Abstract
Myocardial ischemia is a frequent finding in hypertrophic cardiomyopathy (HCM) potentially resulting from microvascular dysfunction, extravascular compression, reduced coronary flow reserve, hemodynamic abnormalities, or concomitant atherosclerotic coronary artery disease (CAD). From June 1991 to October 2024, 40 congenital coronary artery anomalies (CCAA) were reported in 30 adults with HCM [age 18–87 (47.6 ± 19.7); 60% male; 37% obstructive; 20% apical variant]. Clinical data on ischemia, therapy, and outcomes were reviewed. Coronary anatomy was categorized by number of ostia: single (n=12; 40%), two (n=13; 43%), three (n=4; 13%), or four (n=1; 4%). The anomalous vessel was the left main (20%), left anterior descending (25%), left circumflex (18%), right coronary artery (35%), or ramus intermedius (2%). Two patients (7%) with apical HCM also had coronary fistulae. Myocardial ischemia was present in 13 patients (43%), including 5 with anomalous vessels coursing between the great vessels. Two of five patients with sudden cardiac arrest had a high-risk anomaly. Pharmacologic therapy was provided to 13 (43%), coronary surgery to 3 (10%), and septal reduction to 8 (27%). Among 15 patients with follow-up, all reported symptomatic improvement. CCAA may occur in HCM and contribute to ischemia; management should be guided by anomaly type and objective evidence of ischemia.
Keywords
Hypertrophic cardiomyopathy, HCM, Coronary artery, Coronary anomalies, Myocardial ischemia
Abbreviations
CAD: Coronary Artery Disease; CCAA: Congenital Coronary Artery Anomalies HCM: Hypertrophic Cardiomyopathy SCA: Sudden Cardiac Arrest
Introduction
Myocardial ischemia is frequently detected in adults with hypertrophic cardiomyopathy (HCM) [1] and is commonly attributed to microvascular disease, extravascular compressive forces, reduced coronary flow reserve, hemodynamic abnormalities, and concomitant atherosclerotic coronary artery disease (CAD) [2]. Congenital coronary artery anomalies (CCAA) may be present in some adults with HCM with potential independent contribution to myocardial ischemia. However, such association has not been systematically evaluated. We aimed to examine the spectrum of reported CCAA in adults with HCM and their potential association with myocardial ischemia.
Methods
A systematic, worldwide literature search, using keywords of “hypertrophic cardiomyopathy and congenital coronary anomalies”, was conducted across three databases: PubMed/MEDLINE, Google Scholar, and the Temple University Library search engine to identify published case reports and case series highlighting patients with both HCM and CCAA (Figure 1). In addition to database searching, reference lists of all retrieved articles were manually screened to identify additional eligible studies not captured through the primary search. Studies were included if they met all of the following criteria: (1) reported at least one patient; (2) included a diagnosis of HCM as reported by the original authors; (3) described the presence of one or more congenital coronary artery anomalies; and (4) provided sufficient patient-level data to identify both patient demographics (age and sex) and the type and identity of the anomalous coronary vessel and its anatomy. Case reports and case series were eligible. No language restrictions were applied.
