showed a strong inclination toward TNBC, consistent with its aggressive clinical behavior.
While histologic type showed descriptive differences across subtypes, the association did not achieve statistical significance (p > 0.05), likely due to small numbers in rare categories.
Ki-67 and molecular subtype
High Ki-67 expression (≥20%) was most frequently observed in HER2-enriched, Luminal B, and TNBC tumors, whereas Luminal A tumors were predominantly low-proliferative. This marked difference aligns with the biological framework that positions Luminal A tumors as more indolent, while the remaining subtypes are characterized by heightened mitotic activity.
Discussion
This study provides a comprehensive characterization of the molecular landscape of breast cancer in a Nigerian cohort using immunohistochemistry as a surrogate for intrinsic subtyping. Several important patterns emerge, with meaningful clinical and biological implications for breast cancer care in sub-Saharan Africa [13,15].
Predominance of triple-negative breast cancer (TNBC)
The most striking finding is the high proportion of TNBC (33.7%), which stands out as the predominant molecular subtype in this cohort. This aligns with other African and African-diaspora data showing disproportionate representation of basal-like tumors among women of African ancestry [14,23]. However, the proportion identified here is substantial and reinforces concerns about the aggressive phenotype that characterizes breast cancer in many African populations. Biologically, TNBC tumors tend to exhibit basal-like gene expression, enriched for [24,25].
TP53 mutations
Proliferation pathway upregulation, defects in DNA repair pathways (including BRCA-associated mechanisms) and enhanced immune infiltration in some subsets.
These tumors typically present with rapid growth, early metastasis, and limited therapeutic options in settings with constrained access to immunotherapy or PARP inhibitors [26].
Clinically, the predominance of TNBC has sobering implications. Because TNBC lacks ER, PR, and HER2 targets, treatment largely depends on cytotoxic chemotherapy, an option that may not be readily accessible or well tolerated in many low-resource environments. Furthermore, supportive care for chemotherapy toxicities is not uniformly available, which can worsen outcomes. The high frequency of TNBC in this cohort, therefore, underscores an urgent need for earlier diagnosis, integration of genetic counseling, and enhanced chemotherapy support services.
Distribution of luminal tumors
Luminal A tumors accounted for 20.5% of cases, and Luminal B tumors (HER2– and HER2+) together accounted for approximately 29.7%. These proportions differ significantly from Western populations, where Luminal A tumors often comprise nearly half of all cases [22].
Several factors may explain this lower proportion of luminal disease:
- Younger age structure of African populations, with higher premenopausal incidence [21].
- Genetic predispositions influencing hormone receptor expression.
- Delayed presentation, which may lead to down-regulation of ER and PR in advanced tumors.
- Tumor dedifferentiation associated with chronic inflammation or microenvironmental factors.
- Luminal tumors typically respond well to endocrine therapy, which is relatively accessible and affordable [19]. Ensuring consistent access to tamoxifen, aromatase inhibitors, and ovarian suppression strategies may therefore produce substantial survival gains.
HER2-enriched and Luminal B (HER2+) subtypes
HER2 overexpression was identified in 19.8% of tumors, with HER2-enriched and Luminal B (HER2+) subtypes each contributing 16.1% to the total sample. This frequency is comparable to global estimates [27], suggesting that HER2-driven biology may be relatively stable across populations.
However, the clinical consequences differ greatly based on treatment availability. HER2-targeted therapies, trastuzumab, pertuzumab, and newer antibody, drug conjugates remain limited in many parts of Africa due to high cost. Consequently, women with HER2-positive tumors may not derive the survival benefits seen in high-resource countries. This situation highlights the importance of advocating for access programs, biosimilar trastuzumab formulations, and national insurance coverage [28].
Age–subtype relationship
A significant association was observed between age group and molecular subtype, with TNBC disproportionately affecting younger and middle-aged women. This age pattern has profound implications: Younger women often present with higher-grade, higher-proliferation tumors.
