Abstract
Objective: Anastomotic insufficiency in colorectal surgery can lead to serious complications and high morbidity and mortality rates. This study aimed to evaluate the effect of tirofiban hydrochloride, an antiplatelet agent, on ischemic colon anastomosis healing.
Materials and Methods: A total of 21 male Wistar albino rats were randomly divided into three groups: Group A (Sham), Group B (Ischemia-Control), and Group C (Ischemia-Tirofiban). Ischemia was induced in the colon by ligating the marginal arteries, followed by end-to-end anastomosis. Group C received intraperitoneal tirofiban (0.125 mcg/1cc) for five postoperative days. On the fifth postoperative day, bursting pressure, hydroxyproline (HP) levels, and histopathological ischemic necrosis scores were assessed.
Results: Bursting pressures were measured as 97.85±16.17 mmHg in Group B and 170.85±8.15 mmHg in Group C (p<0.0001). HP levels were 2.42±0.84 mg/g in Group A, 3.70±0.29 mg/g in Group B, and 4.80±0.99 mg/g in Group C. Histopathological scores were significantly better in Group C (p=0.024).
Conclusion: Tirofiban significantly improved ischemic colon anastomosis healing in terms of bursting pressure, collagen production, and histological tissue integrity. These findings suggest that tirofiban may be a potential agent to enhance anastomotic safety in ischemic surgical settings.
Keywords
Colon anastomosis, Ischemia, Tirofiban, Bursting pressure, Hydroxyproline
Introduction
Colonic anastomoses are among the most crucial surgical procedures in gastrointestinal surgery [1]. However, anastomotic leakage following these procedures may lead to serious postoperative complications. Insufficient blood supply is one of the primary factors impairing healing. Tirofiban has the potential to improve microcirculation by inhibiting platelet aggregation. In this study, we investigated the effects of tirofiban on colon anastomosis healing under ischemic conditions. Experimental and clinical studies have demonstrated that ischemia disrupts the balance between collagen synthesis and degradation, leading to impaired tensile strength and delayed anastomotic maturation [2,4,5]. In addition, ischemia promotes microvascular thrombosis and endothelial dysfunction, further compromising local tissue perfusion during the critical early phases of wound healing [6].
Tirofiban hydrochloride is a reversible non-peptide antagonist of the platelet glycoprotein IIb/IIIa (GPIIb/IIIa) receptor and is widely used in the treatment of acute coronary syndromes to prevent platelet aggregation [7,8].
By inhibiting platelet aggregation, tirofiban has the potential to improve microcirculatory flow, reduce ischemia-induced thrombosis, and preserve endothelial integrity [9].
Although various pharmacological agents have been investigated to enhance colonic anastomotic healing under ischemic conditions, evidence regarding the role of glycoprotein IIb/IIIa inhibitors in this setting remains extremely limited [10,11]. To the best of our knowledge, no previous experimental study has specifically evaluated the effect of tirofiban on ischemic colon anastomosis healing.
Therefore, the present study aimed to investigate the effects of tirofiban hydrochloride on ischemic colon anastomosis healing in a rat model by evaluating mechanical strength, collagen content, and histopathological changes.
Materials and Methods
Animals and experimental design
A total of 21 male Wistar-Albino rats, weighing between 200 and 205 grams, were included in the study. All rats were kept under standard laboratory conditions and were fed with commercial rat chow and water. The rats were fast for eight hours before and after the operation. Wistar-Albino rats were randomly allocated into three equal groups (n=7 per group) using a simple randomization method generated by sealed opaque envelopes prior to surgery.
Randomization was performed by an investigator not involved in the surgical procedures to minimize selection bias. All surgical interventions and outcome assessments were conducted by investigators blinded to group allocation.
All procedures in this experimental study were performed in accordance with the National Guidelines for the Use and Care of Laboratory Animals and were approved by the Animal Ethics Committee of Mustafa Kemal University.
- Group A (Sham): Only laparotomy and colon mobilization were performed.
- Group B (Control): Colonic ischemia and anastomosis were performed, but no treatment was applied.
- Group C (Tirofiban): Colonic ischemia and anastomosis were performed, followed by tirofiban administration.
