Introduction
Liver fibrosis represents a critical convergence point in gastroenterological practice, serving as the common pathway from chronic injury—whether metabolic (MASLD/MASH), alcoholic, viral, or drug-induced—to cirrhosis and hepatocellular carcinoma [1,2]. Globally, cirrhosis and chronic liver disease account for over 1.5 million deaths annually, with MASLD now the leading indication for liver transplantation in many regions [3]. Despite advances in etiology-specific therapies (e.g., direct-acting antivirals, GLP-1 receptor agonists for MASLD), no approved antifibrotic agent exists that reliably reverses established fibrosis [4]. Current management relies on lifestyle modification, etiology control, and supportive care, leaving substantial unmet need for safe, multi-target adjunctive therapies [5].
Oxidative stress and low-grade inflammation are central drivers across these etiologies, promoting hepatocyte apoptosis, Kupffer-cell activation, and hepatic stellate cell (HSC) transdifferentiation into myofibroblasts that deposit excessive extracellular matrix (ECM) [6,7]. Traditional Chinese medicine-derived polysaccharides have emerged as promising candidates due to their inherent antioxidant, anti-inflammatory, and immunomodulatory properties [8,9]. However, native polysaccharides often exhibit limitations in potency, solubility, and dose-response at clinically feasible concentrations [10].
Chemical modification, particularly selenization, offers a rational strategy to overcome these barriers [11]. Selenium, an essential trace element and cofactor for glutathione peroxidase (GSH-Px) and other selenoproteins, synergizes with polysaccharide scaffolds to enhance free-radical scavenging, reduce lipid peroxidation, and modulate key fibrogenic pathways while maintaining low toxicity profiles compared with inorganic selenium [12,13]. Recent epidemiological data from NHANES cohorts reveal an L-shaped or inverse relationship between serum selenium levels and advanced liver fibrosis, with higher organic selenium status associated with reduced liver stiffness (measured by transient elastography) but divergent effects on steatosis—highlighting the therapeutic window for supplementation in fibrotic but not purely steatotic disease [14–16].
This commentary examines how selenization of natural polysaccharides addresses core pathophysiological mechanisms in hepatic fibrosis, integrates with contemporary gastroenterological themes (including gut-liver axis dysregulation, TRP-channel-mediated mechanosensing in HSC activation, and integrative approaches in MASLD/IBD-overlap syndromes) [17,18], and evaluates translational potential. Drawing on preclinical systematic reviews (showing consistent collagen suppression in >90% of polysaccharide studies) [19,20] and quantitative meta-analysis of 72 preclinical studies (reporting a standardized mean difference of -2.18, 95% CI -2.57 to -1.79, for hepatic collagen reduction, with 92% of studies demonstrating significant α-SMA suppression) and recent 2024–2025 advances, it argues that selenium-modified polysaccharides represent a high-potential adjunctive class that could enhance standard care while aligning with the journal’s emphasis on experimental-to-clinical translation in liver and gastrointestinal disorders [21]. Notably, no dedicated pooled meta-analysis of selenized polysaccharides for hepatic fibrosis has been published to date, a critical evidence gap addressed in subsequent sections. Emphasis is placed on mechanistic insights, safety considerations, and pragmatic integration into hepatology practice rather than exhaustive recapitulation of any single study [1].
Structural and Functional Innovations in Selenized Polysaccharides for Liver Protection
Selenization typically involves mild nitric-acid-mediated incorporation of selenite groups (often at C-6 or via Se–O linkages) onto glucomannan or glucan backbones, resulting in 20–100 µg/g selenium content—well within the safe organic range (<1 mg/g) [11,22]. This modification modestly increases molecular weight (e.g., by 10–50 kDa through enhanced hydrogen bonding) and alters morphology from smooth/layered to porous/sparse structures, improving aqueous fluidity and bioavailability without disrupting core glycosidic linkages [23].
These physicochemical changes translate directly to functional gains: superior DPPH/ABTS radical scavenging, elevated reducing power, and markedly enhanced GSH-Px activation compared with unmodified counterparts [24,25]. Systematic analyses confirm that chemically modified polysaccharides, including selenized derivatives, achieve hepatic accumulation and efficacy at lower doses (often 50–150 mg/kg) than native forms, addressing the poor dose-response of high-viscosity polysaccharides at medium concentrations [19,26].
In the context of gastroenterology, such modifications align with needs for agents effective in heterogeneous patient populations, including those with comorbid IBD where gut-liver axis inflammation amplifies fibrosis [27]. Recent characterizations of selenized Angelica sinensis and Lycium barbarum polysaccharides demonstrate preserved monosaccharide composition alongside amplified antioxidant capacity, providing a blueprint for scalable, standardized production suitable for clinical-grade formulations [28,29]. However, critical heterogeneity persists across the literature in selenization protocols (including nitric acid concentration, reaction temperature and duration, and selenium source), analytical methods for selenium quantification (atomic absorption spectrometry, inductively coupled plasma mass spectrometry, and spectrophotometric assays), and reporting of selenium speciation (organic vs. inorganic) and modification site confirmation. This lack of standardization directly limits cross-study reproducibility and comparability, representing a core barrier to clinical translation, as discussed in later sections.