They may carry hereditary predispositions, including BRCA1 mutations known to drive basal-like/TNBC phenotypes [29,30]. Fertility preservation, psychosocial support, and survivorship planning become more complex. This pattern emphasizes the need for genetic risk assessment, particularly for young women presenting with TNBC.
Histologic grade and molecular subtype
Despite descriptive differences, histologic grade was not significantly associated with molecular subtype in this study. While TNBC and HER2-enriched tumors often trend toward higher grades, luminal tumors can also demonstrate grade heterogeneity, especially in resource-limited settings where delays in diagnosis may permit tumor progression.
This lack of association reinforces the view that grade alone is insufficient to determine tumor biology, and that molecular classification provides indispensable complementary prognostic information [22].
Ki-67 proliferation index and tumor behavior
High Ki-67 expression (≥20%) was most common among HER2-enriched, Luminal B, and TNBC tumors. This is consistent with the aggressive clinical behavior associated with these subtypes [8].
Ki-67, despite variability in scoring across laboratories, remains a useful marker of proliferative activity and is central to distinguishing Luminal A from Luminal B tumors. In this cohort, the strong alignment between high Ki-67 and aggressive subtypes supports its continued use and the need for standardized scoring protocols [20].
Histologic type and subtype distribution
The predominance of infiltrating ductal carcinoma (85.3%) mirrors global patterns. Lobular carcinoma cases were mostly luminal, reflecting their hormone receptor-rich biology, while inflammatory carcinoma clustered toward TNBC, consistent with its aggressive natural history.
Although the association between histologic type and subtype did not reach statistical significance, the descriptive patterns observed align with established tumor biology and provide additional context for clinical interpretation.
Clinical and public health implications
This molecular profile paints a clear picture: breast cancer in this population is characterized by a higher-than-expected burden of aggressive subtypes, which contributes to poor outcomes seen across sub-Saharan Africa [28,31].
Implications include
- Urgent need for earlier detection, particularly among younger women.
- Expansion of IHC-based molecular testing across the region to support risk stratification.
- Improved access to chemotherapy and endocrine therapy, which remain relatively cost-effective.
- Advocacy for affordable HER2-targeted therapies, including biosimilars.
- Genetic counseling and BRCA testing, especially for young TNBC patients.
- This study adds valuable evidence toward developing more nuanced, resource-appropriate breast cancer treatment guidelines in Nigeria and comparable settings.
Comparison to existing evidence and explanation of differences
The findings align with reports from East, West, and Southern Africa but contrast sharply with European and North American cohorts where luminal tumors dominate. Reasons for these differences include:
Genetic ancestry and the influence of genomic architecture, Variations in reproductive patterns and hormonal exposures, Delays in detection leading to more advanced, dedifferentiated tumors.
Limited access to screening and diagnostic services.
These factors collectively shape the unique molecular epidemiology observed in the region.
Conclusion
This study provides a detailed and contemporary characterization of breast cancer molecular subtypes in a Nigerian cohort using immunohistochemistry as a practical surrogate for genomic profiling. The molecular landscape observed is dominated by Triple-Negative Breast Cancer, accompanied by relatively fewer Luminal A tumors than typically reported in Western populations. These findings underscore a significant burden of biologically aggressive disease [24], particularly among younger women, and highlight the need for targeted strategies to improve outcomes.
Routine incorporation of IHC-based molecular classification into diagnostic workflows can empower clinicians to make more individualized treatment decisions, even in resource-limited environments. The high prevalence of TNBC and HER2-enriched tumors further emphasizes the importance of expanding access to effective systemic treatments, strengthening chemotherapy delivery systems, and advocating for affordable HER2-targeted therapies. This work therefore contributes valuable evidence supporting regional efforts toward precision oncology and provides a foundation for future studies, particularly those integrating genetic profiling and longitudinal outcome data.