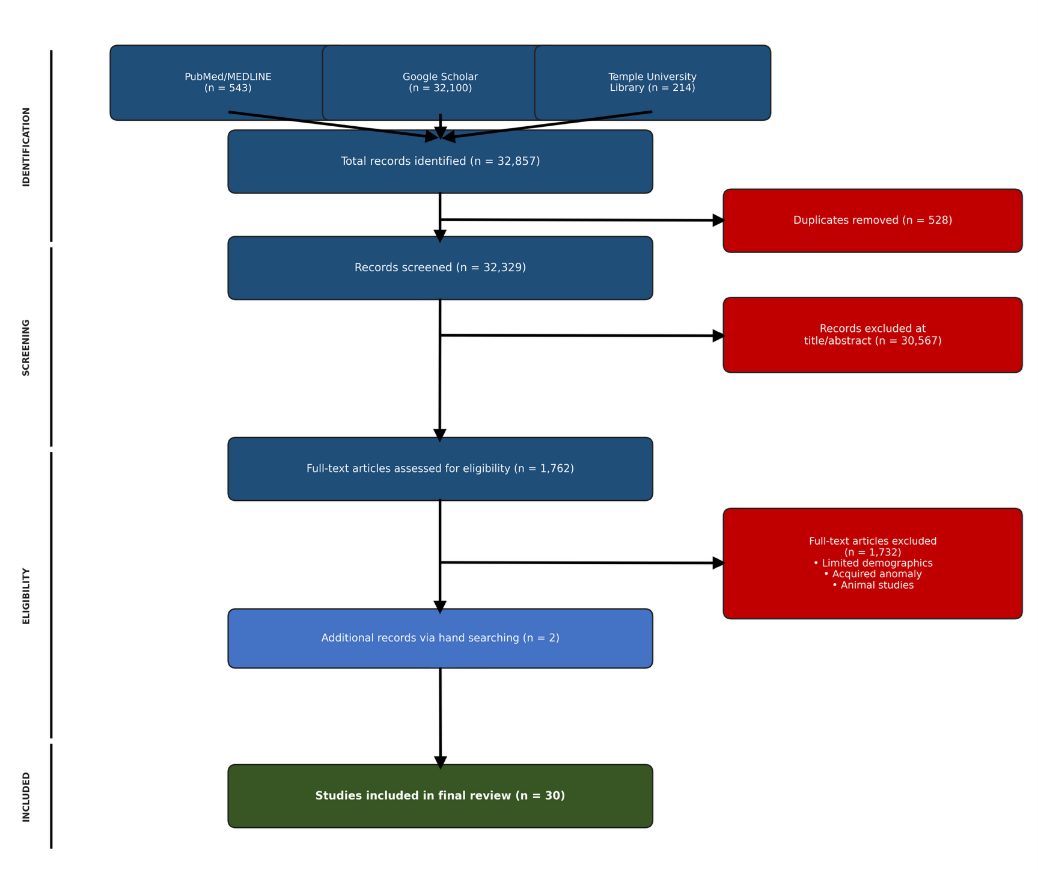
Figure 1. PRISMA flow diagram illustrating the systematic search and study selection.
Exclusion criteria were as follows: (1) animal studies; (2) cases in which the coronary anomaly was acquired rather than congenital in etiology (e.g., secondary to Kawasaki disease or atherosclerosis); (3) cases lacking patient demographic data (age or sex) or an adequate description of the coronary anomaly; and (4) duplicate reports identified as describing the same patient across multiple publications, in which case only the most detailed or original report was retained.
Clinical data on patient history, family history, the anomalous vessel(s), presence or absence of myocardial ischemia, mode of therapy, and outcomes were extracted from each case. A subset of these characteristics can be found in Table 1. From June 1991 to October 2024, a total of 40 individual anomalous arteries were reported in 30 adults with HCM. Institutional review board approval was not required as all data were obtained from de-identified, previously published case reports and case series.
|
Figure #1 Reference |
Author [manuscript reference] |
Publication Year |
Journal |
Age |
HCM Phenotype |
Presence of Ischemia? |
|
XVII |
Liddy [20] |
2014 |
BMJ |
40 |
HCM |
No |
|
XXIII |
Moza [26] |
2011 |
AMJS |
29 |
HOCM |
NA |
|
XVIII |
Tyczynski [21] |
2013 |
Studium Przypadku |
65 |
HOCM |
Yes, dipyridamole induced |
|
VII |
Atmaca [10] |
2002 |
Journal of Invasive Cardiology |
38 |
HCM |
No |
|
XIII |
Niwayama [16] |
1991 |
Kokyu To Junkan |
47 |
HCM |
Yes, exercise stress induced |
|
VIII |
Lv [11] |
2022 |
Journal of Cardiothoracic Surgery |
64 |
HOCM |
NA |
|
XIV |
Georgekutty [17] |
2014 |
Cardiology Young |
18 |
Apical HCM |
Yes |
|
I |
Chow [4] |
2009 |
The Canadian Journal of Cardio. |
70 |
Apical HCM |
Yes, dipyridamole induced |
|
XV |
Alqarqaz [18 |
2011 |
Journal of Cardiovascular Medicine |
44 |
HCM |
Yes, SPECT positive |
|
XXVI |
Ferreira [29] |
2008 |
International Journal of Cardiology |
26 |
HCM |
NA |
|
XXVIII |
Georgiadou [31] |
2006 |
International Journal of Cardiology |
68 |
HCM |
Yes, elevated troponins |
|
XXI |
Dermengiu [24] |
2010 |
Romanian Journal of Legal Medicine |
20 |
HCM |
NA |
|
XXX |
Beach [33] |
2001 |
Arch Pathol Lab Med |
49 |
HCM |
NA |
|
XXVIII |
Bush [30] |
2005 |
Images in Cardiovascular Medicine |
87 |
HOCM |
NA |
|
XXIX |
Cifcti [32] |
2009 |
Cardiology in the Young |
31 |
HCM |
Yes, exercise stress induced |
|
IX |
Zuccarino [12 |
2009 |
European Soceity of Cardiology |
23 |
HCM |
