Abstract
Background: There is limited data on the effectiveness and safety of vedolizumab for Indian patients, making this study the first focused evaluation of its use in this population.
Methods: This prospective, multicenter, open-label phase IV study enrolled patients aged 18–65 with moderate to severe ulcerative colitis (UC) or Crohn's disease (CD), who received vedolizumab 300 mg IV at weeks 0, 2, and 6 (induction) and at weeks 14, 22, 30, 38, and 46 (maintenance). The primary objective was safety; the secondary, effectiveness. Descriptive statistics were used to summarize demographic, baseline disease characteristics, prior/concomitant medications, and treatment outcomes.
Results: A total of 150 patients were enrolled (102 with UC; 48 with CD). Adverse Events (AE) affected 83 patients (55.3%), with mild AE in 72 (48%). Treatment related adverse events (TEAE) occurred in 52 (51%) patients with UC and 29 (60.4%) with CD, Serious adverse events (SAE) occurred in 8 (5.3%) patients. Additionally, 3.3% had at least 1 Adverse drug reaction (ADR), and 2.7% had adverse events of special interest (AESI). Reported ADR/AESI cases included one patient each with pulmonary tuberculosis, tuberculous pleurisy, rectal adenocarcinoma, and hypertension. Overall clinical response rates for weeks 14, 30, and 46 were 60.7%, 65.3%, and 72% and remission rates were 42%, 44%, and 53.3% respectively. Mucosal healing was observed in 18% patients at week 46 (23 (22.5%) with UC; 4 (8.3%) with CD). Quality of life scores showed improvements across both UC and CD groups, with increases noted from baseline of 5 at week 14 to 13 at week 46 in both groups.
Conclusion: This study demonstrates that vedolizumab has a good safety and effectiveness profile in Indian patients with moderate-to-severe UC or CD.
Keywords
Vedolizumab, Safety, Effectiveness, Inflammatory Bowel Disease, India, Ulcerative colitis, Crohn's disease
Abbreviations
ADR: Adverse Drug Reaction; AE: Adverse Events; AESI: Adverse Events of Special Interest; BEPs: Biologic-Experienced Patients; BNPs: Biologic-Naïve Patients; CRP: C-reactive Protein; CD: Crohn’s Disease; CS: Clinically significant; DCGI: Drugs Controller General of India; EAIRs: Exposure-adjusted Incidence Rates; ECGs: Electrocardiograms; EOS: End of the Study; ET: Early Terminations; GCP: Good Clinical Practice; GI: Gastrointestinal; HBI: Harvey-Bradshaw Index; IBD: Inflammatory Bowel Disease; IV: Intravenous ; JAK: Janus Kinase; MedDRA: Medical Dictionary for Regulatory Activities; PPAS: Per-Protocol Analysis Set; PTE: Pre-Treatment Event; QoL: Quality of Life; SAE: Serious Adverse Events; SAS: Safety Analysis Set; SD: Standard Deviations; SES-CD: Simple Endoscopic Score for Crohn’s Disease; SIBDQ: Short Inflammatory Bowel Disease Questionnaire; SOC: System Organ Class; TB: Tuberculosis; TEAE: Treatment Related Adverse Events; TNF-α: Tumor Necrosis Factor-alpha; UC: Ulcerative Colitis; UTI: Urinary Tract Infection
Introduction
Inflammatory bowel disease (IBD), comprising ulcerative colitis (UC) and Crohn’s disease (CD), represents a growing global health concern due to its chronic, relapsing, and remitting nature. UC is characterized by inflammation confined to the colonic mucosa and submucosa, while CD can involve any part of the gastrointestinal (GI) tract in a transmural pattern. Both conditions significantly impact patients' quality of life and impose substantial healthcare burdens. Over the last three decades, the prevalence of IBD has risen dramatically, with a 47% increase globally from 1990 to 2019, according to the Global Burden of Disease (GBD) study [1,2]. Traditionally seen as a disease of the Western world, IBD is now increasingly recognized in newly industrialized nations, including India, where rapid urbanization and lifestyle changes have contributed to its rising incidence [3].
