Abstract
Introduction: Our group has described the nitrergic pathway as a key analgesic mechanism of drugs and as part of the endogenous control of peripheral inflammatory pain. Studies have shown that epoxyeicosatrienoic acids (EETs) can activate different isoforms of nitric oxide synthase, leading to nitric oxide release and subsequent vasodilation.
Aim: Therefore, the goal of this study was to determine whether the nitrergic pathway mediates the peripheral antinociceptive effects of EETs.
Methods: The mechanical paw pressure test was used as an algesimetric method. Hyperalgesia was induced by intraplantar injection of prostaglandin E2 to assess the effects of exogenous EETs administration and its impact on inhibiting the nitrergic pathway. The EETs (5,6-, 8,9-, 11,12-, and 14,15-EET) and drugs targeting the nitrergic pathway were administered intraplantarly to male Swiss mice (n = 4). Statistical analysis was performed using ANOVA followed by Bonferroni post-test.
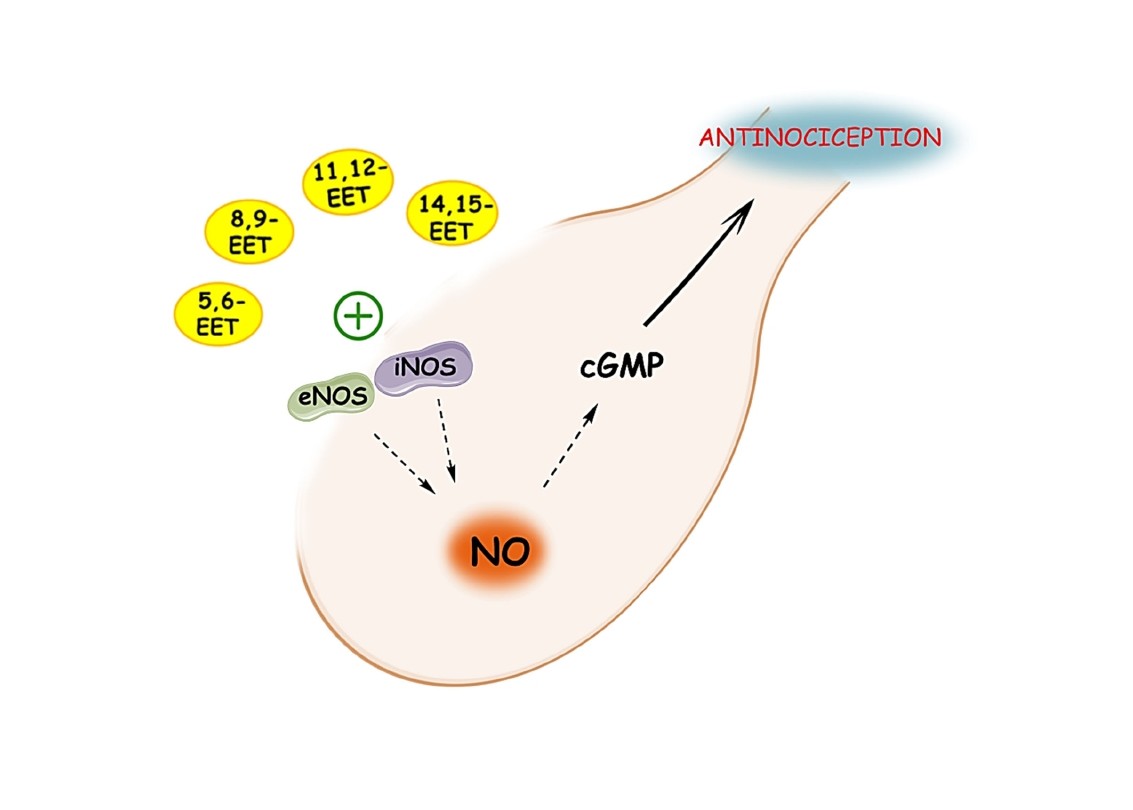
Results: The peripheral antinociceptive effects of 5,6-, 8,9-, 11,12-, and 14,15-EET were antagonized by L-NOArg (12, 18, and 24 µg/paw), a non-selective nitric oxide synthase (NOS) inhibitor. The selective inhibitors of inducible (iNOS) (LNIL; 24 μg/paw) and endothelial (eNOS) (LNIO; 24 μg/paw) isoforms partially reduced the antinociceptive effects of EETs. In contrast, selective inhibition of neuronal NOS (nNOS) with LNPA (24 µg/paw) did not affect the peripheral antinociceptive effects of EETs. The soluble guanylate cyclase (sGC) inhibitor ODQ (50, 100, and 200 μg/paw) decreased the peripheral antinociceptive effects of EETs. Conversely, Zaprinast (50 μg/paw), a selective phosphodiesterase (PDE) inhibitor, enhanced the peripheral antinociceptive effects of low doses of EETs. The results suggest that EETs may be linked with iNOS and eNOS activation, but not nNOS, and this process leads to the production of nitric oxide, which stimulates sGC, raises cGMP, and ultimately produces the antinociceptive effect observed with these substances.
Keywords
Epoxyeicosatrienoic acids, Peripheral antinociception, Nitric oxide
Introduction
Epoxyeicosatrienoic acids (EETs) are metabolites of arachidonic acid produced by cytochrome P450 epoxygenases responsible for important signaling that mediates a series of events, such as inflammation [1], pain [2], cancer [3], angiogenesis [4], and cardiovascular regulation [5].
Like EETs, nitric oxide (NO) has an intrinsic mechanism for regulating vascular tone [6]. Studies have shown that EETs can be considered an endothelium-derived hyperpolarizing factor (EDHF), as they cause hyperpolarization and subsequent relaxation of vascular smooth muscle cells by opening high-conductance calcium-activated potassium channels [7–10], whereas NO is an endothelium-derived relaxing factor (EDRF) that induces vasodilation through its pathway [11].
EETs and NO also have controversial roles in the nociceptive pathway and can be pro-nociceptive [2,12] or antinociceptive [13,14], depending on their concentration and route of administration. However, NO can also act as a mediator in the antinociceptive signaling of other antinociceptive molecules [15–18].
The description of the nitric oxide pathway in pain relief mechanisms was first outlined in the 1970s, with early findings showing that blocking mediators of this pathway prevents the pain-relieving effects of specific molecules, such as acetylcholine [19]. Later, Duarte et al. (1990) [13] demonstrated that nitroprusside, a non-enzymatic producer of NO, produced antinociception in a paw compression test in rats. They also found that L-arginine, a precursor of NO synthesis, induced antinociception, and that L-NMMA, an inhibitor of NO biosynthesis, blocked it. Ultimately, the researchers demonstrated that the pain-relieving effects of sodium nitroprusside and ACh were enhanced by the intraplantar injection of a cyclic GMP phosphodiesterase inhibitor (MY 5445). Therefore, it was only in the early 1990s that the link between this mediator and the NO pathway was established, providing evidence of the role of the NO pathway in peripheral antinociception.