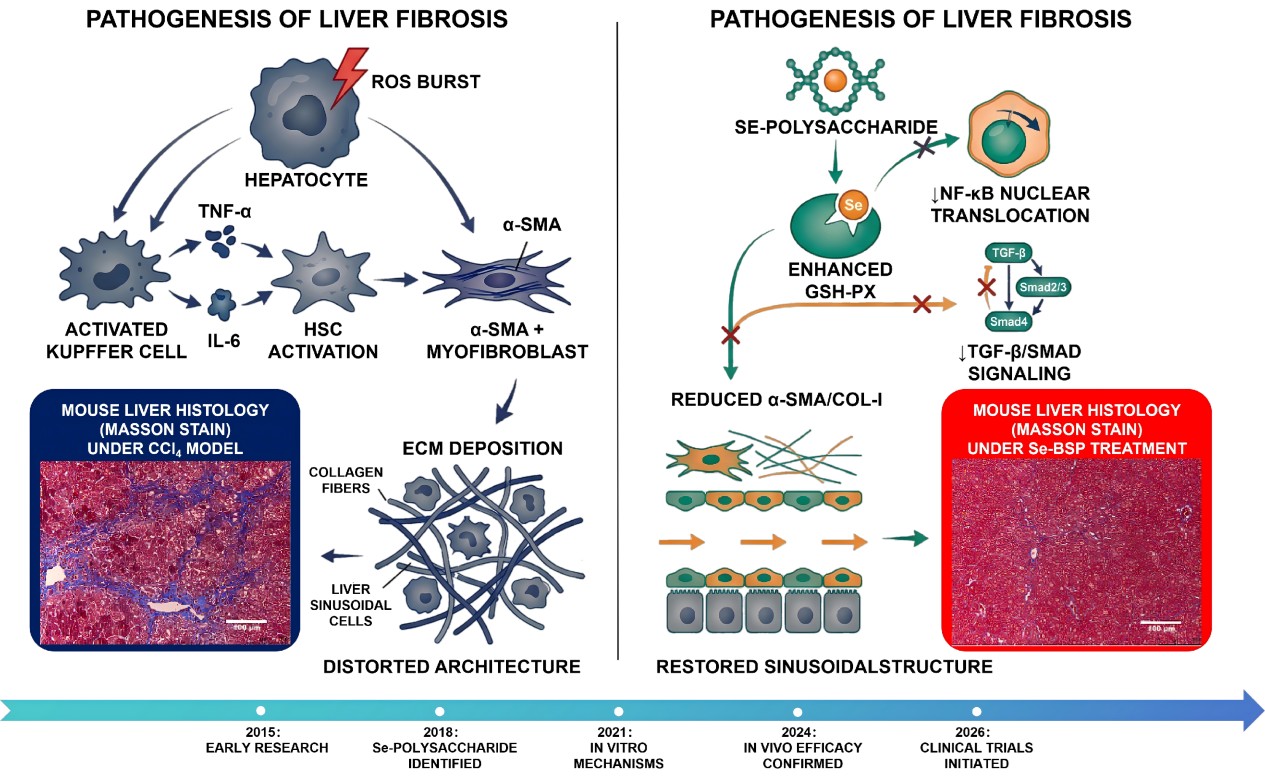
Figure 1. Schematic overview of liver fibrosis pathogenesis and intervention by selenium-modified polysaccharides [1].
Mechanistic Insights: Multi-Target Regulation of Oxidative Stress, Inflammation, and Fibrogenesis
Selenized polysaccharides exert multi-level protection by addressing the oxidative stress–inflammation–fibrogenesis axis. Selenium incorporation augments endogenous antioxidant enzymes (GSH-Px, SOD), directly reducing malondialdehyde and reactive oxygen species that otherwise trigger hepatocyte necrosis and release damage-associated molecular patterns [1,12]. This dampens TLR4/NF-κB signaling, lowering pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) that recruit and activate HSCs [30,31].
Downstream, inhibition of TGF-β/Smad3 phosphorylation and nuclear translocation prevents HSC transdifferentiation, evidenced by reduced α-SMA and collagen I expression [1, 32]. Additional pathways include MAPK (↓p-ERK/JNK/p38), NLRP3 inflammasome suppression, and IL-22/STAT3-mediated anti-apoptotic effects on hepatocytes [33,34]. Gut-liver axis modulation—via prebiotic effects on short-chain fatty acid-producing bacteria—further reduces LPS translocation, reinforcing anti-inflammatory actions relevant to MASLD and IBD-associated liver injury [35,36].