India has emerged as a hotspot for IBD in Asia, with prevalence rates comparable to Western countries. Between 1990 and 2019, the estimated number of IBD cases in India increased from 0.13 million to 0.27 million [4]. Studies reported the prevalence of UC ranging between 43 and 44 per 100,000 individuals, while CD, although less common, demonstrated an increasing trend [5,6]. The rise in IBD cases in India has been partially linked to improvements in diagnostic technologies, greater public awareness, and better access to specialized healthcare [7]. The management of IBD in India remains challenging, as conventional therapies often fail to address the needs of patients with moderate-to-severe disease.
Current treatment options for IBD include corticosteroids, immunomodulators, and biologic therapies, with tumor necrosis factor-alpha (TNF-α) inhibitors forming the cornerstone of biologic treatment. While these therapies have shown efficacy, their limitations, such as suboptimal response rates, loss of efficacy over time, and increased risk of infections, highlight the need for alternative therapeutic options [8,9].
Recent advancements in IBD treatment have introduced novel therapeutic options, including anti-integrin biologics like vedolizumab, anti-IL12/23 biologics such as ustekinumab, and Janus Kinase (JAK) inhibitors like tofacitinib [10]. Vedolizumab, a gut-selective monoclonal antibody targeting the integrin α4β7, has shown promising results in reducing GI inflammation without causing systemic immunosuppression [11]. Global clinical trials and real-world studies have established vedolizumab’s efficacy in inducing and maintaining clinical remission in patients with moderate-to-severe UC or CD, particularly those unresponsive to conventional therapies or TNF-α antagonists [12,13].
Despite its established global efficacy, regional differences in disease epidemiology, environmental exposures, and healthcare systems necessitate localized evidence to confirm the applicability of vedolizumab in Indian patients. Recognizing this need, the Drugs Controller General of India (DCGI) mandated a Phase 4 study to evaluate the safety and effectiveness of vedolizumab in Indian patients as part of its marketing authorization requirements.
This post-hoc exploratory analysis presents the findings of the first prospective, multicenter, Phase 4 study assessing the safety and effectiveness of vedolizumab in Indian patients with moderate-to-severe UC or CD. By addressing the unique challenges faced by Indian IBD patients, this study aims to provide critical insights into the role of vedolizumab as a therapeutic option in this population.
Methodology
This open-label, single-arm, prospective, Phase 4 study (NCT04804540) was conducted across 17 sites in India. The primary aim was to evaluate the safety and effectiveness of vedolizumab 300 mg intravenous (IV) infusion in patients with moderate-to-severe UC or CD who demonstrated an inadequate response, loss of response, or intolerance to conventional therapy or TNF-α antagonists.
Study design
A total of 150 patients were enrolled, 102 with UC and 48 with CD. The study duration for each patient was up to 74 weeks, encompassing a 4-week screening period, a 46-week treatment period, and a 16-week safety follow-up period after the last dose of the study drug. Additionally, a long-term safety follow-up survey was conducted 6 months post-treatment via a telephonic visit.
Inclusion and exclusion criteria
Patients aged 18–65 years with a diagnosis of moderate-to-severe UC or CD for at least three months prior to screening were eligible for the study. UC patients required a Full Mayo Score of 6–12, while CD patients needed a Harvey-Bradshaw Index (HBI) score of ≥8. Endoscopic confirmation of disease activity within 30 days prior to or during screening was mandatory.
Key exclusion criteria included evidence of abdominal abscess, recent extensive bowel resections, ileostomy or colostomy, active or latent tuberculosis (TB), hepatitis B or C infections, and a history of malignancy or severe neurological disorders. Patients with a history of hypersensitivity to vedolizumab or related monoclonal antibodies, recent use of investigational drugs or live vaccines, and specific laboratory abnormalities (e.g., hemoglobin <8 g/dL, ALT or AST >3× ULN) were also excluded.