Our group has described the influence of nitrergic pathway modulation as part of the endogenous control of inflammatory pain in the periphery [15] and as a key peripheral antinociceptive mechanism of drugs such as peptides from animal toxins [20], hydrogen peroxide [18], tramadol [16], and even cannabidiol [17].
Recently, our group described the antinociceptive potential of the four regioisomers of EETs in an acute pain model induced by PGE2. Now, in this study, our objective was to determine whether the nitrergic pathway mediates the analgesic effects of EETs.
Methods
Animals
Male Swiss mice weighing between 30 and 40 g, with free access to food and water, were kept on a 12-hour light/dark cycle (6:00-18:00 h) at a controlled temperature of 24 ± 2°C at the Bioterism Center of the Federal University of Minas Gerais (CEBIO-ICB/UFMG). After the tests, the animals were euthanized via high-dose intraperitoneal injection of an anesthetic mixture (300 mg/kg ketamine hydrochloride and 15 mg/kg xylazine hydrochloride, both from Sigma-Aldrich, USA). The Ethics Committee on Animal Experimentation approved the project under protocol number 75/2017.
Drugs
Prostaglandin E2 (PGE2; Sigma-Aldrich, USA) was diluted in 2% ethanol within an aqueous solution of sterile 0.9% sodium chloride (saline). 5,6-epoxieicosatrienoic acid (5,6-EET) (Cayman Chemical, USA) was dissolved in methyl acetate mixed with saline. 8,9-epoxieicosatrienoic acid (8,9-EET) (Cayman Chemical, USA) and 11,12-epoxieicosatrienoic acid (11,12-EET) (Cayman Chemical, USA) were dissolved in 6.4% ethanol in saline. 14,15-epoxieicosatrienoic acid (14,15-EET) (Cayman Chemical, USA) was dissolved in 25.6% ethanol in saline. Nonselective NO synthase (NOS) inhibitor L-NOarg [NG-nitro-L-arginine (RBI, USA)]; selective neuronal NO synthase inhibitor L-NPA [NW-propyl-L-arginine (Sigma-Aldrich)]; selective endothelial NO synthase inhibitor L-NIO [N5-(1-iminoethyl)-L-ornithine dihydrochloride (Sigma-Aldrich)]; and selective inducible NO synthase inhibitor L-NIL [L-N6-(1-iminoethyl) lysine hydrochloride (Sigma-Aldrich)] were dissolved in isotonic saline. Specific soluble guanylyl cyclase and phosphodiesterase 5 and 6 inhibitors ODQ (1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one; RBI) and Zaprinast (Cayman Chemical, USA) were dissolved in 10% DMSO in saline. All drugs were injected via intraplantar at a volume of 20 μg/paw.
Measurement of nociceptive threshold
Hyperalgesia was induced with prostaglandin E2 (PGE2; 2 µg) through a right hind paw intraplantar injection. The mechanical nociceptive threshold was measured in grams (g) using a paw pressure test (Ugo-Basile; SRL, Varese, Italy), as described in rats [21] and mice [22]. The test uses a cone-shaped paw-presser with a rounded tip that gradually increases force on the hind paw. The force required to elicit paw withdrawal was recorded as the nociceptive threshold. A cutoff value of 150 g was set to reduce the risk of paw damage.
The nociceptive threshold was measured in the right paw and calculated as the average of three consecutive trials recorded before and 180 minutes after PGE2 injection. Hyperalgesia was determined by the difference between these two averages (Δ of nociceptive threshold).
Experimental protocol
The animals were first randomly split into several groups.
The basal threshold was measured just before the experiments started, with no drugs present. Then, PGE2 was administered at zero minutes. Nociceptive threshold measurements were taken after 180 minutes, which is when hyperalgesia peaks, to evaluate the nitrergic pathway. EETs were given 5 minutes before measurement, representing the peak of the antinociceptive effect previously shown by our group [14]. The non-selective inhibitor of nitric oxide synthase isoforms, L-NAME, along with the selective isoform inhibitors L-NIL, L-NIO, and L-NPA, were injected at 135 minutes. The specific soluble guanylyl cyclase inhibitor ODQ was administered at 165 minutes. The specific phosphodiesterase 5 and 6 inhibitor Zaprinast was given at 115 minutes. The drug dosage and timing protocols in this study were based on previous research from our group and existing literature [15–18]. The timing of drug injections was coordinated with each inhibitor's peak activity to ensure that all inhibitors reached maximum effect when the nociceptive threshold was measured (180 minutes after the PGE2 injection in the hind paw).
The experiments were conducted in a double-blind manner, where the person who injected the solutions was not the same as the one who performed the nociceptive behavioral assessment.
Data and statistical analysis
All results are shown in the figures as the mean ± standard error of the mean (S.E.M.). Statistical analysis was performed using GraphPad Prism 8.0.2, followed by one-way ANOVA and the Bonferroni post hoc test for multiple comparisons. Only p-values less than 0.05 (p<0.05) were considered statistically significant.