Yes, elevated troponins |
|
X |
Kurflaklloglu [13] |
2005 |
Olgu Sunumu |
22 |
HOCM |
NA |
|
III |
Serino [6] |
1998 |
Heart |
71 |
HOCM |
NA |
|
IV |
Hara [7] |
2006 |
Journal of Invasive Cardiology |
80 |
HOCM |
NA |
|
XI |
Efthimiadis [14] |
2013 |
Herz |
45 |
HCM |
No |
|
V |
Muller [8] |
2023 |
JACC |
77 |
Apical HCM |
Yes, elevated troponins |
|
XX |
Santos [23] |
2017 |
JACC |
57 |
Apical HCM |
Yes, dobutamine stress induced |
|
XIX |
Sarkar [22] |
2019 |
CHEST |
66 |
Apical HCM |
Yes, exercise stress induced |
|
VI |
Penciu [9] |
2014 |
Ann Thoracic Surgery |
48 |
HOCM |
Yes |
|
XXIV |
Sato [27] |
2016 |
HEARTS Original |
41 |
HCM |
NA |
|
XXII |
Zheng [25] |
2019 |
BMC Cardiovascular Disorders |
26 |
HCM |
NA |
|
XII |
Sinha [15] |
2022 |
ARYA Athero |
21 |
HOCM |
No |
|
XVI |
Guo [19] |
2015 |
Angiology |
55 |
Apical HCM |
No |
|
XXV |
Woznica [28] |
2018 |
Kardiologia Polska |
61 |
HOCM |
NA |
|
II |
O'Gorman [5] |
2024 |
JACC Case Report |
39 |
HOCM |
Yes, elevated troponins |
|
NA: Not Available; HCM: Non-obstructive Hypertrophic Cardiomyopathy; HOCM: Obstructive Hypertrophic Cardiomyopathy; SPECT: Single Photon Emission Computerized Tomography |
||||||
Results
Patients were 18 to 87 (47.6 ±19.7) years old at the time of report; 60% were male; 37% had an obstructive phenotype of HCM while 20% demonstrated the apical variant. The coronary anatomy of the individual cases were graphically reproduced (Figure 2) and were categorized based on the number of coronary ostia present in the aorta {single ostium [n=12 (40%)]; 2 ostia [n=13 (43%)]; 3 ostia [n=4 (13%)] and 4 ostia [n=1 (4%)}. The anomalous coronary artery was the left main coronary artery in 8 (20%); left anterior descending coronary artery in 10 (25%); left circumflex coronary artery in 7 (18%); right coronary artery in 14 (35%) or the ramus intermedius coronary artery in 1 (2%). Two patients (7%) with apical variant HCM also had coronary artery fistulae in the left anterior descending coronary artery. Overall, there were 38 instances of anomalous origin, 33 instances of anomalous course and 2 instances of anomalous termination (fistulae) among the congenitally abnormal coronary arteries. Of the 30 patients reviewed, 13 (43%) showed objective evidence of myocardial ischemia with 5 (38%) having an anomalous coronary artery coursing in between the great arteries. In addition, 2 of 5 patients with sudden cardiac death had a high-risk coronary anomaly, defined as either having a slit-like ostium, acute take-off angle or coursing in between the great arteries. Pharmacologic treatment was offered to 13 (43%) patients, and 3 (10%) patients underwent coronary artery surgery (bypass grafting in 2 and unroofing an intramural epicardial coronary artery in 1). Septal reduction therapy was performed in 8 (27%). Follow up data was available in 15 patients including 2 of the 3 who underwent surgical correction. Symptomatic improvement was noted on follow up in all 15.