Ischemia model and surgical technique
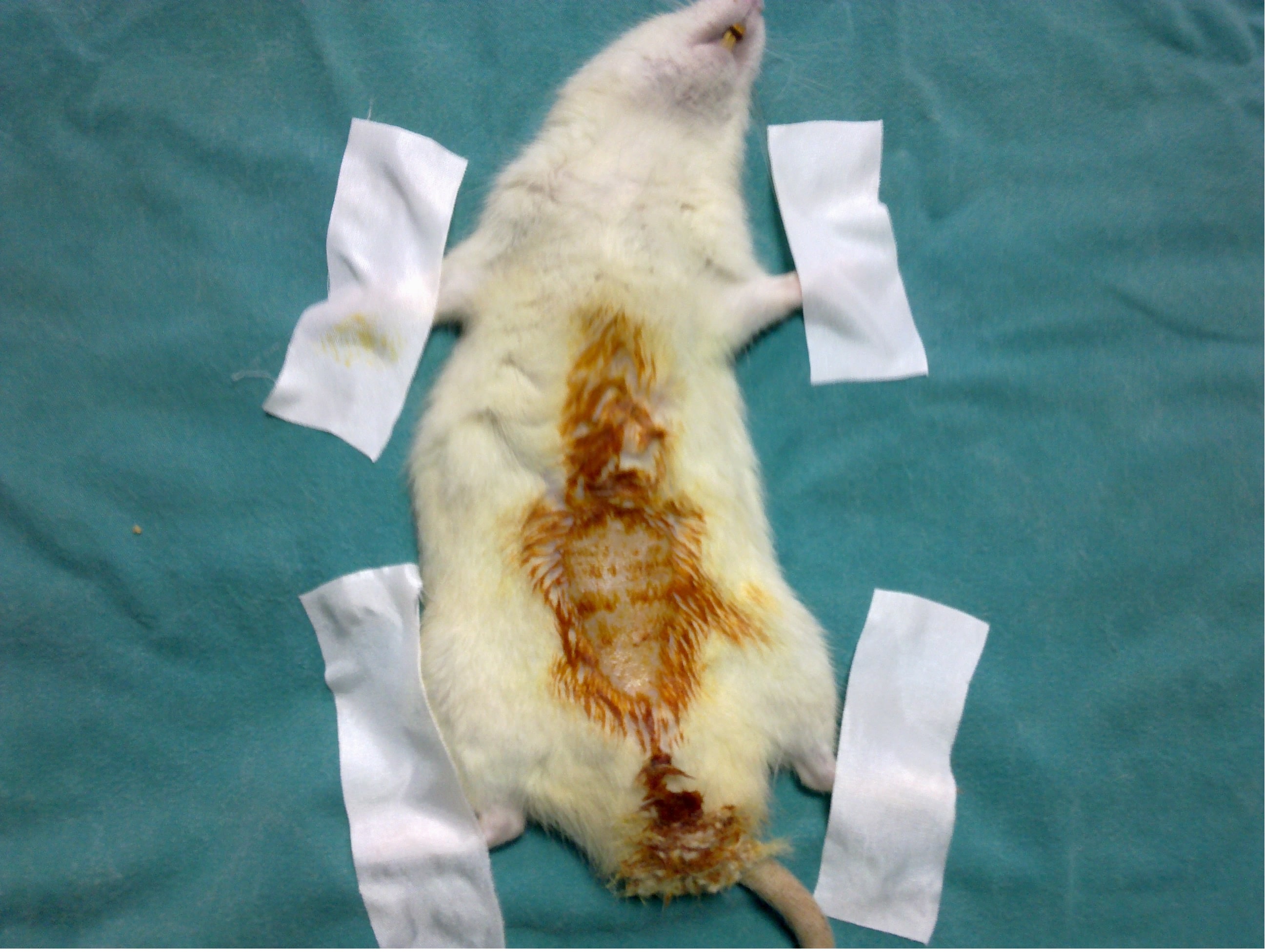
In the study conducted at the Experimental Animal Research Center Laboratory of Mustafa Kemal University Faculty of Medicine, anesthesia was induced using 2 mg/kg of xylazine (Rompun 2%, 0.1 mL, BAYER) and 25 mg/kg of ketamine (Ketalar 50 mg/mL, 0.5 mL, PFIZER). Following aseptic preparation, a midline laparotomy was performed.