Results
Participation of nitric oxide (NO) in EETs peripheral antinociceptive effect against PGE2-induced hyperalgesia
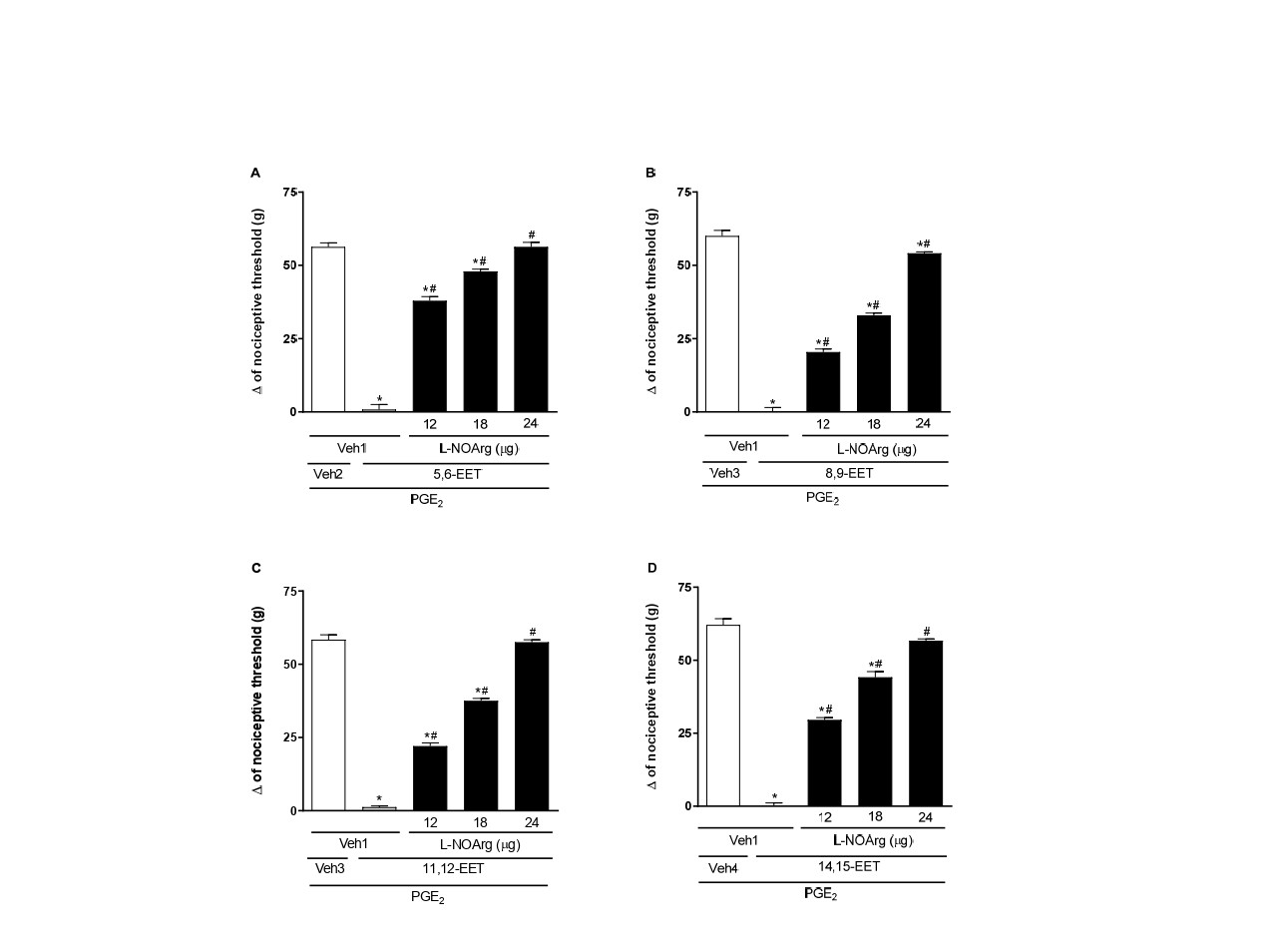
The peripheral antinociceptive effect of 5,6-, 8,9-, 11,12-, and 14,15-EET was blocked by L-NOArg (12, 18, and 24 µg/paw), a non-selective inhibitor of nitric oxide synthase (NOS) enzymes (Figure 1).
Figure 1. Administration of a NOS inhibitor prevents 5,6- (A), 8,9- (B), 11,12- (C), and 14,15-EET (D) -induced peripheral antinociception in hyperalgesic paws. Each column represents the mean ± SEM (n = 4). * and # indicate a significant difference (P<0.05) compared to PGE2 + Veh and PGE2 + EET, respectively. ANOVA with Bonferroni post test.
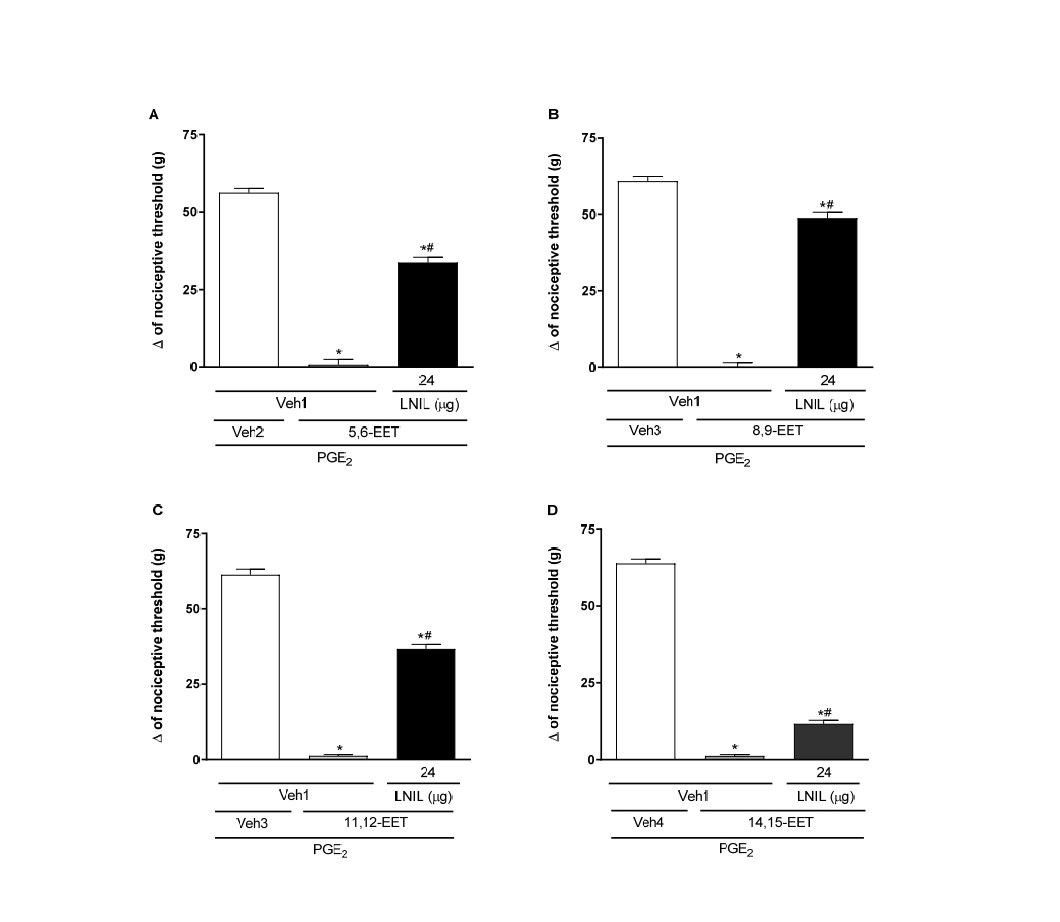
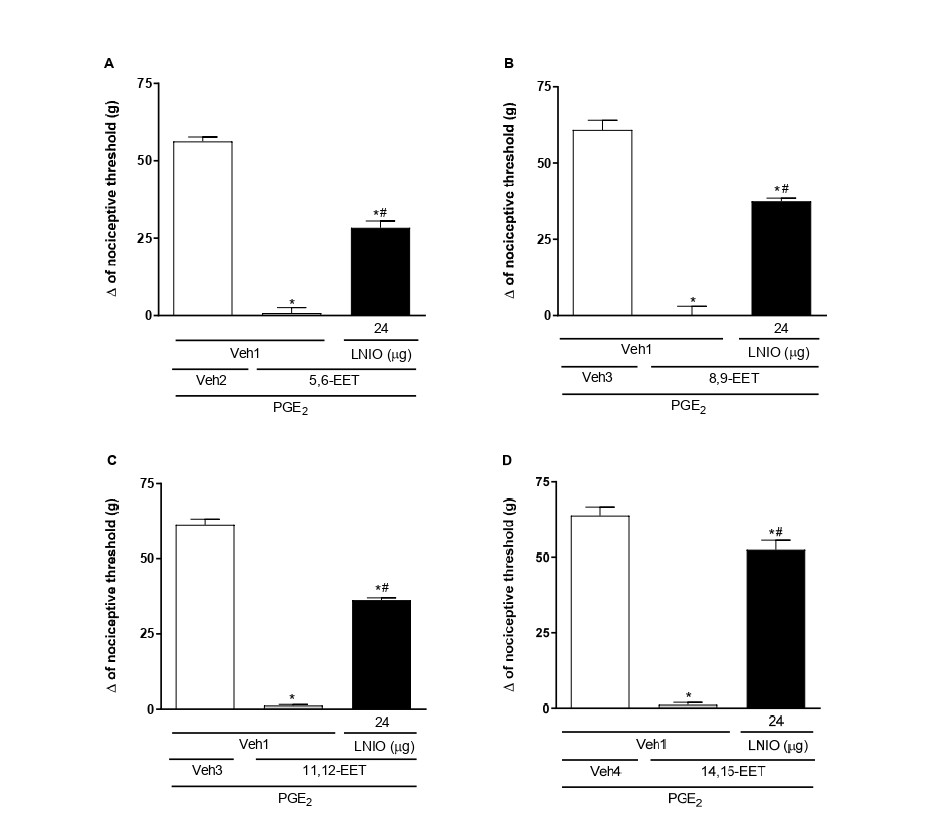
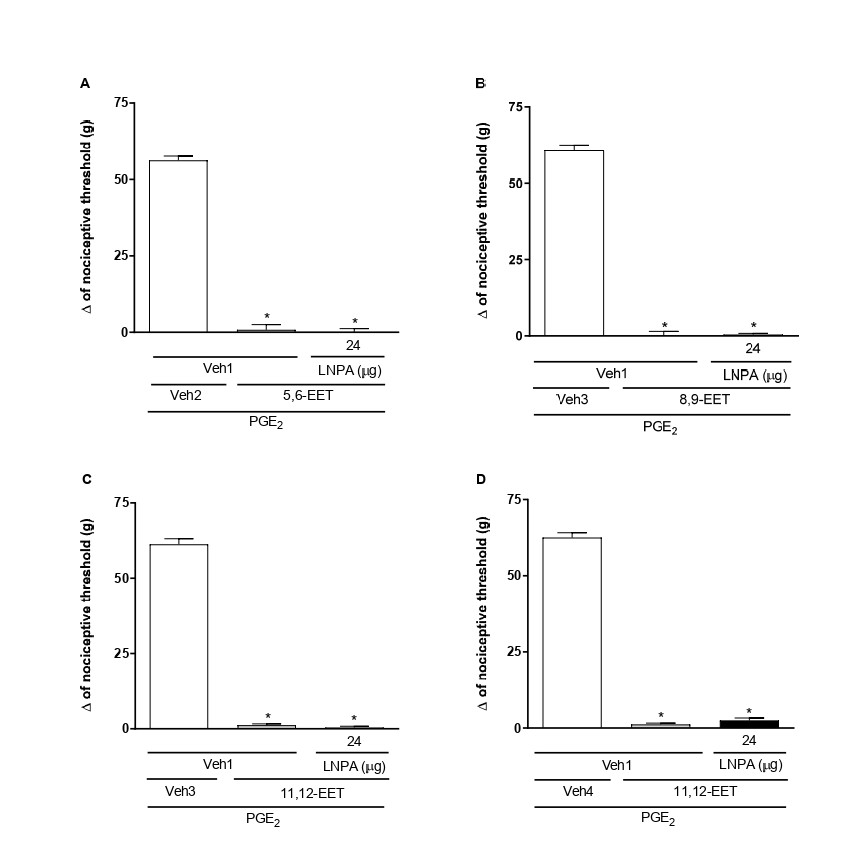
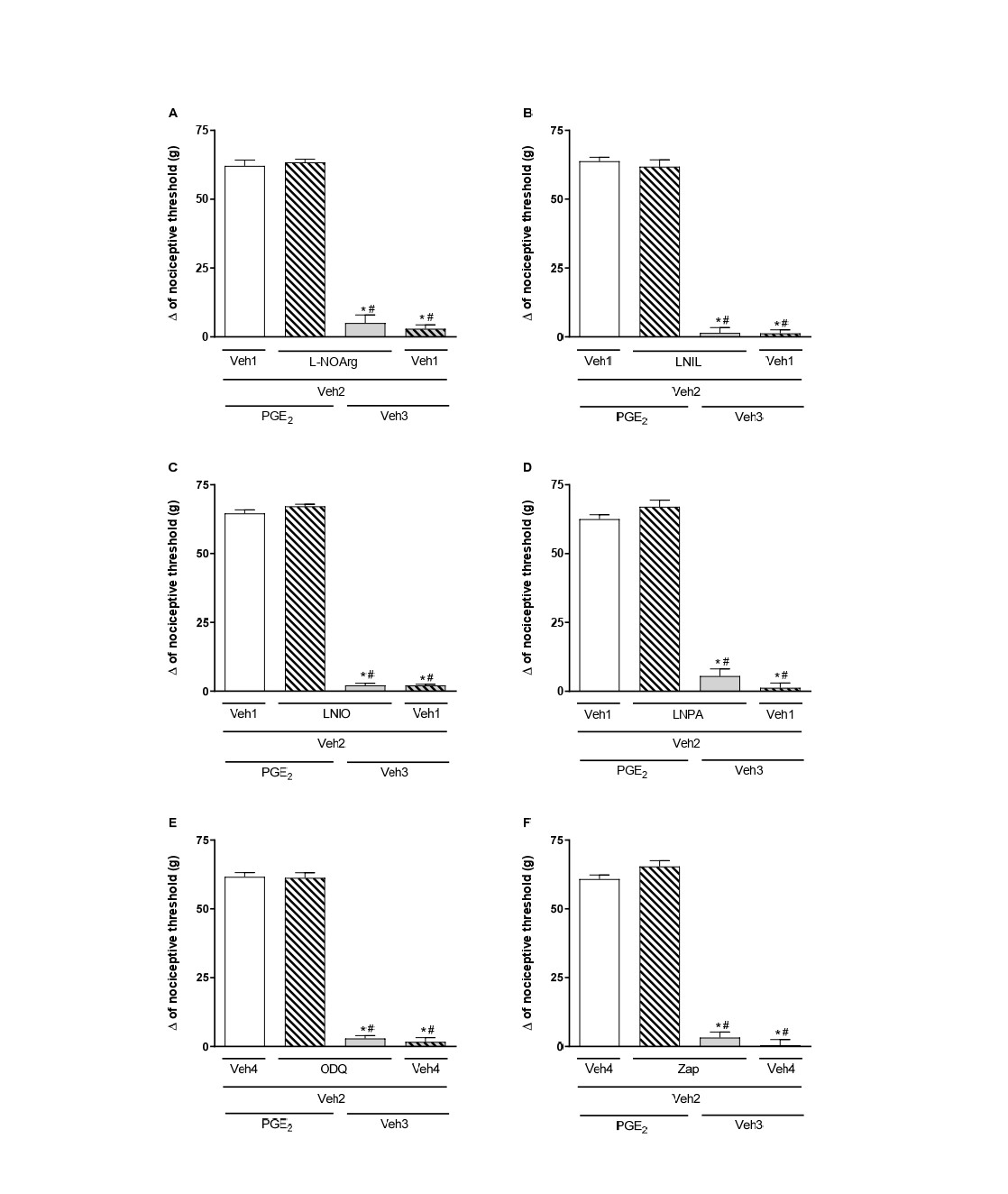