Patients who met the inclusion criteria received vedolizumab 300 mg IV infusions at specified intervals. Doses were administered at Weeks 0, 2, and 6 (induction phase) and subsequently at Weeks 14, 22, 30, 38, and 46 (maintenance phase). Patients with CD who did not show a clinical response were eligible for an additional dose at Week 10, and those experiencing a decrease in response during the maintenance phase were eligible to receive infusions every four weeks.
Study procedures
Screening procedures were conducted within 28 days prior to the first dose and included demographic and medical history collection, disease activity assessments, endoscopic evaluations, and laboratory testing. Baseline assessments included the Full Mayo Score for UC and the HBI for CD. Endoscopy was scored using the Mayo Score for UC and the Simple Endoscopic Score for Crohn’s Disease (SES-CD) for CD.
Safety assessments included physical examinations, vital signs, laboratory tests, and electrocardiograms (ECGs) conducted at regular intervals. Long-term safety data, including infections requiring hospitalization, UC or CD-related surgeries, and progressive multifocal leukoencephalopathy (PML), were collected during the follow-up telephonic survey.
Effectiveness and safety evaluations
The primary endpoint was the safety of vedolizumab, measured by the incidence of adverse events (AEs), with specific attention to treatment-emergent adverse events (TEAEs), serious adverse events (SAEs), adverse drug reactions (ADRs), and adverse events of special interest (AESIs). Safety events were summarized descriptively by severity, relationship to the study drug, and outcome. Exposure-adjusted incidence rates (EAIRs) were calculated using the Poisson method.
Secondary endpoints included the proportion of patients achieving clinical response, clinical remission, mucosal healing, or endoscopic response at defined time points. Disease activity scores and patient-reported quality of life (QoL) measures were analyzed for changes from baseline.
Statistical analysis
Safety analyses included all patients who received at least one dose of vedolizumab. Primary safety endpoints included analysis of AE, TEAE, ADR, AESI, and SAE, summarized by severity, relationship to the study drug, and system organ class using Medical Dictionary for Regulatory Activities (MedDRA) version 27.0 with EAIRs, and confidence intervals calculated using the Poisson method.
Secondary effectiveness endpoints analyzed binary outcomes with percentages and 95% confidence intervals by the exact Clopper-Pearson method, and continuous outcomes like QoL scores using descriptive statistics by using the number of non-missing observations (n), mean, standard deviations (SD), median, first quartile (Q1), third quartile (Q3), minimum and maximum value at each visit by UC and CD groups. Missing binary data were considered non-responders. Effectiveness analyses were performed for the safety analysis set (SAS) and the per-protocol analysis set (PPAS). Subgroup analyses were performed for biologic-naïve patients (BNPs) and biologic-experienced patients (BEPs).
Ethical considerations
The study was conducted in compliance with Good Clinical Practice (GCP) guidelines and adhered to local regulatory requirements. The study’s protocol was reviewed and approved by the Institutional Review Board (No. AIG/IEC-CT 50/07.2021-02). Informed consent was obtained from all participants or their legally authorized representatives prior to any study-related procedures.