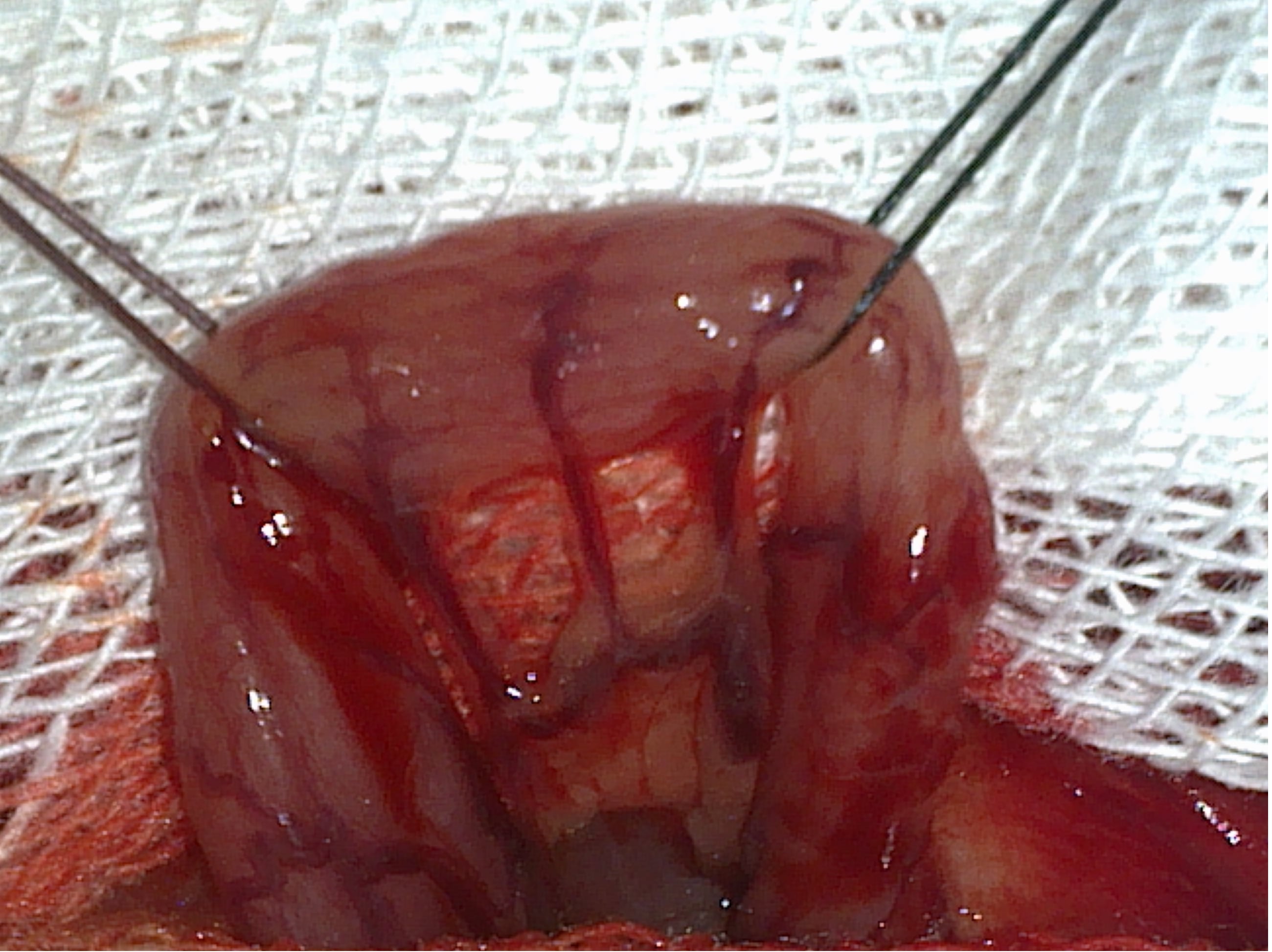
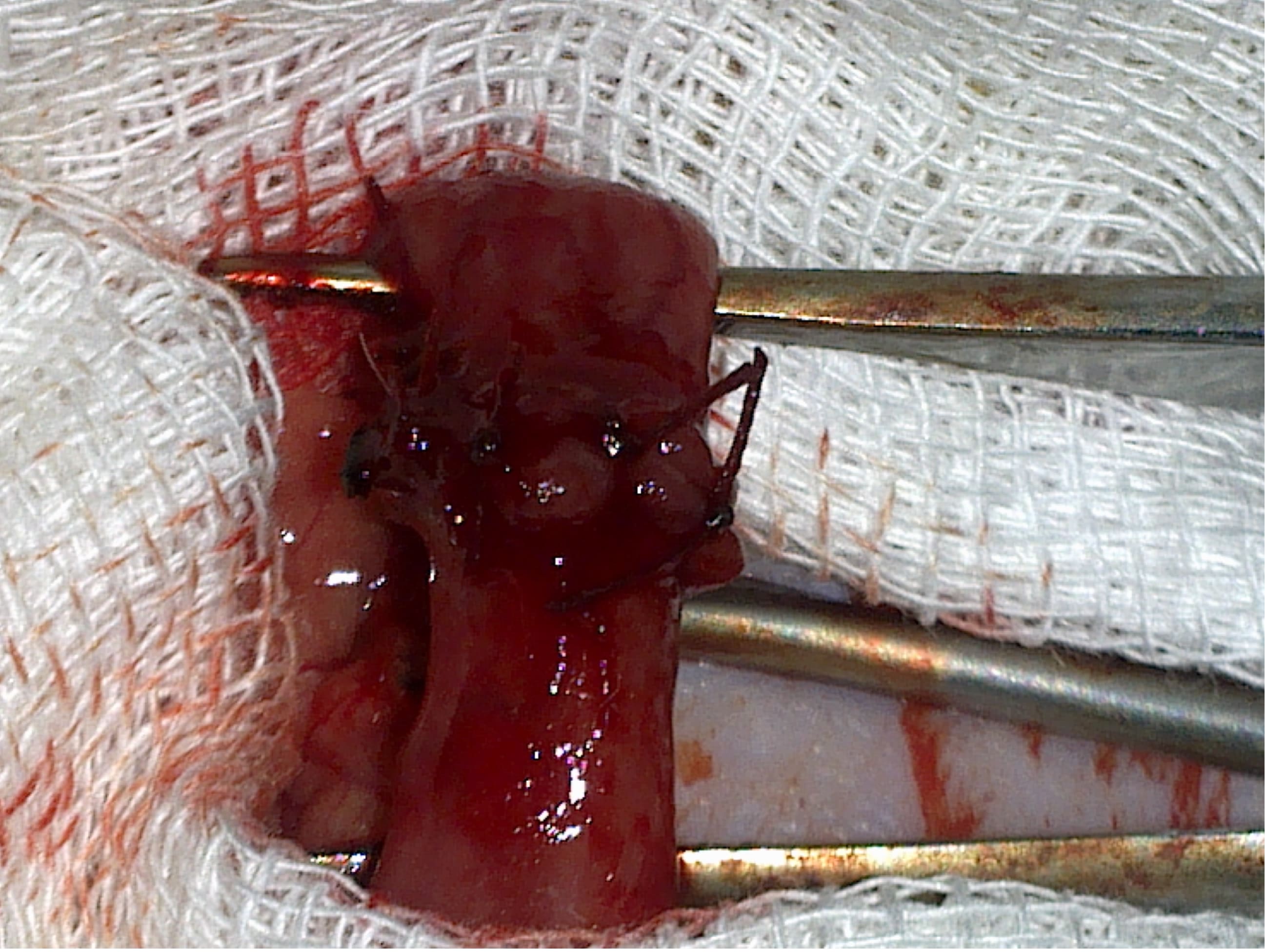
In Groups B and C, the marginal arteries 2 cm proximally and distally from the selected anastomosis site were ligated with 4/0 silk sutures and then cut to induce ischemia. After waiting for 10 minutes, the colon was transected full-thickness, and end-to-end anastomosis was performed in a single layer using 4/0 polyglactin sutures models [5,6] (Vicryl, Ethicon LTD).
After anastomosis, Group B received 1 cc of intraperitoneal saline (0.9% NaCl), whereas Group C received 0.125 mcg of tirofiban (AGGRASTAT, Merck-Co Inc.) in 1 cc intraperitoneally.
The selected dose of 0.125 mcg/1 cc and intraperitoneal route were based on previous experimental studies demonstrating effective platelet inhibition and local vascular modulation without causing systemic bleeding in small-animal. The intraperitoneal route was preferred to achieve localized action around the anastomotic site and minimize systemic exposure.