Selective inhibitors of inducible (iNOS; LNIL, 24 μg/paw) and endothelial (eNOS; LNIO, 24 μg/paw) isoforms of NOS partially reduced the antinociceptive effect of EETs. That is, there was a decrease in antinociception caused by EETs, but it did not return to the control threshold of hyperalgesia (Figures 2 and 3, respectively). Conversely, the selective inhibitor of neuronal NOS (nNOS; LNPA, 24 μg/paw) did not affect the peripheral antinociception induced by EETs (Figure 4). All these inhibitors, when administered alone, did not alter the response to PGE2 or the vehicle (Figure 7).
Figure 2. Administration of iNOS inhibitor partially blocks 5,6- (A), 8,9- (B), 11,12- (C), and 14,15-EET (D)-induced peripheral antinociception in hyperalgesic paws. Each column shows the mean ± SEM (n = 4). * and # indicate a significant difference (P<0.05) compared with PGE2 + Veh and PGE2 + EET, respectively. ANOVA with Bonferroni post hoc test.
Figure 3. Administration of eNOS inhibitor partially blocks 5,6- (A), 8,9- (B), 11,12- (C), and 14,15-EET (D)-induced peripheral antinociception in hyperalgesic paws. Each column shows the mean ± SEM (n = 4). * and # indicate significant differences (P<0.05) compared with PGE2 + Veh and PGE2 + EET, respectively. ANOVA with Bonferroni post test.
Figure 4. Administration of nNOS inhibitor did not block 5,6- (A), 8,9- (B), 11,12- (C), and 14,15-EET (D)-induced peripheral antinociception in hyperalgesic paws. Each column shows the mean ± SEM (n = 4). * and # indicate a significant difference (P<0.05) compared with PGE2 + Veh and PGE2 + EET, respectively. ANOVA with Bonferroni post hoc test.
Role of cyclic guanosine monophosphate (cGMP) in the peripheral antinociceptive effects of EETs
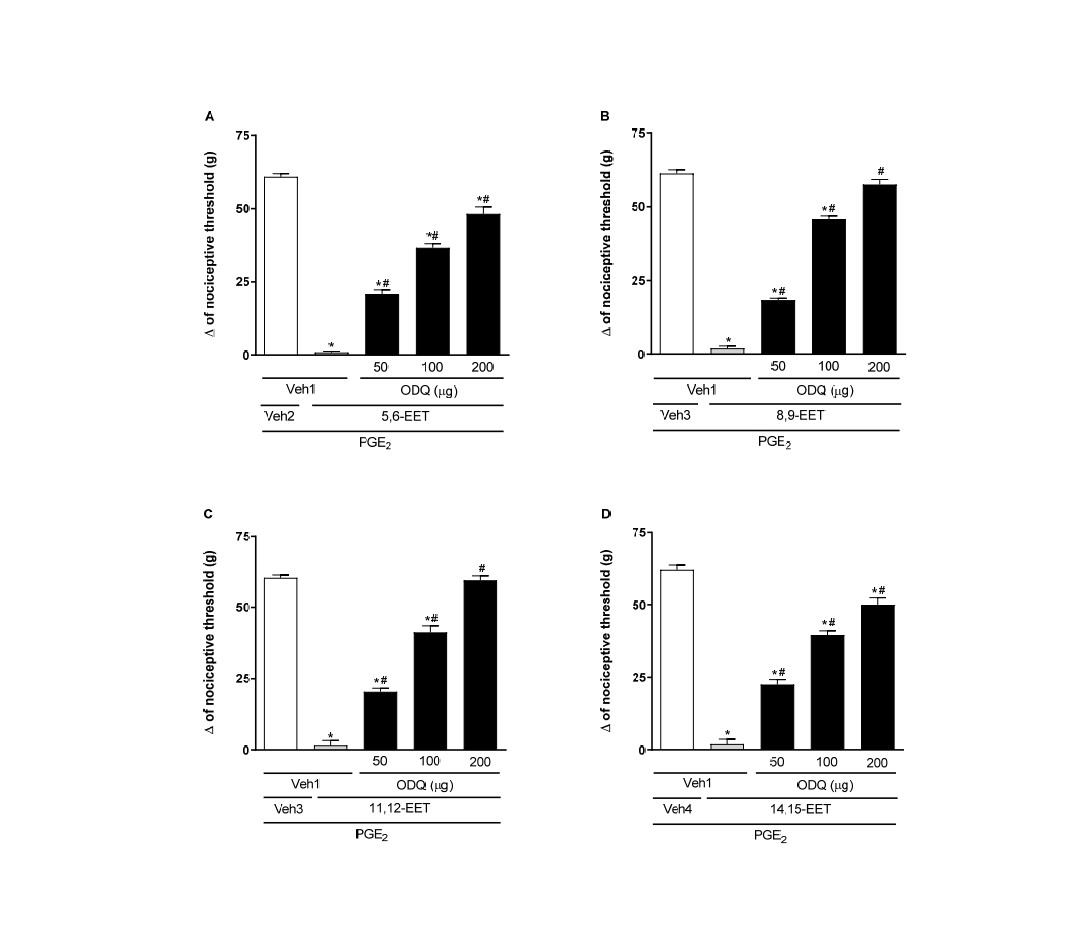
The soluble guanylate cyclase (sGC) inhibitor, ODQ (50, 100, and 200 μg/paw), reversed the peripheral antinociceptive effect of EETs in a dose-dependent manner (Figure 5).