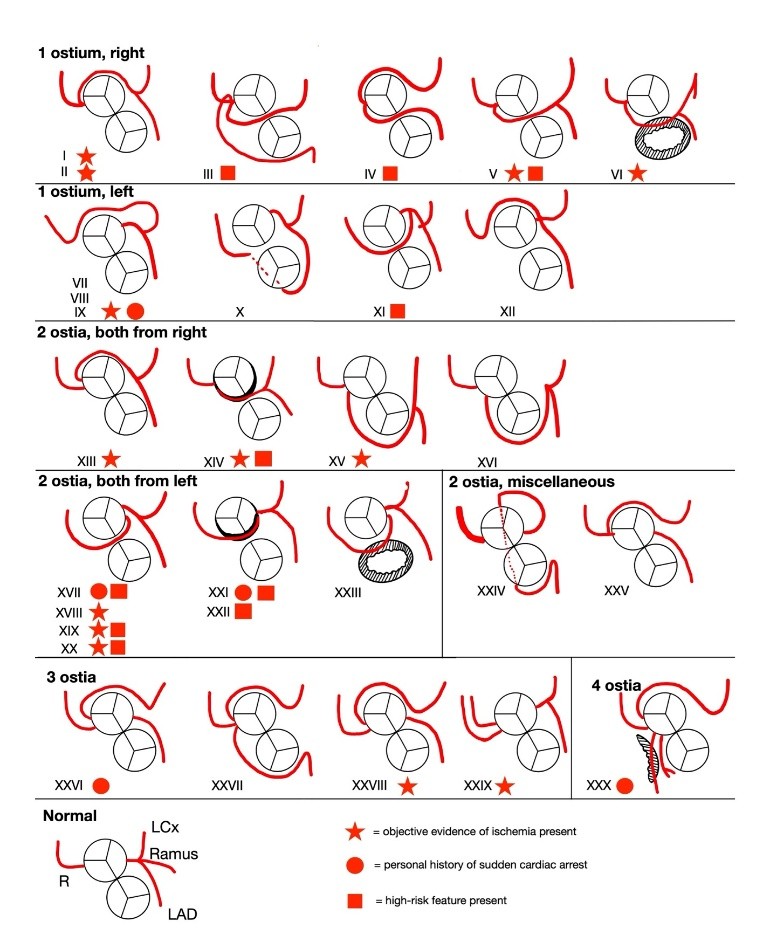
Figure 2. Graphical representation of origin and course of coronary artery anomalies.
Discussion
CCAA refers to a heterogeneous group of coronary malformations divided into three main categories: anomalies of origin, anomalies of course, and anomalies of termination [3]. The literature of each main CCAA category within HCM patients is limited; however, one of the most studied sub-variants are myocardial bridges which are nearly twice as prevalent in HCM patients and are theorized to potentially contribute to myocardial ischemia and SCD [4]. Beyond myocardial bridges, there is sparse literature which describes anomalies of origin and anomalies of termination in HCM patients.
Our study shows that CCAA may be concomitantly present in patients with phenotypic HCM and in certain cases (~43%) may be associated with myocardial ischemia. Of interest, most reported cases (~63%) had the non-obstructive phenotype and the 2 patients with coronary cameral fistula had the apical variant of HCM [34]. While the right coronary was involved in ~35% of these anomalies, the remaining cases were similarly distributed among left main (8), left descending (10) and left circumflex (7) coronary arteries. Overall, coronary anatomy varied widely among patients with HCM and no predominant specific anomaly was identifiable. There were no instances of tunneled (bridged) coronary artery among patients with anomalous coronary artery.
Among the 30 cases, 12 (40%) were considered to have the “high risk” features of coursing in between the great vessels, having a slit-like ostium, or having an acute take-off angle. Ischemia was present in 5 of the latter 12 patients. Only 2 of 5 patients with sudden cardiac arrest (SCA) had a “high risk” coronary anomaly. Further research into the risk of SCD in patients with HCM and CCAAs will help optimize treatment of this population.
While it can be speculated that the presence of CCAA would heighten risk of sudden cardiac death among competitive athletes, only one of the cases reported died suddenly during soccer training session. Based on our observations from these published case reports, patients with CCAA and HCM present in three main categories: asymptomatic and subsequently found incidentally, symptomatic with recurrent angina or syncope on exertion found via echocardiogram or coronary angiography, and post-mortem autopsies secondary to SCA.
Conclusion
The true prevalence of CCAA in patients with HCM is not known. CCAA is rarely acknowledged as a potential contributor to myocardial ischemia in the presence of multiple other potential etiologies (microvascular dysfunction, extravascular compressive forces, reduced coronary flow reserve, hemodynamic abnormalities, intramyocardial course, and concomitant atherosclerotic coronary artery disease) in patients with HCM. While evidence-based treatment strategies are lacking for specific management of coronary anomalies in patients with HCM, application of general approaches to CCAA appears to have been effective in most cases. Further studies are needed to understand the true scope of CCAA in HCM and its impact on treatment and outcomes of the patients.