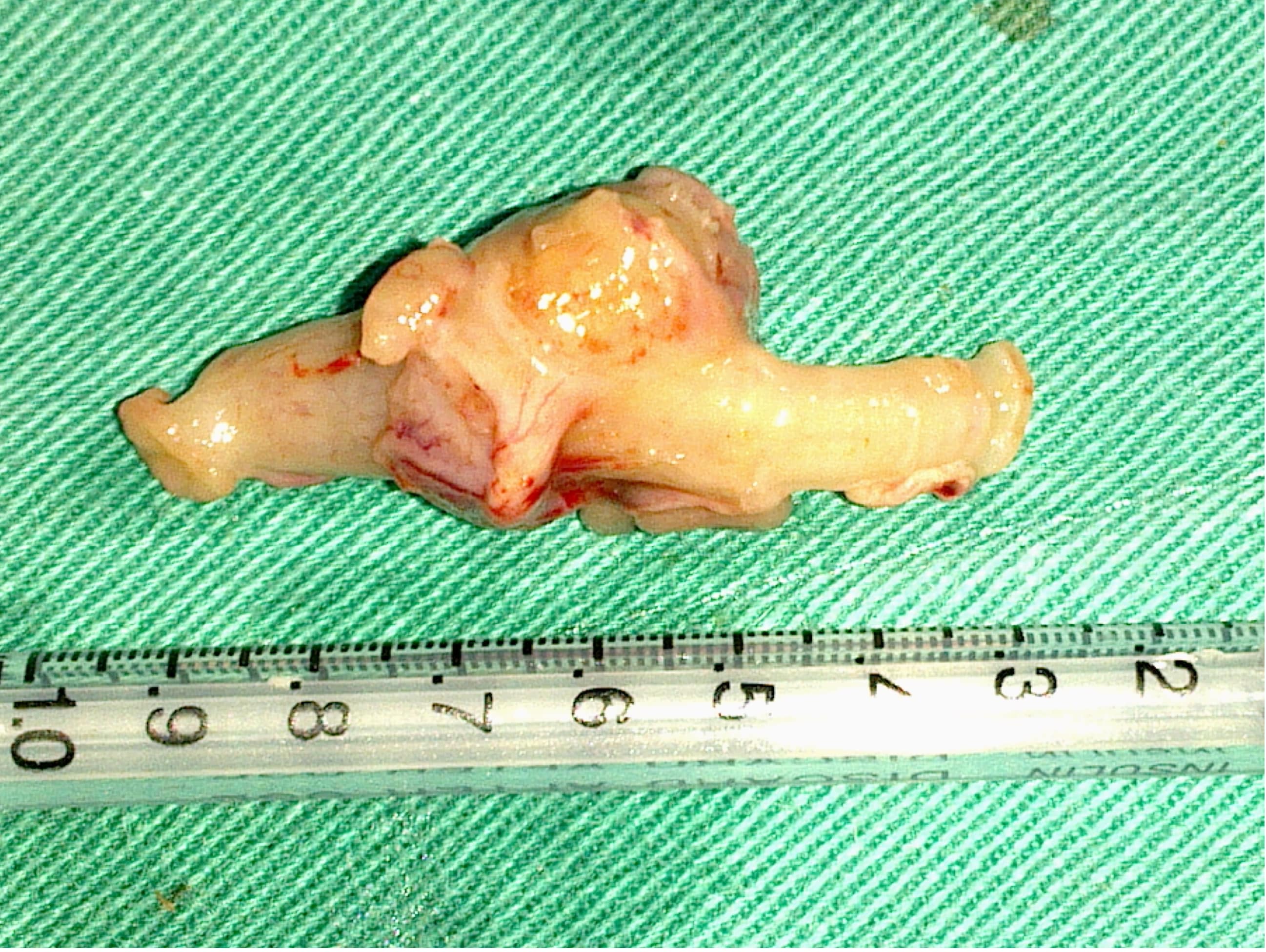
The abdominal layers were closed with 4/0 monofilament polypropylene sutures. All procedures were performed by the same surgeon. Postoperatively, only water was allowed for 24 hours, then oral feeding was resumed. For five days, the control group received saline, and the tirofiban group received 0.125 mcg tirofiban daily. On day 5, the anastomotic segments were resected for bursting pressure, biochemical, and histopathological analyses.
Figure 1. Preoperative image of rat.
Figure 2. Determination of the anastomosis line.
Figure 3. The completed anastomosis line.
Figure 4. Removed anastomosis line.
Assessment criteria
Bursting pressure
Measured with a manometric system; pressure was recorded when methylene blue appeared outside the anastomosis line.
Hydroxyproline measurement
Conducted spectrophotometrically using the Woessner method.
Histopathology
Evaluated per Garcia’s modified scoring system; necrosis grading determined under light microscopy.
Statistical analysis
All statistical analyses were performed using SPSS v23.0. Data were expressed as mean ± standard deviation. Non-parametric tests (Kruskal–Wallis, Mann–Whitney U) were used for non-normally distributed variables, whereas one-way ANOVA was applied for normally distributed data. A p<0.05 was considered significant.