Results
Demographic characteristics
The study included 150 patients, with a median age of 36 years (range: 18–63 years). Patients with UC had a median age of 39 years, while those with CD had a median age of 31 years. Male patients comprised 55.3% (83) and female patients comprised of 44.7% (67) of the cohort, and the median body mass index (BMI) was 20.41 kg/m². The median duration of IBD was 2.8 years, with a wide range from 0.3 to 34.3 years, reflecting both newly diagnosed and chronic cases. The majority of patients, 93.3% (140) had disease localized to the large intestine, with all UC patients and 79.2% (38) of CD patients affected, followed by 14.7% (22) in the ileocecal region (2% (2) in UC; 41.7% (20) in CD) and 10% (15) in the small intestine (3.9% (4) in UC; 22.9% (11)in CD). Extra-intestinal manifestations (oral cavity) were observed in 1.0% (1) of UC patients and 4.2% (2) of CD patients. IBD-related surgeries were reported in 1.0% (1) of UC patients and 14.6% (7) of CD patients. Disease severity scores revealed a mean (SD) Full Mayo Score of 8.64 (1.488) for UC patients and a mean HBI score of 10.90 (2.469) for CD patients. Prior biologics were used by 10.0% (15) of patients, with the remainder (90.0%; 135) being biologic-naïve (Table 1).
|
Characteristics |
UC (n=102) |
CD (n=48) |
Total (N=150) |
|
Age: years, median (min-max) |
39 (18–63) |
31 (18–59) |
36 (18–63) |
|
Gender n (%) Male |
|
|
|
|
Duration of IBD, years, mean ± SD |
4.12 (4.947) |
4.58 (4.309) |
4.26 (4.742) |
|
Full mayo score for UC, mean ±SD |
8.64 (1.488) |
N/A |
8.64 (1.488) |
|
HBI score for CD, mean ±SD |
N/A |
10.90 (2.469) |
10.90 (2.469) |
|
Treatment duration: days, median (min-max) |
325 (1–390) |
323 (15–345) |
324 (1–390) |
|
Percentages are based on number of patients in safety analysis set. CD: Crohn’s Disease; HBI: Harvey Bradshaw Index; IBD: Inflammatory Bowel Disease; NA: Not Applicable; UC: Ulcerative Colitis |
|||
Of the enrolled patients, 51.6% (111) patients (76 patients with UC and 35 patients with CD) completed the study and 18.1% (39) patients were terminated early. The common reasons for discontinuation were voluntary withdrawal 8.4% (18), pre-treatment event (PTE) or AE 4.2% (9), lost to follow-up 2.8% (6), lack of efficacy 2.3% (5) and 1 subject discontinued the study due to other reasons.
Approximately 31.3% (47) of patients had at least one medical condition at baseline, with infections (e.g., pulmonary tuberculosis) being the most common system organ class (SOC). Gastrointestinal disorders, including anal fistulas, were also reported. A history of surgical procedures was noted in 8.7% (13) of patients, with hemorrhoid surgeries and hysterectomies being the most common.
All patients received at least one prior medication within 30 days of screening, primarily for alimentary tract and metabolism conditions. Common prior medications included nervous system drugs (56.0%) and antineoplastic/immunomodulating agents (48.7%). Biologics such as adalimumab received 5.3% (8) and infliximab were used by 6.7% (10) of patients, with 3 receiving both. Concomitant medications were also widely used, with mesalazine (82.7%), folic acid (50.0%), and azathioprine (43.3%) being the most frequently administered.
Effectiveness
These results were analyzed using both the SAS and PPAS populations, with dropouts and missing data imputed as non-responders for binary endpoints. The number of patients assessed for effectiveness endpoints varied at Weeks 14, 30, and 46, primarily due to early terminations (ET).
At Week 14, 30, and 46, clinical response was observed in 60.7% (91), 65.3% (98), and 72.0% (108) of patients in the SAS population, respectively. In the UC and CD subgroups, the clinical response at these weeks was 54.9% (56), 62.7% (64), and 73.5% (75) for UC, and 72.9% (35), 70.8% (34), and 68.8% (33) for CD. For PPAS, 58.3% (81), 64.0% (89), and 70.5% (98) of patients had a clinical response at these timepoints, with UC patients showing 53.1% (51), 61.5% (59), and 71.9% (69), and CD patients showing 69.8% (30), 69.8% (30), and 67.4% (29) (Figure 1).