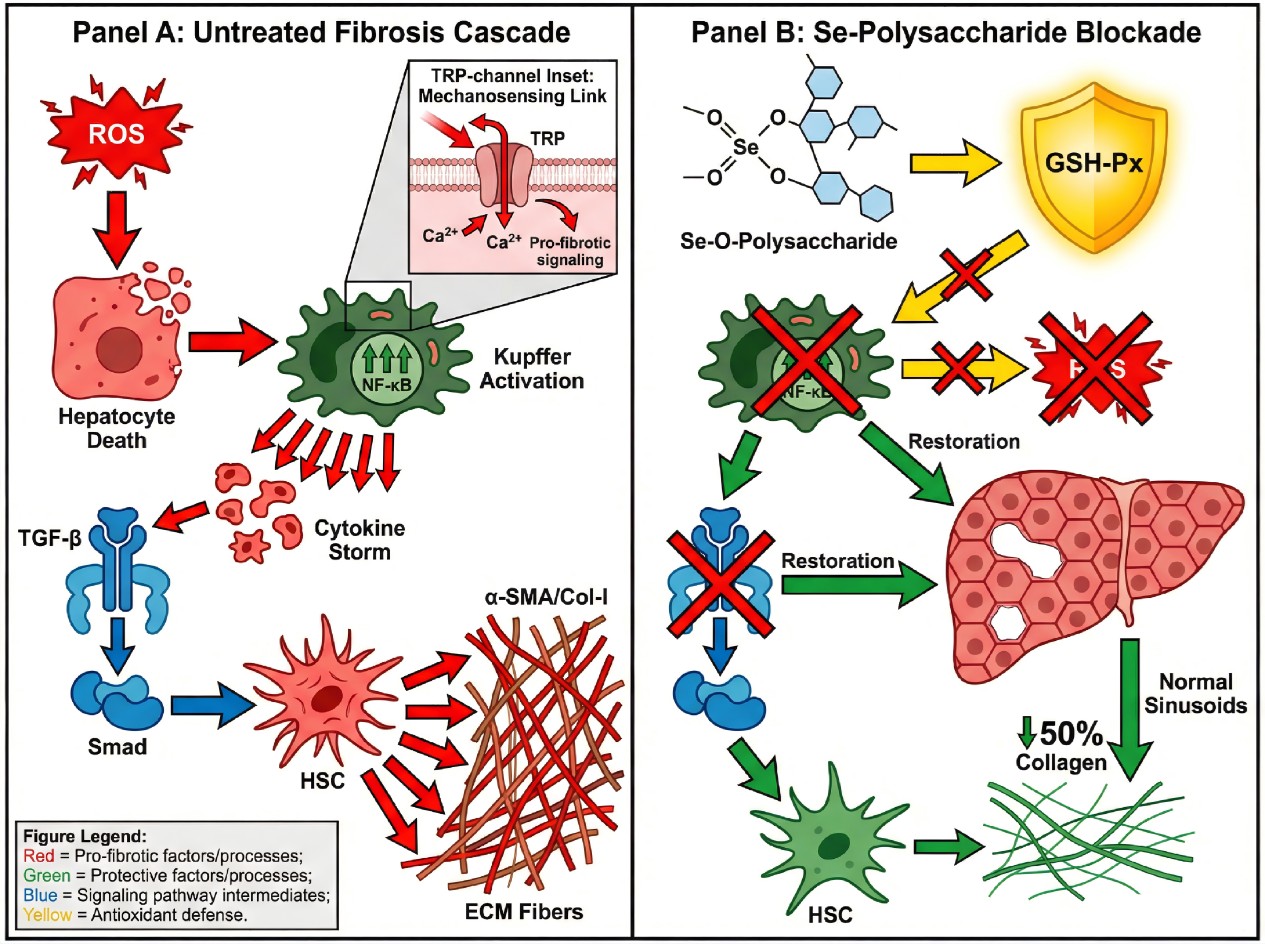
Figure 2. Multi-pathway mechanistic diagram of selenium-modified polysaccharides in alleviating hepatic fibrosis.
Clinical Relevance and Translational Opportunities in Gastroenterology
Preclinical superiority at medium doses (150 mg/kg) in rodent models provides a preliminary basis for human dose estimation. Per U.S. Food and Drug Administration (FDA) guidance for allometric scaling of preclinical dosing, the human equivalent dose (HED) for a 60 kg adult is ~14.6 mg/kg, equivalent to 700–1200 mg/day, accounting for inter-species variability [1]. Critical caveats to this estimate must be emphasized: this scaling assumes linear dose proportionality, which has not been validated in humans, particularly in patients with impaired hepatic function, who have altered absorption, distribution, metabolism, and excretion (ADME) of oral polysaccharides and selenium compounds. No published human pharmacokinetic data for selenized Bletilla striata polysaccharide or related derivatives exist to date, representing a mandatory prerequisite for clinical trial design and safe clinical translation.
NHANES data support this: higher serum selenium correlates with lower liver stiffness (inverse association with advanced fibrosis, OR 0.55 in highest quartile), particularly in older adults and those with metabolic risk—common gastroenterology cohorts [14–16]. To maximize clinical benefit and minimize risk, evidence-based patient selection and biomarker stratification are essential. The optimal target population includes: (1) patients with F2–F3 (moderate-to-advanced) compensated hepatic fibrosis, the population with reversible fibrogenic activity and the primary target of contemporary antifibrotic clinical trials; (2) patients with baseline selenium deficiency (serum selenium <70 μg/L, per World Health Organization guidelines), as epidemiological data show no significant antifibrotic benefit in selenium-sufficient individuals (>125 μg/L); and (3) patients with etiologies driven by oxidative stress, gut-liver axis dysregulation, and low-grade inflammation, including MASLD, alcoholic liver disease, and IBD-associated liver injury.
In practice, selenium-modified polysaccharides could serve as adjuncts in compensated cirrhosis, MASLD fibrosis staging, or IBD patients on biologics (e.g., vedolizumab) where liver comorbidity is rising [9]. Their gut-modulatory effects may synergize with microbiome-targeted therapies, while low toxicity (organic Se profile) suits long-term use unlike inorganic forms [37,38].