Results
Bursting pressure
Bursting pressures were recorded from the anastomosis lines in all rats. The bursting pressure values for each rat and the intergroup data are presented in Table 1. The bursting pressures were significantly higher in Group C (170.85±8.15 mmHg, p<0.0001).
|
Rat |
Group A (Sham) |
Group B (Control) |
Group C (Treatment) |
|
1 |
220 |
101 |
159 |
|
2 |
211 |
122 |
182 |
|
3 |
226 |
103 |
171 |
|
4 |
232 |
108 |
168 |
|
5 |
240 |
71 |
177 |
|
6 |
206 |
89 |
163 |
|
7 |
228 |
91 |
176 |
The mean bursting pressure (± standard deviation) was 223.28±11.87 mmHg in Group A, 97.85±16.17 mmHg in Group B, and 170.85 ± 8.15 mmHg in Group C. The difference in bursting pressures between Groups B and C was statistically significant (p<0.0001).
The mean bursting pressure (± standard deviation) was 223.28±11.87 mmHg in Group A, 97.85±16.17 mmHg in Group B, and 170.85±8.15 mmHg in Group C. The difference in bursting pressures between Groups B and C was statistically significant (p<0.0001).
Hydroxyproline levels
The hydroxyproline levels, measured via biochemical analysis and expressed as mg/g of dry tissue, are shown in Table 2. Group C exhibited significantly higher hydroxyproline levels (4.80±0.99 mg/g) compared to the other groups (p<0.05).
|
Rat |
Group A (Sham) |
Group B (Control) |
Group C (Treatment) |
|
1 |
2.586 |
4.115 |
3.103 |
|
2 |
1.149 |
3.593 |
3.829 |
|
3 |
1.724 |
3.789 |
5.722 |
|
4 |
2.605 |
3.621 |
4.658 |
|
5 |
3.736 |
3.598 |
5.393 |
|
6 |
3.017 |
4.003 |
5.493 |
|
7 |
2.155 |
3.231 |
5.446 |
The mean hydroxyproline levels (± standard deviation) were 2.42±0.848 mg/g in Group A, 3.70±0.294 mg/g in Group B, and 4.80±0.995 mg/g in Group C. Pairwise comparisons between the groups revealed statistically significant differences:
- Group A vs. Group B: p = 0.009
- Group A vs. Group C: p = 0.003
- Group B vs. Group C: p = 0.042
Overall, the difference between Groups B and C was considered statistically significant (p<0.05).
Histopathological analysis
Histopathological evaluation was performed using Garcia’s modified ischemic necrosis scoring system, which grades tissue injury on a scale from Grade 1 to Grade 4 based on epithelial integrity, inflammatory cell infiltration, hemorrhage, and depth of necrosis.
- Grade 1: Normal histological architecture
- Grade 2: Mild epithelial degeneration and focal inflammation
- Grade 3: Moderate necrosis with transmural inflammatory infiltration
- Grade 4: Severe transmural necrosis with loss of tissue integrity
All histological assessments were conducted by a blinded pathologist under light microscopy.
The histological evaluation of ischemic necrosis among the groups revealed the following scores:
- Group A: HS = 28
- Group B: HS = 18
- Group C: HS = 25
The comparison between Groups B and C showed a statistically significant difference (p=0.024), indicating more severe ischemic necrosis in Group B than in Group C. No significant difference was observed between Groups A and C (p=0.441). One-way ANOVA also showed significant differences among the three groups (p=0.002). The histological score (HS) was calculated as the sum of individual grades for each specimen.
Group B exhibited the highest ischemic necrosis scores, while Group C significantly reduced these scores (p = 0.024).
|
Ischemic Necrosis Grade |
Group A (n, %) |
Group B (n, %) |
Group C (n, %) |
|
Grade 1 |
7 (100%) |
1 (14.3%) |
4 (57.1%) |
|
Grade 2 |
0 (0%) |
3 (42.8%) |
3 (42.8%) |
|
Grade 3 |
0 (0%) |
2 (28.6%) |
0 (0%) |
|
Grade 4 |
0 (0%) |
1 (14.3%) |
0 (0%) |
|
Histological Score (HS) |
28 |
18 |
25 |
Discussion
Perianastomotic blood flow is a key determinant of anastomotic healing, and impaired perfusion has consistently been identified as one of the principal factors leading to anastomotic failure. Adequate microcirculation is essential for oxygen delivery, fibroblast migration, collagen synthesis, and neovascularization during the early phases of wound repair [14,16]. In ischemic conditions, disruption of these processes results in reduced anastomotic strength and increased susceptibility to leakage.