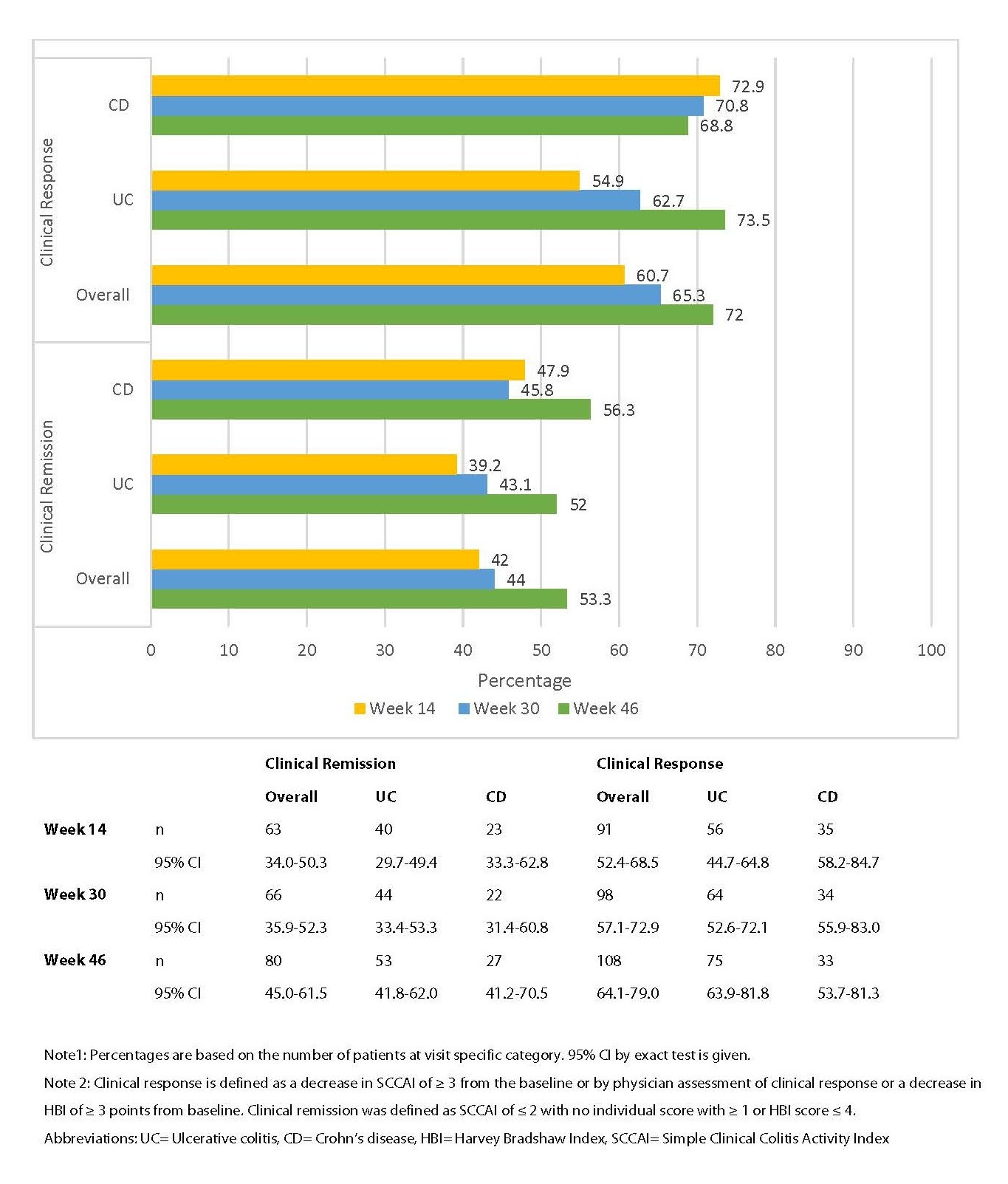
Figure 1. Clinical response and clinical remission in patients with UC and CD.