Strengths and Limitations
Strengths
This study leverages a relatively large sample size (n = 273), providing robust insights into the molecular profile of breast cancer in a Nigerian population.
The use of a standardized IHC panel (ER, PR, HER2, Ki-67) ensures reliable subtype classification, consistent with global surrogate definitions.
Uniform re-evaluation of histology using the 2019 WHO classification enhances diagnostic accuracy and comparability.
The study reflects a real-world diagnostic environment, offering pragmatic data relevant to clinical decision-making in low-resource settings.
Limitations
As a retrospective study, it is subject to limitations related to variability in documentation, incomplete clinical information, and selection bias.
HER2 2+ cases lacking confirmatory FISH testing may have led to minor misclassification of HER2 status, a constraint imposed by resource availability.
Absence of genomic profiling means intrinsic molecular subtypes could not be directly compared with their genetic counterparts.
Clinical outcome data (e.g., survival, response to therapy) were not available, limiting the ability to correlate molecular subtypes with prognosis.
Despite these limitations, the study offers valuable insights into the regional biology of breast cancer and highlights priority areas for strengthening diagnostic and therapeutic services.
Declarations
Ethics approval and consent to participate
Ethical approval for this study was granted by the appropriate institutional ethics committee. All patient data were anonymized prior to analysis. Given the retrospective nature of the study, the requirement for informed consent was waived.
Consent for publication
Not applicable.
Availability of data and materials
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request and with appropriate institutional approval.
Competing interests
The authors declare that they have no competing interests.
Funding
This study received no external funding. All research activities were self-funded.
Authors’ contributions
The corresponding author conceptualized the study, participated in data curation and analysis, and led manuscript development. All authors contributed to critical review and approved the final version of the manuscript.
Acknowledgements
The authors acknowledge the technical and administrative support provided by the SynLab Nigeria histopathology laboratory team and express appreciation to the staff involved in the archival management of pathology materials.
References
2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018 Nov;68(6):394–424.
3. Adetona AE, Osungbade KO, Akinyemi OO, Obembe TA. Uptake of Breast Screening Among Female Staff at A Tertiary Health Institution in South-West Nigeria. Journal of Preventive Medicine and Care. 2021 Mar 5;3(2):17–30.
4. Galukande M, Wabinga H, Mirembe F. Breast cancer survival experiences at a tertiary hospital in sub-Saharan Africa: a cohort study. World J Surg Oncol. 2015 Jul 19;13:220.
5. Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A. 2001 Sep 11;98(19):108–74.
6. Parker JS, Mullins M, Cheang MCU, Leung S, Voduc D, Vickery T, et al. Supervised Risk Predictor of Breast Cancer Based on Intrinsic Subtypes. J Clin Oncol. 2023 Sep 10;41(26):419–99.
7. Coates AS, Winer EP, Goldhirsch A, Gelber RD, Gnant M, Piccart-Gebhart M, et al. Tailoring therapies--improving the management of early breast cancer: St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2015. Ann Oncol. 2015 Aug;26(8):1533–46.
8. Nishimura R, Osako T, Okumura Y, Hayashi M, Toyozumi Y, Arima N. Ki-67 as a prognostic marker according to breast cancer subtype and a predictor of recurrence time in primary breast cancer. Exp Ther Med. 2010 Sep;1(5):747–54.
9. Voduc KD, Cheang MC, Tyldesley S, Gelmon K, Nielsen TO, Kennecke H. Breast cancer subtypes and the risk of local and regional relapse. J Clin Oncol. 2010 Apr 1;28(10):168–91.
10. Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. Jama. 2006 Jun 7;295(21):2492–502.
11. Newman LA. Breast cancer in African-American women. Oncologist. 2005 Jan;10(1):1–14.
12. Dietze EC, Sistrunk C, Miranda-Carboni G, O'Regan R, Seewaldt VL. Triple-negative breast cancer in African-American women. Nat Rev Cancer. 2015 Apr;15(4):248–54.