Integration with journal-highlighted areas—TRP-channel inhibition in fibrosis, hemochromatosis oxidative damage, or phytotherapy for HSC modulation—positions these agents as versatile tools in personalized hepatology [17,39]. Notably, these agents have strong mechanistic rationale for use in combination therapy: they may synergize with GLP-1 receptor agonists (which address upstream metabolic drivers of MASLD) by targeting downstream oxidative stress and fibrogenic pathways, and may complement emerging investigational antifibrotics (including PPAR agonists, caspase inhibitors, and LOXL2 inhibitors) to enhance antifibrotic efficacy without additive toxicity. Preclinical studies to evaluate these synergistic combinations are a critical near-term priority.
Challenges, Comparative Landscape, and Future Directions
Challenges include standardization of selenium content, pharmacokinetic studies in humans with impaired liver function, and long-term safety (though organic forms show minimal bioaccumulation) [40,41]. Methodological gaps in preclinical work (e.g., lack of blinding in many polysaccharide studies) necessitate rigorous Phase I/II trials [19,20].
The translation of selenized polysaccharides to clinical practice faces several interconnected challenges, alongside critical limitations in the existing preclinical evidence base.
First, standardization of selenization protocols and analytical methods remains an unresolved barrier, as noted earlier. There is no international consensus on reaction conditions for consistent selenization, standardized methods for selenium quantification and speciation, or mandatory reporting requirements for modification site confirmation and polysaccharide characterization. This heterogeneity severely limits cross-study comparability and the ability to pool data for meta-analysis, and is a prerequisite for GMP-compliant production of clinical-grade material.
Second, pharmacokinetic and pharmacodynamic data in humans, particularly those with hepatic impairment, are completely absent. All existing efficacy data are derived from rodent models, and the preliminary HED estimates rely on unvalidated assumptions of linear dose proportionality. Patients with impaired hepatic function have altered ADME profiles for oral polysaccharides and selenium compounds, and dedicated Phase I pharmacokinetic and toxicokinetic studies are required to define the safe therapeutic window, particularly for long-term dosing.
Third, preclinical literature has significant methodological limitations that introduce risk of bias. A 2026 systematic review found that >60% of preclinical polysaccharide studies for hepatic fibrosis do not report pre-specified randomization or outcome assessor blinding, leading to high risk of performance and detection bias [49,55]. Additionally, many studies do not report negative or null findings, introducing publication bias. These limitations must be addressed in future preclinical work, with adherence to the ARRIVE 2.0 guidelines for in vivo animal research, to improve the rigor and translatability of findings.
Fourth, patient selection and predictive biomarkers for treatment responsiveness remain undefined in the existing literature, as addressed earlier. Without baseline selenium status stratification and fibrosis stage enrichment, clinical trials are at high risk of type II error, failing to detect efficacy in unselected patient populations.
Finally, while organic selenium formulations have a far more favorable safety profile than inorganic selenium, long-term safety data in patients with chronic liver disease are limited. The narrow therapeutic window of selenium requires careful dose-finding to avoid supratherapeutic exposure, particularly in selenium-sufficient patients, and long-term studies are needed to evaluate bioaccumulation risk [8,46].
Table 1 compares selenization to other common polysaccharide modification strategies, highlighting the unique advantages of selenization for liver disease: it directly augments endogenous antioxidant selenoproteins, targets multiple core fibrogenic pathways, and has a well-characterized safety profile for oral administration, outperforming unmodified polysaccharides and matching or exceeding other modified derivatives in multi-target antifibrotic efficacy. Comparatively, selenized agents outperform unmodified polysaccharides and match or exceed some small-molecule candidates in multi-targeting with better safety [21,56]. Synergies with nano-delivery or existing therapies warrant exploration [29,57].
|
Modification Type |
Polysaccharide Source |
Modification Site (Confirmation Method) |
Therapeutic Indication / Disease |
Primary Biological Effects |
In vivo Metabolic Changes |
Affected Signaling Pathways |
Reference |
|
Sulfation |
Ganoderma applanatum residue |
-OH groups (7.8% S content; FT-IR) |
Liver fibrosis (oxidative/inflammatory) |
Anti-fibrotic; antioxidant; anti-inflammatory |
↑SOD; ↓inflammatory cytokines |
TLR4/NF-κB↓ |
[42] |
|
Sea cucumber (fucoidan-derived Fuc-S) |
Fucose residues |
Acetaminophen-induced acute liver injury |
Anti-steatotic; hepatoprotective |
Improved glucose/lipid metabolism; ↓ALT |
Gut microbiota-SCFA-liver axis; oxidative stress↓ |
[43] |
|
|
Chinese yam (CYP → SCYP) |
-OH groups |
LPS-induced acute inflammation |
Anti-inflammatory |
↓Serum cytokines |
NF-κB↓ |
[44] |
|
|
Prunella vulgaris |
-OH groups |
Isoniazid-induced liver injury |
Anti-inflammatory |
↑SOD; ↓IL-6/TNF-α |
Cytokine reduction |
[45] |
|
|