Tirofiban hydrochloride is a non-peptide tyrosine derivative that functions as a reversible antagonist of the platelet glycoprotein IIb/IIIa (GPIIb/IIIa) receptor, a critical mediator in the final common pathway of platelet aggregation. By blocking the binding of fibrinogen, von Willebrand factor, and other adhesive ligands to the GPIIb/IIIa receptor, tirofiban effectively inhibits platelet aggregation and thrombus formation [7,8,13]. Although platelet–fibrin interactions are essential for physiological hemostasis, excessive platelet activation under ischemic and inflammatory conditions may promote microvascular thrombosis and further impair tissue perfusion [6,12].
Experimental ischemic colon anastomosis models commonly involve ligation of marginal arteries followed by colonic transection and single-layer anastomosis. The technique described by Karatas et al. has become a widely accepted and standardized model for evaluating ischemic anastomotic healing in rats [3,11]. We adopted this well-established approach to ensure reproducibility and comparability with existing experimental data.
In the present study, tirofiban administration resulted in a significant increase in anastomotic bursting pressure and collagen synthesis under ischemic conditions. These findings suggest that platelet inhibition may improve anastomotic integrity not only by enhancing microcirculatory flow but also by modulating downstream inflammatory pathways. Previous experimental studies have demonstrated that inhibition of platelet activation attenuates neutrophil recruitment, reduces cytokine release, and limits endothelial dysfunction in ischemic tissues [18,19]. Such effects may collectively contribute to improved wound healing and tissue preservation at the anastomotic site.
Hydroxyproline levels, an established surrogate marker for collagen content, were highest in the tirofiban-treated group, indicating enhanced fibroblast activity and extracellular matrix deposition. The lower hydroxyproline levels observed in the sham group compared with the anastomotic groups are expected, as uninjured colonic tissue lacks the active fibroblast-driven collagen synthesis characteristic of wound repair [14]. Interestingly, hydroxyproline levels were also higher in the ischemic control group than in the sham group. This finding likely reflects a compensatory wound-healing response triggered by ischemic injury, characterized by increased fibroblast recruitment and collagen turnover despite impaired tissue perfusion [15,17].
Importantly, increased collagen synthesis alone does not necessarily translate into improved mechanical strength. Experimental studies have shown that ischemia-induced collagen deposition may be disorganized and structurally insufficient, resulting in reduced tensile strength despite elevated hydroxyproline levels [16,17]. In contrast, tirofiban treatment in our study appeared to enhance not only the quantity but also the functional organization of collagen, as evidenced by the markedly higher bursting pressure values observed in Group C.
Histopathological analysis further supported these findings by demonstrating reduced ischemic necrosis and better preservation of tissue architecture in the tirofiban-treated group. Microscopic studies of ischemic intestinal anastomoses have emphasized that preservation of microvascular patency is crucial for preventing transmural necrosis and maintaining anastomotic viability [17,20]. The improved histological scores observed in our study are consistent with these observations.
From a translational perspective, the potential use of tirofiban as an adjunctive agent to enhance anastomotic healing is particularly relevant for high-risk surgical settings characterized by compromised perfusion. Previous experimental studies have reported beneficial effects of pharmacological agents such as sildenafil citrate, dexpanthenol, and coenzyme Q10 on ischemic colonic anastomosis healing, primarily through vasodilation, antioxidant activity, and improved microcirculation [10,11,19,20]. Our findings extend this body of evidence by suggesting that targeted platelet inhibition represents an alternative and complementary strategy to improve ischemic anastomotic healing.
Although no macroscopic bleeding was observed in the tirofiban-treated group, glycoprotein IIb/IIIa inhibitors are known to be associated with bleeding complications in clinical practice. The absence of bleeding in our study may be attributed to the low-dose regimen and the intraperitoneal route of administration, which likely limited systemic exposure. Nevertheless, careful dose optimization and route selection will be essential for any potential clinical application.
This study has several limitations. The relatively small sample size may limit statistical power, and the assessment was restricted to short-term outcomes on postoperative day 5, precluding conclusions regarding long-term anastomotic maturation. Furthermore, only a single antiplatelet agent was evaluated, and the optimal dose and duration of tirofiban therapy remain uncertain. Future studies should investigate dose–response relationships, alternative routes of administration, and comparisons with other antiplatelet or microcirculatory agents. Translational studies incorporating human-equivalent dosing may further clarify the clinical applicability of tirofiban in colorectal surgery.
Despite these limitations, our results demonstrate that tirofiban significantly improves ischemic colon anastomosis healing based on mechanical, biochemical, and histopathological parameters. These findings suggest that tirofiban or similar platelet-targeted agents may serve as promising adjunctive therapies in ischemic surgical conditions. However, larger experimental studies and subsequent clinical investigations are required to validate this hypothesis and determine its relevance to routine surgical practice.