Conflicts of Interest
The authors declare that we have no conflicts of interest.
Funding
Not applicable.
Acknowledgements
Not applicable.
Author Contributions
LM: Conceptualization, Data curation, Formal analysis, Investigation, Visualization, Writing-original draft, Writing-review & editing. MS, KG, NR: Writing-original draft, Writing-review and editing. JS: Conceptualization, Formal analysis, Supervision, Validation, Writing-original draft, Project administration, Writing-review & editing. All authors read and approved the submitted version.
References
2. Coleman JA, Ashkir Z, Raman B, Bueno-Orovio A. Mechanisms and prognostic impact of myocardial ischaemia in hypertrophic cardiomyopathy. Int J Cardiovasc Imaging. 2023;39(10):1979–96.
3. Gentile F, Castiglione V, De Caterina R. Coronary Artery Anomalies. Circulation. 2021;144(12):983–96.
4. Chow B, Ling M, Ascah K. Single coronary artery in a patient with apical variant hypertrophic cardiomyopathy. Can J Cardiol. 2009;25(6):e205.
5. O’Gorman KJ, Moulton MN, Gustafson JD, Cahill MS, Gore RS. Patient with an anomalous left coronary artery and hypertrophic obstructive cardiomyopathy. JACC Case Rep. 2024;29(19):102571.
6. Serino W, Sigwart U. Septal ablation in a patient with hypertrophic obstructive cardiomyopathy and a unique variant of anomalous origin of the left coronary artery. Heart. 1998;79(6):629–30.
7. Hara H, Ishii K, Nakamura M. A case of hypertrophic obstructive cardiomyopathy complicated by a single coronary artery treated by transcoronary septal ablation. J Invasive Cardiol. 2006;18(5):234–8.
8. Muller L, Patel S, Mehlhorn D, et al. Right-sided single coronary artery and apical hypertrophic cardiomyopathy. J Am Coll Cardiol. 2023;81(8_Suppl):3515.
9. Penciu OM, Mojibian H, Sugeng L, Cleman M, Brennan J, DePasquale E, et al. Anomalous left coronary artery in hypertrophic cardiomyopathy. Ann Thorac Surg. 2014 Jun;97(6):2190–3.
10. Atmaca Y, Dandachi R, Oral D. A rare variant of single coronary artery and non-obstructive hypertrophic cardiomyopathy. J Invasive Cardiol. 2002 Aug;14(8):469–70.
11. Lv X, Li J, Liu C, Wei R, Meng L, Kong X, et al. Single coronary ostium with obstructive hypertrophic cardiomyopathy treated using the Morrow procedure: a case report. J Cardiothorac Surg. 2022 Dec 28;17(1):340.
12. Zuccarino F, Moral S, Pujol E. Sudden death: hypertrophic cardiomyopathy with myocardial scarring and extremely rare coronary origin. Eur Heart J. 2009 Jun;30(11):1355.
13. Kurşaklioğlu H, Iyisoy A, Celik T, Barçin C, Köse S, Işik E. An unusual type of single coronary artery anomaly in a patient with hypertrophic obstructive cardiomyopathy. Anadolu Kardiyol Derg. 2005 Sep;5(3):232–3.
14. Efthimiadis GK, Theofilogiannakos EK, Gossios TD, Paraskevaidis S, Vassilikos VP, Styliadis IH. Hypertrophic cardiomyopathy associated with an anomalous origin of right coronary artery. Case report and review of the literature. Herz. 2013 Jun;38(4):427–30.
15. Sinha S, Aggarwal P, Samrat S, Razi M, Sharma A, Pandey U. Unusual trifurcation of left main coronary artery in a patient with hypertrophic obstructive cardiomyopathy: Issues in management strategy. ARYA Atheroscler. 2022 Jul;18(4):1–4.
16. Niwayama H, Morooka S, Takaoka N, Inagaki M, Yoshida H, Shukuya M, et al. [Hypertrophic cardiomyopathy associated with anomalous origin of the left coronary artery from the right sinus of valsalva]. Kokyu To Junkan. 1991 Jun;39(6):613–6.
17. Georgekutty J, Cross RR, Rosenthal JB, Heath DM, Sinha P, John AS. Anomalous left coronary artery from the right coronary cusp with gene positive apical hypertrophic cardiomyopathy: a case report and literature review. Cardiol Young. 2014 Jun;24(3):397–402.