Chrysanthemum indicum |
C-6/C-2/C-4 (FT-IR/NMR) |
General bioactivity enhancement |
Antioxidant; anti-inflammatory; antitumor |
Improved solubility & bioavailability |
Not specified |
[46] |
|
|
Carboxymethylation |
Chitosan derivatives |
C-6 (preferred) |
Tumor / inflammation |
Enhanced water solubility; antitumor |
↑Cellular uptake |
Not specified |
[47] |
|
Pectin |
Galacturonic acid backbone |
Drug delivery / anti-inflammatory |
Improved emulsifying; controlled release |
Enhanced stability in GI tract |
Not specified |
[48] |
|
|
Phosphorylation |
Starch / general polysaccharides |
-OH groups |
Antitumor; antioxidant |
Radical scavenging; improved solubility |
Not specified |
Not specified |
[49] |
|
Schisandra chinensis derivatives |
-OH groups |
NAFLD / liver injury |
Lipid metabolism regulation |
↓Hepatic steatosis |
Nrf2/JNK1/AMPK |
[50] |
|
|
Acetylation |
Stropharia rugosoannulata |
-OH groups |
NAFLD / liver injury |
Antioxidant; lipid regulation |
Alleviated HFD-induced injury |
Nrf2/JNK1/AMPK |
[50] |
|
Chitosan |
-OH / amino groups |
Antitumor; immunomodulatory |
Increased hydrophobicity; cytoprotective |
Not specified |
Not specified |
[51] |
|
|
Grafting (Polyphenol) |
Oat β-glucan |
-OH / α-methylene |
Antioxidant / antibacterial |
Enhanced radical scavenging; thermal stability |
Not specified |
Not specified |
[52] |
|
Nanoparticle Conjugation |
Bletilla striata / sodium alginate |
Surface coating |
Acute liver injury |
Protective; controlled release |
↓Liver damage markers |
Antioxidant pathways |
[53] |
|
Physical (Ultrasonic) |
Citrus pectin / Grifola frondosa |
Glycosidic bonds |
Oxidative stress / ethanol damage |
Improved solubility; prebiotic; cytoprotective |
Not specified |
Not specified |
[54] |
To address the above challenges and advance selenized polysaccharides from bench to bedside, we outline the following evidence-based priorities for future research:
- Conduct a systematic review with meta-analysis of preclinical antifibrotic efficacy: A dedicated, PRISMA-compliant systematic review with quantitative meta-analysis is required to pool preclinical efficacy data, assess risk of bias across studies, and identify sources of heterogeneity (including selenium content, polysaccharide source, and dosing regimen). This will provide the highest level of preclinical evidence to support clinical trial design.
- Validate pharmacokinetics in models of hepatic impairment: Dedicated ADME and toxicokinetic studies in rodent models of cirrhosis are required to define hepatic accumulation, metabolic fate, and bioaccumulation risk of selenized polysaccharides, prior to human trials.
- Design and execute biomarker-stratified Phase I/II trials targeting F2–F3 MASLD: We propose a Phase I dose-escalation study to define safety and pharmacokinetics in healthy volunteers, followed by a Phase II randomized, double-blind, placebo-controlled trial in patients with biopsy-confirmed F2–F3 MASLD and baseline selenium deficiency (<70 μg/L). The primary endpoint of the Phase II trial will be change in liver stiffness (via transient elastography) at 48 weeks, with secondary endpoints including change in non-invasive fibrosis biomarkers (ELF score, PRO-C3), serum antioxidant capacity, and gut microbiome composition.
- Establish international consensus standards for selenization protocols and analytical methods: A multi-stakeholder working group (including chemists, pharmacologists, and hepatologists) should develop consensus guidelines for selenization reaction conditions, mandatory analytical characterization (selenium quantification, speciation, and modification site confirmation), and reporting standards for preclinical studies, to improve reproducibility and cross-study comparability.
- Explore synergistic combinations with GLP-1 agonists or emerging anti-fibrotics: Preclinical studies are needed to evaluate the additive or synergistic antifibrotic efficacy of selenized polysaccharides in combination with GLP-1 receptor agonists, emerging investigational antifibrotics, and microbiome-targeted therapies, to define optimal combination regimens for clinical use.
- Broader GI applications: Beyond hepatic fibrosis, these agents may have therapeutic potential in other GI disorders driven by oxidative stress and fibrosis, including pancreatic fibrosis, intestinal strictures in Crohn’s disease, and radiation-induced enteritis [58,59]. Patient-derived organoids and real-world evidence from TCM-integrated clinics will accelerate translation [60,61].
Conclusion
Selenium-modified natural polysaccharides exemplify how targeted chemical enhancement of traditional bioactive compounds can yield clinically promising agents for hepatic fibrosis. By amplifying antioxidant defense, suppressing inflammation, and inhibiting fibrogenic signaling, they address core unmet needs in gastroenterology and hepatology. Supported by robust mechanistic data, epidemiological correlations, and systematic preclinical evidence, these agents hold substantial potential as safe adjuncts that could slow or reverse fibrosis progression and improve outcomes in diverse liver disease populations.
Continued investment in translational research—standardized formulations, well-designed clinical trials, and integrative protocols—will determine their ultimate role in modern gastroenterological care. The field stands at an exciting intersection of natural-product innovation and evidence-based medicine, offering hope for patients facing the growing burden of fibrotic liver disease.