Figure 5. Administration of an sGC inhibitor blocks 5,6- (A), 8,9- (B), 11,12- (C), and 14,15-EET (D)-induced peripheral antinociception in hyperalgesic paws. Each column shows the mean ± SEM (n = 4). * and # indicate a significant difference (P<0.05) compared to PGE2 + Veh and PGE2 + EET, respectively. ANOVA with Bonferroni post test.
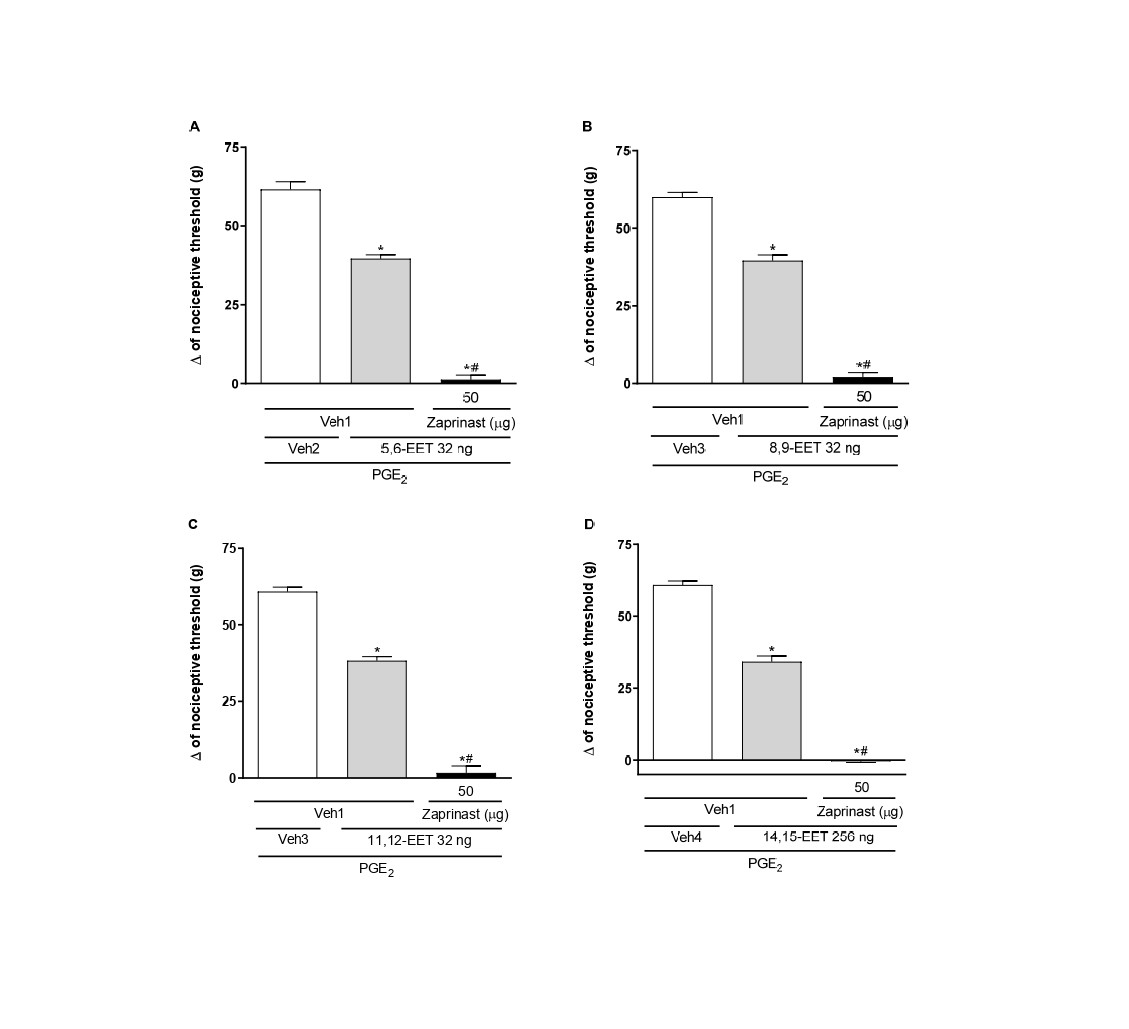
Additionally, a selective phosphodiesterase (PDE) inhibitor, Zaprinast (50 μg/paw), increased the peripheral antinociceptive effect of low doses of EETs, producing an effect similar to that of the maximum doses of these substances (Figure 6).ODQ and zaprinast, at the doses used, showed no effect when injected separately into either normal or hyperalgesic paws (Figure 7).
Figure 6. Potentiation by a PDE inhibitor of a lower dose of 5,6- (A), 8,9- (B), 11,12- (C), and 14,15-EET (D)-induced peripheral antinociception in hyperalgesic paws. Each column shows the mean ± SEM (n = 4). * and # indicate a significant difference (P<0.05) compared to PGE2 + Veh and PGE2 + EET, respectively. ANOVA with Bonferroni post hoc test.
Figure 7. Effect of L-NOArg (A), LNIL (B), LNIO (C), LNPA (D), ODQ (E), and Zaprinast (Zap; F) alone and on PGE2-induced hyperalgesia. Each column shows the mean ± SEM (n = 4). * and # indicate a significant difference (P<0.05) compared to PGE2 + Veh and PGE2 + (LNOArg, LNIL, LNIO, LNPA, ODQ, or Zap) + Veh, respectively. ANOVA with Bonferroni post test.
Discussion
In Fonseca and Duarte (2024) [14], our group described the antinociceptive effects of the four regioisomeric forms of EETs and their receptor involvement in these effects in an acute pain model induced by PGE2. We used the paw pressure test to measure the mechanical nociceptive threshold, as outlined in rats by Randall and Selitto (1957) [21] and in mice by Kawabata et al. (1992) [22]. The maximum analgesic effects were observed at doses of 128 ng for 5,6-, 8,9-, and 11,12-EETs, and at 512 ng for 14,15-EET. The doses producing sub-maximal analgesic effects were 32 ng for 5,6-, 8,9-, and 11,12-EETs, and 256 ng for 14,15-EET. Therefore, both dose ranges were used in this study to examine the role of the nitric oxide pathway in the antinociceptive mechanism of EETs.