Conclusion
Tirofiban increased bursting pressure, promoted collagen production, and reduced ischemic necrosis in ischemic colon anastomoses. These findings suggest that tirofiban may serve as a supportive agent for anastomotic healing under ischemic surgical conditions. Further clinical and dose-optimization studies are warranted.
References
2. Jiborn H, Ahonen J, Zederfeldt B. Healing of experimental colonic anastomoses. II. Breaking strength of the colon after left colon resection and anastomosis. Am J Surg. 1978 Nov;136(5):595–9.
3. Karataş A, et al. The effect of amelogenin on colon anastomosis healing. Turkish Journal of Surgery. 2020.
4. Woessner JF Jr. The determination of hydroxyproline in tissue and protein samples containing small proportions of this amino acid. Arch Biochem Biophys. 1961 May;93:440–7.
5. Shogan BD, Belogortseva N, Luong PM, Zaborin A, Lax S, Bethel C, et al. Collagen degradation and MMP9 activation by Enterococcus faecalis contribute to intestinal anastomotic leak. Sci Transl Med. 2015 May 6;7(286):286ra68.
6. Rosendorf J, Klicova M, Herrmann I, Anthis A, Cervenkova L, Palek R, et al. Intestinal Anastomotic Healing: What do We Know About Processes Behind Anastomotic Complications. Front Surg. 2022 Jun 7;9:904810.
7. Shah HD, Goyal RK. Glycoprotein IIb/IIIa receptor and its inhibition: A platelet-directed therapeutic strategy. Indian J Pharmacol. 2004 May 1;36(3):133–9.
8. Kong DF, Califf RM, Miller DP, Moliterno DJ, White HD, Harrington RA, et al. Clinical outcomes of therapeutic agents that block the platelet glycoprotein IIb/IIIa integrin in ischemic heart disease. Circulation. 1998 Dec 22-29;98(25):2829–35.
9. York A, et al. Tirofiban and vascular patency in rat femoral artery. J Vasc Res. 2005.
10. Hasanoğlu A. Sildenafil Sitratın Kolon Anastomozundaki Yara İyileşmesi Üzerine Etkisi. Turkish Medical Journal. 2007;1(2):66–75.
11. Pehlivanlı F, Aydin O, Karaca G, Aydin G, Devrim T, Bulut H, et al. Healing of Ischemic Colon Anastomosis in Rats Could Be Provided by Administering Dexpanthenol or Coenzyme Q10. J Clin Med. 2018 Jun 25;7(7):161.
12. Rosendorf J, Klicova M, Cervenkova L, Horakova J, Klapstova A, Hosek P, et al. Reinforcement of Colonic Anastomosis with Improved Ultrafine Nanofibrous Patch: Experiment on Pig. Biomedicines. 2021 Jan 21;9(2):102.
13. Blanco-Colino R, Espin-Basany E. Intraoperative use of ICG fluorescence imaging to reduce the risk of anastomotic leakage in colorectal surgery: a systematic review and meta-analysis. Tech Coloproctol. 2018 Jan;22(1):15–23.
14. Thornton FJ, Barbul A. Healing in the gastrointestinal tract. Surg Clin North Am. 1997 Jun 1;77(3):549–73.
15. Attard JA, Raval MJ, Martin GR, Kolb J, Afrouzian M, Buie WD, et al. The effects of systemic hypoxia on colon anastomotic healing: an animal model. Dis Colon Rectum. 2005 Jul;48(7):1460–70.
16. Reijnen MM, Bleichrodt RP, van Goor H. Pathophysiology of anastomotic healing. Br J Surg. 1996;83(4):489–98.
17. Verhofstad MH, Lange WP, van der Laak JA, et al. Microscopic evaluation of ischemic intestinal anastomoses. J Surg Res. 2001;98(1):8–15.
18. Topaloğlu U, Aydın A, et al. The effect of antiplatelet agents on intestinal ischemia–reperfusion injury in rats. Eur Surg Res. 2010;45(3–4):160–6.
19. Cevik O, Oba S, et al. Platelet inhibition attenuates inflammation and oxidative injury in experimental intestinal ischemia. Inflammation. 2012;35(4):1219–28.
20. Wu Z, Li M, et al. Pharmacological strategies to improve intestinal anastomotic healing under ischemic conditions: Experimental evidence. World J Gastroenterol. 2019;25(28):3650–63.