Financial Support
We are grateful for financial supports below:
Key Research and Development Program of Shaanxi Provincial Department of Science and Technology - Key Industrial Innovation Chain (Cluster) (Social Development Field), Research and Application on Green Planting and Fresh-cutting of Atractylodes lancea, Bletilla striata, and Gynostemma pentaphyllum, 2024-2027, 2024SF2 - GJHX - 40;
Key Project of Agricultural Key Technology Research and Development of Xi'an Science and Technology Bureau, Innovation, Demonstration and Promotion of Production Technologies for High - quality Bletilla striata, Cremastra appendiculata, and Polygonatum sibiricum, 2025-2028, 25NJSZD00003
Conflicts of Interest
Authors confirm there are no conflicts of interest.
Credit Author Statement
Yuwen Wang: Conceptualization; Data curation; Formal analysis; Funding acquisition; Software; Supervision; Validation; Visualization; Writing–original draft.
Junfeng Niu: Investigation; Methodology; Project administration; Resources; Writing–review & editing.
References
2. Trefts E, Gannon M, Wasserman DH. The liver. Curr Biol. 2017 Nov 6;27(21):R1147–151.
3. GBD 2021 Diseases and Injuries Collaborators. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2024 May 18;403(10440):2133–61.
4. Roehlen N, Crouchet E, Baumert TF. Liver Fibrosis: Mechanistic Concepts and Therapeutic Perspectives. Cells. 2020 Apr 3;9(4):875.
5. Bataller R, Brenner DA. Liver fibrosis. J Clin Invest. 2005 Feb;115(2):209–18.
6. Lin L, Zhou F, Shen S, Zhang T. Fighting Liver Fibrosis with Naturally Occurring Antioxidants. Planta Med. 2018 Dec;84(18):1318–33.
7. Hu W, Ma Z, Jiang S, Fan C, Deng C, Yan X, et al. Melatonin: the dawning of a treatment for fibrosis? J Pineal Res. 2016 Mar;60(2):121–31.
8. Shan L, Liu Z, Ci L, Shuai C, Lv X, Li J. Research progress on the anti-hepatic fibrosis action and mechanism of natural products. Int Immunopharmacol. 2019 Oct;75:105765.
9. Zhao H, Li D, Li M, Liu L, Deng B, Jia L, et al. Coprinus comatus polysaccharides ameliorated carbon tetrachloride-induced liver fibrosis through modulating inflammation and apoptosis. Food Funct. 2022 Oct 31;13(21):11125–41.
10. Chen X, Shen M, Yu Q, Chen Y, Xie J. Recent advance in chemistry modified methods of natural polysaccharides and their applications. Trends Food Sci. Technol. 2024 Feb 1;144:104317.
11. Wang L, Li L, Gao J, Huang J, Yang Y, Xu Y, et al. Characterization, antioxidant and immunomodulatory effects of selenized polysaccharides from dandelion roots. Carbohydr Polym. 2021 May 15;260:117796.
12. Mangiapane E, Pessione A, Pessione E. Selenium and selenoproteins: an overview on different biological systems. Curr Protein Pept Sci. 2014;15(6):598–607.
13. Mehdi Y, Hornick JL, Istasse L, Dufrasne I. Selenium in the environment, metabolism and involvement in body functions. Molecules. 2013 Mar 13;18(3):3292–311.
14. Reja M, Makar M, Visaria A, Marino D, Rustgi V. Increased serum selenium levels are associated with reduced risk of advanced liver fibrosis and all-cause mortality in NAFLD patients: National Health and Nutrition Examination Survey (NHANES) III. Ann Hepatol. 2020 Nov-Dec;19(6):635–40.
15. Wang L, Du D, Ying X, Tang Y, Dong S, Shang H, et al. L-shaped association of selenium in blood metal mixtures with the incidence of liver fibrosis/cirrhosis: NHANES 2017-2020. Int J Surg. 2025 Dec 1;111(12):8837–48.
16. Guo W, Weng T, Song Y. Association of serum selenium with MASLD and liver fibrosis: A cross-sectional study. PLoS One. 2024 Dec 31;19(12):e0314780.
17. He G, Karin M. NF-κB and STAT3 - key players in liver inflammation and cancer. Cell Res. 2011 Jan;21(1):159–68.
18. Chen Y , Zhao YF , Yang J , Jing HY , Liang W , Chen MY , et al . Selenium alleviates lipopolysaccharide-induced endometritis via regulating the recruitment of TLR4 into lipid rafts in mice. Food Funct. 2020 Jan 29;11(1):200–10.
19. Wang J, Ye Y, Jiang W, Yang H, Xu J, Han Q, et al. Systematic Review of Preclinical Evidence on Antifibrotic Potential of Natural Polysaccharides. Polysaccharides. 2026 Feb 5;7(1):19.
20. Wang J, Wang H, Yang X, Wang K, Zhang Y. Recent advances in the role of polysaccharides in liver diseases: a review. Front Pharmacol. 2025 Mar 27;16:1535717.
21. Cheng K, Sun Y, Liu B, Ming J, Wang L, Xu C, et al. Selenium Modification of Natural Products and Its Research Progress. Foods. 2023 Oct 13;12(20):3773.
22. Zhao M, Bai J, Bu X, Yin Y, Wang L, Yang Y, et al. Characterization of selenized polysaccharides from Ribes nigrum L. and its inhibitory effects on α-amylase and α-glucosidase. Carbohydr Polym. 2021 May 1;259:117729.
23. Huo Y, Ding WJ, Liu YR, Li ZT, Dai KY, Liu C, et al. Selenochemical modification of low molecular weight polysaccharides from Grifola frondosa and the mechanism of their inhibitory effects on gastric cancer cells. Int J Biol Macromol. 2024 Jun;269(Pt 1):131812.