18. Alqarqaz M, Zaidan M, Al-Mallah MH. Hypertrophic cardiomyopathy and anomalous left coronary artery: a rare combination. J Cardiovasc Med (Hagerstown). 2011 Dec;12(12):915–8.
19. Guo J, Xu R, Chen W. Left Main Coronary Artery Arising From the Right Sinus of Valsalva in a Patient with Apical Hypertrophic Cardiomyopathy. Angiol. 2015;3(157):2.
20. Liddy S, Roy AK, McCreery C. Double trouble: hypertrophic cardiomyopathy coexistent with malignant anomalous right coronary artery. BMJ Case Rep. 2014 Mar 28;2014:bcr2013203344.
21. Tyczyński P, Dębski A, Pręgowski J, Chojnowska L, Witkowski A. Hypertrophic obstructive cardiomyopathy and anomalous origin of the right coronary artery. A rare coexistence. Kardiol Pol. 2013;71(4):430.
22. Sarkar S, Leung S, Volodarskiy A. A RARE CASE OF AN ABERRANT CORONARY ARTERY CAUSING INCREASED RISK OF SUDDEN CARDIAC ARREST IN APICAL HYPERTROPHIC CARDIOMYOPATHY. Chest. 2019 Oct 1;156(4):A389.
23. Delos Santos JS, Pestano NS, Tangco RV. TCTAP C-205 Triple Threat: Anomalous Origin of the Right Coronary Artery Associated with Coronary-cameral Fistula and Apical Hypertrophic Cardiomyopathy. JACC. 2017 Apr 25;69(16S):S292.
24. Dermengiu D, Ceausu M, Rusu MC, Dermengiu S, Curca GC, Hostiuc S. Sudden death associated with borderline hypertrophic cardiomyopathy and multiple coronary anomalies. Case report and literature review. Rom J Leg Med. 2010 Mar 1;1:3–12.
25. Zheng GM, Bai J, Tang JM, Zhu FC, Jing HX. A case of hypertrophic cardiomyopathy combined with muscular ventricular septal defect and abnormal origin of right coronary artery. BMC Cardiovasc Disord. 2019 Jan 14;19(1):16.
26. Moza A, Prashar R, Bawany M. Anomalous origin of right coronary artery associated with hypertrophic obstructive cardiomyopathy. Am J Med Sci. 2011 Oct;342(4):341–2.
27. Sato T, Mizutani H, Murakami H, Imai Y, Taniguchi M, Okubo S, et al. A Case of Hypertrophic Cardiomyopathy with Congenital Left Anterior Descending Coronary Artery Agenesis and Giant Coronary Artery Aneurysm. Heart. 2016;48(12):1364–70.
28. Woźnica A, Tyczyński P, Brzozowski P, Skowroński J, Różański J, Witkowski A. Hypertrophic obstructive cardiomyopathy with anomalous left circumflex coronary artery. Kardiol Pol. 2018;76(7):1118.
29. Ferreira AM, Rizzo E, Redheuil A, Leclercq JF, Grenier O, Mousseaux E. An unusual combination of possible causes of sudden death imaged by 64-slice computed tomography. Int J Cardiol. 2008 Aug 29;128(3):e91–2.
30. Bush HS, Nolan J, Shen MH. Use of CT angiography for evaluation of 3 coronary ostia originating from the right sinus of Valsalva in a patient with hypertrophic obstructive cardiomyopathy. Tex Heart Inst J. 2005;32(2):246–7.
31. Georgiadou P, Sbarouni E, Kremastinos DT. Midventricular hypertrophic cardiomyopathy coexistent with anomalous origin of circumflex artery. Int J Cardiol. 2006 Jun 7;110(1):102–3.
32. Ciftci H, Tacoy G, Yazici G. Duplication of the right coronary artery in a patient with hypertrophic cardiomyopathy and myocardial bridging. Cardiol Young. 2009 Dec;19(6):627–9.
33. Beach L, Burke A, Chute D, Virmani R. Anomalous origin of 4 coronary ostia from the right sinus of Valsalva in a patient with hypertrophic cardiomyopathy. Arch Pathol Lab Med. 2001 Nov;125(11):1489–90.
34. Miller L, Shelly M, Graver K, Roma N, Cohen B, Shirani J. Coronary artery fistulae in patients with hypertrophic cardiomyopathy. Int J Cardiol Cardiovasc Dis. 2026;6(1):1-3.