Besides their antinociceptive effects in the periphery, the review paper by Turnbull and Chapman (2024) [23] discusses how EETs also exert antinociceptive effects through mechanisms and pain control pathways in the central nervous system. 14,15-EET has been shown to reduce pain behavior after intrathalamic administration in a preclinical model of post-stroke pain. In this model, 14,15-EET increased allopregnanolone production, thereby enhancing γ-aminobutyric acid A receptor (GABA-AR) signaling [24]. The administration of 14,15-EET, but not 5,6-EET, 8,9-EET, or 11,12-EET, into the ventrolateral PAG also proved effective in inhibiting acute nociceptive responses via opioidergic mechanisms [25]. In addition to the literature describing how EETs can induce antinociception through central mechanisms, it is also important to explore other ways these molecules can cause peripheral antinociception.
Therefore, to determine whether the NO pathway is involved in the peripheral antinociception of EETs, we used a non-selective NOS inhibitor (L-NOArg). It was observed that blocking these enzymes reduced the peripheral antinociceptive effect of EETs, indicating that NOS enzymes play a role in this process. It is known that NOS has three different isoforms: neuronal (nNOS), endothelial (eNOS), and inducible (iNOS) [26]. To identify which NOS isoform is related to EET-induced antinociception, we used selective inhibitors for each isoform. The selective inhibitors of iNOS (L-NIL) and eNOS (L-NIO) partially reduced the antinociceptive effect of EETs. In contrast, the selective nNOS inhibitor (L-NPA) did not affect the peripheral antinociception caused by EETs. Thus, it is suggested that EETs are somehow associated with iNOS and eNOS, but not nNOS, leading to NO production.
Consistent with our findings, studies show that EETs can activate eNOS, leading to NO release and, consequently, vasodilation [27,28]. Tan et al. [29] concluded in their study that the vasodilation mechanism of 5,6-EET in the rabbit pulmonary circulation is mainly EDRF-NO dependent, as the use of LNNA (Nω-nitro-L-arginine, 10−4 M), an NO synthase inhibitor, blocked the vasodilation caused by EETs. Hercule et al. [27] reported that removing the endothelium or administering L-NAME, an NO synthase inhibitor, blocked 8,9-EET-dependent dilation and altered vascular function in mice. They showed that 8,9-EET could induce NO production in primary mouse aortic endothelial cells. These effects were also blocked by L-NAME, indicating that EETs can influence the availability and/or action of NO to produce vasodilation.
In antinociceptive signaling, NO promotes sGC activation, thereby increasing the conversion of GTP to cGMP and elevating intracellular cGMP levels [30]. This second messenger is also linked to antinociceptive effects in the periphery [13,31,32]. Therefore, we examine the role of the sGC enzyme in the peripheral antinociceptive effects of EETs using its inhibitor, ODQ. Inhibiting sGC reduces the antinociceptive effect of EETs, indicating that cGMP is involved in this process.
After the GTP-cGMP conversion, cGMP is quickly broken down by phosphodiesterases [33]. Therefore, inhibiting these enzymes would prolong the action of this second messenger. We used the selective cGMP-specific phosphodiesterase E (PDE) inhibitor, Zaprinast, to investigate cGMP's role in the peripheral antinociceptive effects of EETs. It was observed that PDE inhibition increased the antinociceptive effects of low doses of EETs, indicating that cGMP levels are key in this signaling.
Zhang et al. [34] showed that in vitro incubation of human platelets with 11,12-EET increases NOS activity, nitrite production, and cGMP levels in a concentration-dependent manner, indicating that some EETs can activate the NO pathway in different cells through various mechanisms. The findings of Hung et al. [35] reveal that 11,12-EET can also strongly promote neovasculogenesis, not only by increasing NO production but also through the endothelial nitric oxide synthase (eNOS) signaling pathway.
Our data, consistent with the literature, offers insight into the functions of EETs in nociception and how these substances interact with nitric oxide production and signaling, which seem to occur during their antinociceptive effects.
Conclusion
These results collectively indicate that 5,6-, 8,9-, 11,12-, and 14,15-EET, when administered peripherally, could be associated with iNOS and eNOS activation, but not nNOS. This process results in the production of nitric oxide, which stimulates sGC, increases cGMP, and ultimately produces the antinociceptive effect observed with these substances.
Competing Interests
The authors declare that they have no conflicts of interest regarding this publication.
Acknowledgments
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; 303876/2023-9) and Fundação de Amparo à Pesquisa de Minas Gerais (FAPEMIG).
Author Contributions Statement
FCSF: Data curation, Investigation, Methodology, Writing – original draft and Writing – review & editing. WCPB: Data curation, Methodology, Writing – original draft. DPDM: Data curation, Investigation and Methodology. DLA: Data curation, Investigation and Methodology. DDA: Data curation, Investigation and Methodology. BFGQ: Data curation, Investigation and Methodology. A.C.P.: Resources. T.R.L.R.: Resources. I.D.G.D.: Conceptualization, Data curation, Formal analysis, Funding acquisition, Project administration, Resources, Supervision, Validation, Visualization and Writing – review & editing.
References
2. Brenneis C, Sisignano M, Coste O, Altenrath K, Fischer MJ, Angioni C, et al. Soluble epoxide hydrolase limits mechanical hyperalgesia during inflammation. Mol Pain. 2011 Oct 4;7:78.
3. Guo Z, Johnson V, Barrera J, Porras M, Hinojosa D, Hernández I, et al. Targeting cytochrome P450-dependent cancer cell mitochondria: cancer associated CYPs and where to find them. Cancer Metastasis Rev. 2018 Sep;37(2-3):409–23.
4. Rand AA, Rajamani A, Kodani SD, Harris TR, Schlatt L, Barnych B, et al. Epoxyeicosatrienoic acid (EET)-stimulated angiogenesis is mediated by epoxy hydroxyeicosatrienoic acids (EHETs) formed from COX-2. J Lipid Res. 2019 Dec;60(12):1996–2005.
5. Zhang G, Kodani S, Hammock BD. Stabilized epoxygenated fatty acids regulate inflammation, pain, angiogenesis and cancer. Prog Lipid Res. 2014 Jan;53:108–23.
6. Félétou M, Vanhoutte PM. EDHF: an update. Clin Sci (Lond). 2009 Jul 16;117(4):139–55.
7. Baron A, Frieden M, Bény JL. Epoxyeicosatrienoic acids activate a high-conductance, Ca(2+)-dependent K + channel on pig coronary artery endothelial cells. J Physiol. 1997 Nov 1;504 ( Pt 3)(Pt 3):537–43.
8. Campbell WB, Gebremedhin D, Pratt PF, Harder DR. Identification of epoxyeicosatrienoic acids as endothelium-derived hyperpolarizing factors. Circ Res. 1996 Mar;78(3):415–23.
9. Fisslthaler B, Popp R, Kiss L, Potente M, Harder DR, Fleming I, et al. Cytochrome P450 2C is an EDHF synthase in coronary arteries. Nature. 1999 Sep 30;401(6752):493–7.