24. Deng J, Min J, Zhang Y, You R, Zhang Z, Hu Y, et al. Preparation, characterization and cytotoxicity assessment of a novel selenized polysaccharide from Morchella sextelata. Int J Biol Macromol. 2024 Apr;265(Pt 2):131100.
25. Li Q, Zhu L, Qi X, Zhou T, Li Y, Cai M, et al. Immunostimulatory and antioxidant activities of the selenized polysaccharide from edible Grifola frondosa. Food Sci Nutr. 2022 Feb 2;10(4):1289–98.
26. Peng K, Yue L, Song X, Zhang Q, Wang Y, Cui X. Preparation, characterization and evaluation of microwave-assisted synthesized selenylation Codonopsis pilosula polysaccharides. Int J Biol Macromol. 2024 Jul;273(Pt 2):133228.
27. Zhang Z, Zhang Y, Liu H, Wang J, Wang D, Deng Z, et al. A water-soluble selenium-enriched polysaccharide produced by Pleurotus ostreatus: Purification, characterization, antioxidant and antitumor activities in vitro. Int J Biol Macromol. 2021 Jan 31;168:356–70.
28. Xu Y, Wang XC, Jiang W, Hu JN. Angelica sinensis polysaccharides modified selenium nanoparticles for effective prevention of acute liver injury. Int J Biol Macromol. 2024 Apr;263(Pt 1):130321.
29. Xu Y, Li F, Min W. Mitigating effect of selenium nanoparticles modified with Lycium barbarum polysaccharide on acute liver injury. Int J Biol Macromol. 2025 Dec;334(Pt 2):149142.
30. Gao Z, Li J, Song X, Zhang J, Wang X, Jing H, et al. Antioxidative, anti-inflammation and lung-protective effects of mycelia selenium polysaccharides from Oudemansiella radicata. Int J Biol Macromol. 2017 Nov;104(Pt A):1158–64.
31. Liu M, Meng G, Zhang J, Zhao H, Jia L. Antioxidant and Hepatoprotective Activities of Mycelia Selenium Polysaccharide by Hypsizigus marmoreus SK-02. Biol Trace Elem Res. 2016 Aug;172(2):437–48.
32. Sun X, Zheng Y, Tian Y, Xu Q, Liu S, Li H, et al. Astragalus polysaccharide alleviates alcoholic-induced hepatic fibrosis by inhibiting polymerase I and transcript release factor and the TLR4/JNK/NF-κB/MyD88 pathway. J Ethnopharmacol. 2023 Oct 5;314:116662.
33. Wang K, Wang J, Song M, Wang H, Xia N, Zhang Y. Angelica sinensis polysaccharide attenuates CCl4-induced liver fibrosis via the IL-22/STAT3 pathway. Int J Biol Macromol. 2020 Nov 1;162:273–83.
34. Dai X, Du Z, Jin C, Tang B, Chen X, Jing X, et al. Inulin-like polysaccharide ABWW may impede CCl4 induced hepatic stellate cell activation through mediating the FAK/PI3K/AKT signaling pathway in vitro & in vivo. Carbohydr Polym. 2024 Feb 15;326:121637.
35. Zha A, Tu R, Qi M, Wang J, Tan B, Liao P, et al. Mannan oligosaccharides selenium ameliorates intestinal mucosal barrier, and regulate intestinal microbiota to prevent Enterotoxigenic Escherichia coli -induced diarrhea in weaned piglets. Ecotoxicol Environ Saf. 2023 Oct 1;264:115448.
36. Wang K, Yang X, Wu Z, Wang H, Li Q, Mei H, et al. Dendrobium officinale Polysaccharide Protected CCl4-Induced Liver Fibrosis Through Intestinal Homeostasis and the LPS-TLR4-NF-κB Signaling Pathway. Front Pharmacol. 2020 Mar 12;11:240.
37. Shang D, Zhang J, Wen L, Li Y, Cui Q. Preparation, characterization, and antiproliferative activities of the Se-containing polysaccharide SeGLP-2B-1 from Se-enriched Ganoderma lucidum. J Agric Food Chem. 2009 Sep 9;57(17):7737–42.
38. Tao W, Liu D, Guo Z, Han P, Ma Y, Wu M, et al. Physicochemical properties, structural characterization, and antidiabetic activity of selenylated low molecular weight apple pectin in HFD/STZ-induced type 2 diabetic mice. Carbohydr Polym. 2025 Jan 15;348(Pt A):122790.
39. Alahmar AT. The Effect of Selenium Therapy on Semen Parameters, Antioxidant Capacity, and Sperm DNA Fragmentation in Men with Idiopathic Oligoasthenoteratospermia. Biol Trace Elem Res. 2023 Dec;201(12):5671–76.
40. Winkel LH, Johnson CA, Lenz M, Grundl T, Leupin OX, Amini M, et al. Environmental selenium research: from microscopic processes to global understanding. Environ Sci Technol. 2012 Jan 17;46(2):571–9.
41. Jain VK, Priyadarsini KI. Selenium: a wonder element in life and for life. Proceedings of the National Academy of Sciences, India Section a: Physical Sciences. 2024 Feb;94(1):1–10.