13. Huo D, Ikpatt F, Khramtsov A, Dangou JM, Nanda R, Dignam J, et al. Population differences in breast cancer: survey in indigenous African women reveals over-representation of triple-negative breast cancer. J Clin Oncol. 2009 Sep 20;27(27):4515–21.
14. Fregene A, Newman LA. Breast cancer in sub-Saharan Africa: how does it relate to breast cancer in African-American women? Cancer. 2005 Apr 15;103(8):1540–50.
15. Jedy-Agba E, Curado MP, Ogunbiyi O, Oga E, Fabowale T, Igbinoba F, et al. Cancer incidence in Nigeria: a report from population-based cancer registries. Cancer Epidemiol. 2012 Oct;36(5):e271–8.
16. WHO/IARC. WHO Classification of Tumours of the Breast. 5th ed. Lyon: IARC; 2019. DOI: 10.1111/his.14091.
17. Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology. 1991 Nov;19(5):403–10.
18. Wolff AC, Hammond MEH, Allison KH, Harvey BE, Mangu PB, Bartlett JMS, et al. Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. J Clin Oncol. 2018 Jul 10;36(20):210–2.
19. Coates AS, Winer EP, Goldhirsch A, Gelber RD, Gnant M, Piccart-Gebhart M, et al. Tailoring therapies--improving the management of early breast cancer: St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2015. Ann Oncol. 2015 Aug;26(8):1533–46.
20. Inwald EC, Klinkhammer-Schalke M, Hofstädter F, Zeman F, Koller M, Gerstenhauer M, et al. Ki-67 is a prognostic parameter in breast cancer patients: results of a large population-based cohort of a cancer registry. Breast Cancer Res Treat. 2013 Jun;139(2):539–52.
21. Brinton LA, Figueroa JD, Awuah B, Yarney J, Wiafe S, Wood SN, et al. Breast cancer in Sub-Saharan Africa: opportunities for prevention. Breast Cancer Res Treat. 2014 Apr;144(3):467–78.
22. Adebamowo CA, Ajayi OO. Breast cancer in Nigeria. West Afr J Med. 2000 Jul-Sep;19(3):179–91.
23. Galukande M, Wabinga H, Mirembe F, Karamagi C, Asea A. Molecular breast cancer subtypes prevalence in an indigenous Sub Saharan African population. Pan Afr Med J. 2014 Apr 5;17:249.
24. Dent R, Hanna WM, Trudeau M, Rawlinson E, Sun P, Narod SA. Pattern of metastatic spread in triple-negative breast cancer. Breast Cancer Res Treat. 2009 May;115(2):423–8.
25. Sasaki Y, Tsuda H. Clinicopathological characteristics of triple-negative breast cancers. Breast Cancer. 2009;16(4):254–9.
26. Oluwasanu M, Olopade OI. Global disparities in breast cancer outcomes: new perspectives, widening inequities, unanswered questions. Lancet Glob Health. 2020 Aug;8(8):e97–9.
27. Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001 Mar 15;344(11):78–92.
28. Rweyemamu LP, et al. Breast cancer in East Africa: Prevalence and spectrum of germline SNV/indel and CNVs in BRCA1 and BRCA2 genes among breast cancer patients in Tanzania. Cancer Med. 2023 Feb;12(3):3395–409.
29. Rakha EA, Reis-Filho JS, Ellis IO. Basal-like breast cancer: a critical review. J Clin Oncol. 2008 May 20;26(15):256–81.
30. Kreike B, van Kouwenhove M, Horlings H, Weigelt B, Peterse H, Bartelink H, et al. Gene expression profiling and histopathological characterization of triple-negative/basal-like breast carcinomas. Breast Cancer Res. 2007;9(5):R65.
31. Anyanwu SN. Temporal trends in breast cancer presentation in the third world. J Exp Clin Cancer Res. 2008 Jul 11;27(1):17.