10. Quilley J, McGiff JC. Is EDHF an epoxyeicosatrienoic acid? Trends Pharmacol Sci. 2000 Apr;21(4):121–4.
11. Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980 Nov 27;288(5789):373–6.
12. Sousa AM, Prado WA. The dual effect of a nitric oxide donor in nociception. Brain Res. 2001 Apr 6;897(1-2):9–19.
13. Durate ID, Lorenzetti BB, Ferreira SH. Peripheral analgesia and activation of the nitric oxide-cyclic GMP pathway. Eur J Pharmacol. 1990 Sep 21;186(2-3):289–93.
14. Fonseca FC, Duarte ID. 5, 6-, 8, 9-, 11, 12-and 14, 15-Epoxyeicosatrienoic Acids (EETs) Induce Peripheral Receptor-Dependent Antinociception in PGE2-Induced Hyperalgesia in Mice. Pharmacology and Clinical Pharmacy Research. 2024 Dec 16;9(2):156–69.
15. Alves DP, da Motta PG, Romero TR, Klein A, Duarte ID. NO/cGMP production is important for the endogenous peripheral control of hyperalgesia during inflammation. Nitric Oxide. 2013 Jan 15;28:8–13.
16. Soares-Santos RR, Machado DP, Romero TL, Duarte IDG. Nitric oxide and potassium channels but not opioid and cannabinoid receptors mediate tramadol-induced peripheral antinociception in rat model of paw pressure withdrawal. Can J Physiol Pharmacol. 2024 Mar 1;102(3):218–27.
17. de Almeida DL, Mendes Ferreira RC, Fonseca FC, Dias Machado DP, Aguiar DD, Guimaraes FS, et al. Cannabidiol induces systemic analgesia through activation of the PI3Kγ/nNOS/NO/KATP signaling pathway in neuropathic mice. A KATP channel S-nitrosylation-dependent mechanism. Nitric Oxide. 2024 May 1;146:1–9.
18. Barra W, Queiroz B, Perez A, Romero T, Ferreira R, Duarte I. Study on peripheral antinociception induced by hydrogen peroxide (H2O2): characterization and mechanisms. Naunyn Schmiedebergs Arch Pharmacol. 2024 Oct;397(10):7927–38.
19. Ferreira SH, Nakamura M. I - Prostaglandin hyperalgesia, a cAMP/Ca2+ dependent process. Prostaglandins. 1979 Aug;18(2):179–90.
20. Freitas AC, Silva GC, Pacheco DF, Pimenta AM, Lemos VS, Duarte ID, et al. The synthetic peptide PnPP-19 induces peripheral antinociception via activation of NO/cGMP/KATP pathway: Role of eNOS and nNOS. Nitric Oxide. 2017 Apr 1;64:31–38.
21. RANDALL LO, SELITTO JJ. A method for measurement of analgesic activity on inflamed tissue. Arch Int Pharmacodyn Ther. 1957 Sep 1;111(4):409–19.
22. Kawabata A, Nishimura Y, Takagi H. L-leucyl-L-arginine, naltrindole and D-arginine block antinociception elicited by L-arginine in mice with carrageenin-induced hyperalgesia. Br J Pharmacol. 1992 Dec;107(4):1096–101.
23. Turnbull J, Chapman V. Targeting the soluble epoxide hydrolase pathway as a novel therapeutic approach for the treatment of pain. Curr Opin Pharmacol. 2024 Oct;78:102477.
24. Chen X, Li Z, Zhang B, Hu R, Li J, Feng M, et al. Alleviation of Mechanical Allodynia by 14,15-Epoxyeicosatrienoic Acid in a Central Poststroke Pain Model: Possible Role of Allopregnanolone and δ-Subunit-Containing Gamma-Aminobutyric Acid A Receptors. J Pain. 2019 May;20(5):577–91.
25. Terashvili M, Tseng LF, Wu HE, Narayanan J, Hart LM, Falck JR, et al. Antinociception produced by 14,15-epoxyeicosatrienoic acid is mediated by the activation of beta-endorphin and met-enkephalin in the rat ventrolateral periaqueductal gray. J Pharmacol Exp Ther. 2008 Aug;326(2):614–22.
26. Nathan C. Nitric oxide as a secretory product of mammalian cells. FASEB J. 1992 Sep;6(12):3051–64.
27. Hercule HC, Schunck WH, Gross V, Seringer J, Leung FP, Weldon SM, et al. Interaction between P450 eicosanoids and nitric oxide in the control of arterial tone in mice. Arterioscler Thromb Vasc Biol. 2009 Jan;29(1):54–60.
28. Wang H, Lin L, Jiang J, Wang Y, Lu ZY, Bradbury JA, et al. Up-regulation of endothelial nitric-oxide synthase by endothelium-derived hyperpolarizing factor involves mitogen-activated protein kinase and protein kinase C signaling pathways. J Pharmacol Exp Ther. 2003 Nov;307(2):753–64.
29. Tan JZ, Kaley G, Gurtner GH. Nitric oxide and prostaglandins mediate vasodilation to 5,6-EET in rabbit lung. Adv Exp Med Biol. 1997;407:561–6.
30. Moncada S, Palmer RM, Higgs EA. The discovery of nitric oxide as the endogenous nitrovasodilator. Hypertension. 1988 Oct;12(4):365–72.
31. Pacheco DF, Reis GM, Francischi JN, Castro MS, Perez AC, Duarte ID. delta-Opioid receptor agonist SNC80 elicits peripheral antinociception via delta(1) and delta(2) receptors and activation of the l-arginine/nitric oxide/cyclic GMP pathway. Life Sci. 2005 Nov 19;78(1):54–60.
32. Romero TR, Duarte ID. alpha(2)-Adrenoceptor agonist xylazine induces peripheral antinociceptive effect by activation of the L-arginine/nitric oxide/cyclic GMP pathway in rat. Eur J Pharmacol. 2009 Jun 24;613(1–3):64–7.
33. Beavo JA. Cyclic nucleotide phosphodiesterases: functional implications of multiple isoforms. Physiol Rev. 1995 Oct;75(4):725–48.
34. Zhang L, Cui Y, Geng B, Zeng X, Tang C. 11,12-Epoxyeicosatrienoic acid activates the L-arginine/nitric oxide pathway in human platelets. Mol Cell Biochem. 2008 Jan;308(1-2):51–6.
35. Hung HC, Syu JN, Chao CY, Huang SM, Lin CC, Yang MD, et al. Neovasculogenic effect of 11,12-epoxyeicosatrienoic acid involves the Akt/eNOS signaling pathways in human endothelial progenitor cells. Biomedicine (Taipei). 2022 Sep 1;12(3):20–30.