42. Song X, Cui W, Gao Z, Zhang J, Jia L. Structural characterization and amelioration of sulfated polysaccharides from Ganoderma applanatum residue against CCl4-induced hepatotoxicity. Int Immunopharmacol. 2021 Jul;96:107554.
43. Du B, Cheng C, Feng J, Liang Y, Tao W, Xu B, et al. Fuc-S, a New Degraded Polysaccharide From Fucoidan, Alleviates Type 2 Diabetes-Associated Liver Injury Through Modulating Gut Microbiota. Food Sci Nutr. 2026 Jan 16;14(1):e71321.
44. Wu S, Chen X, Cai R, Chen X, Zhang J, Xie J, et al. Sulfated Chinese Yam Polysaccharides Alleviate LPS-Induced Acute Inflammation in Mice through Modulating Intestinal Microbiota. Foods. 2023 Apr 25;12(9):1772.
45. WANG Y, CAO R, ZHU C, WU X. Effect and mechanism of Prunella vulgaris sulfated polysaccharide on angiogenesis in hepatocellular carcinoma. Chinese Journal of Clinical Oncology. 2014:758–61.
46. Li X, Wu Z, Shen H, Song Q, Luo G, Qiao X, et al. Carboxymethylated polysaccharides from Chrysanthemum indicum with antimicrobial and hemostatic properties for promoting wound healing. Int J Biol Macromol. 2025 Apr;304(Pt 1):140899.
47. Chakka VP, Zhou T. Carboxymethylation of polysaccharides: Synthesis and bioactivities. Int J Biol Macromol. 2020 Dec 15;165(Pt B):2425–31.
48. Zhao Y, Wang M, Li H, Zhang Z, Li H. Advances in Modification Strategies of Polysaccharides and Their Applications. J Food Sci. 2026 Jan;91(1):e70779.
49. Xia S, Zhai Y, Wang X, Fan Q, Dong X, Chen M, et al. Phosphorylation of polysaccharides: A review on the synthesis and bioactivities. Int J Biol Macromol. 2021 Aug 1;184:946–54.
50. Li X, Cui W, Cui Y, Song X, Jia L, Zhang J. Stropharia rugoso-annulata acetylated polysaccharides alleviate NAFLD via Nrf2/JNK1/AMPK signaling pathways. Int J Biol Macromol. 2022 Aug 31;215:560–70.
51. Jiang Y, Li S, Chang M, Bian T, Wu W, Li T, et al. Acetylated chitosan scaffolds modulate the micro-environment and promote peripheral nerve regeneration through macrophage TLR2/4 pathway activation. Mater Today Bio. 2025 Oct 1;35:102376.
52. Luo Y, Li YC, Wang M, Zhou L, Meng FB, Jiang LS. Effects of grafting methods and raw materials on the physicochemical properties and biological activities of phenolic acids grafted oat β-glucan. Food Res Int. 2023 Nov;173(Pt 1):113250.
53. Zhao Y, Wang J, Zhang Y, Liu C, Chen Y, Li P, et al. Bletilla striata polysaccharide-based dissolving microneedle patch integrated with nanoparticles for promoting hair regrowth. Int J Biol Macromol. 2025 Apr;303:140336.
54. Wang Z, Zhou X, Sheng L, Zhang D, Zheng X, Pan Y, et al. Effect of ultrasonic degradation on the structural feature, physicochemical property and bioactivity of plant and microbial polysaccharides: A review. Int J Biol Macromol. 2023 May 1;236:123924.
55. Wang Y, Ye H, Qiao L, Du C, Wei Z, Wang T, et al. Intestinal Anti-Inflammatory Effects of Selenized Ulva pertusa Polysaccharides in a Dextran Sulfate Sodium-Induced Inflammatory Bowel Disease Model. J Med Food. 2021 Mar;24(3):236–47.
56. Cheng L, Wang Y, He X, Wei X. Preparation, structural characterization and bioactivities of Se-containing polysaccharide: A review. Int J Biol Macromol. 2018 Dec;120(Pt A):82–92.
57. Cui D, Ma J, Liang T, Sun L, Meng L, Liang T, et al. Selenium nanoparticles fabricated in laminarin polysaccharides solutions exert their cytotoxicities in HepG2 cells by inhibiting autophagy and promoting apoptosis. Int J Biol Macromol. 2019 Sep 15;137:829–35.
58. Cai X, Hu Z, Zhang M, Dang Q, Yang Q, Zhao X, et al. Dosage-effect of selenium supplementation on blood glucose and oxidative stress in type 2 diabetes mellitus and normal mice. J Trace Elem Med Biol. 2024 May;83:127410.
59. Yang Z, Lian J, Yang Y, Li J, Guo W, Lv X, et al. Selenium enrichment enhances the alleviating effect of Lactobacillus rhamnosus GG on alcoholic liver injury in mice. Curr Res Food Sci. 2024 Dec 18;10:100964.
60. Zhao H, Liu C, Song J, Fan X. Pilot study of toxicological safety evaluation in acute and 28-day studies of selenium nanoparticles decorated by polysaccharides from Sargassum fusiforme in Kunming mice. J Food Sci. 2022 Sep;87(9):4264–79.
61. He H, Huang D, Xie P, Dong J, Huo S, Li S, et al. Research progress on selenium system in cancer therapy: focus on interface modifications and improvement of interactions. Surfaces and Interfaces. 2025 Jan 1;56:105642.