Regarding clinical remission, the SAS results showed a 42.0%, 44.0%, and 53.3% remission rate at Weeks 14, 30, and 46, respectively, with UC and CD subgroups demonstrating similar trends. In PPAS, clinical remission was seen in 38.8% (54), 41.0% (57), and 51.1% (71) of patients at these timepoints. In terms of vedolizumab discontinuation, 26.0% (39) of patients in SAS discontinued the drug, with UC and CD showing comparable rates. ET reasons for discontinuation were largely voluntary withdrawal and adverse events (Figure 1).
Mucosal healing was based on endoscopic evidence of no inflammation and healing of the mucosa as defined by a Mayo endoscopic sub-score of ≤1 point or SES-CD 0-2 or SES-CD ≤4 and at least a 2-point reduction from baseline with no sub-score >1. The endoscopic response was defined as a decrease in Mayo endoscopic sub-score of ≥1 point in UC and >50% decrease in SES-CD in CD. Mucosal healing/endoscopic response was assessed in a small number of patients, with overall mucosal healing observed in 18.0% (27) of which 23 (22.5%) patients belonged to UC group and 4 (8.3%) patients belonged to CD group and endoscopic response in 18.7% (28); 22 (21.6%) patients in UC group and 6 (12.5%) patients in CD group at Week 46. This response was more frequent in UC patients compared to CD patients.
The QoL of patients was evaluated using the Short Inflammatory Bowel Disease Questionnaire (SIBDQ) score after 14, 30, and 46 weeks of vedolizumab treatment. At Week 14, scores were recorded for 136 patients (UC: 91, CD: 45); at Week 30, for 123 patients (UC: 81, CD: 42); and at Week 46, for 118 patients (UC: 81, CD: 37). Among patients with UC, the median absolute change from baseline in SIBDQ scores was 5.00 (range: -20 to 42) at Week 14, 12.00 (range: -12 to 48) at Week 30, and 13.00 (range: -12 to 45) at Week 46. For CD patients, the median absolute change from baseline was 5.00 (range: -19 to 36) at Week 14, 6.50 (range: -26 to 36) at Week 30, and 13.00 (range: -32 to 44) at Week 46.
Safety
The median duration of vedolizumab treatment was 324 days, with a range from 1 to 390 days. The treatment duration was similar between UC (325 days) and CD (323 days). The median number of doses was 8, with a range of 1 to 16, showing similar treatment patterns in both groups.
Overall, 55.3% (83) of patients experienced at least one AE, with a slightly higher incidence in CD patients (60.4%) compared to UC patients (52.9%). A total of 5.3% (8) of patients had SAEs, with 2 of these being related to vedolizumab. The most common SAE was hospitalization or prolonged hospitalization. A total of 54.0% (81) of patients experienced TEAEs, and 3.3% had drug related TEAEs, all in UC patients. The severity of AEs was mostly mild (48%) or moderate (8%), with severe AEs reported in only 3 patients, primarily in UC patients. No patients died due to AEs, and most AEs were resolved by the end of the study (Table 2).
|
Safety Endpoints |
UC (n=102) |
CD (n=48) |
Total (N=150) |
|
Patients with ≥1 AE, n (%) |
54 (52.9) |
29 (60.4) |
83 (55.3) |
|
Patients having ≥1 TEAEs n (%) * |
52 (51.0) |
29 (60.4) |
81 (54.0) |
|
Drug related |
5 (4.9) |
0 |
5 (3.3) |
|
Patients having ≥1 TEAEs (PT) |
|||
|
Anemia |
8 (7.8) |
6 (12.5) |
14 (9.3) |
|
Pyrexia |
7 (6.8) |
6 (12.5) |
13 (8.6) |
|
Dizziness |
3 (2.9) |
3 (6.2) |
6 (4) |
|
Headache |
5 (4.9) |
4 (8.3) |
9 (6) |
|
Abdominal pain |
3 (2.9) |
2 (4.1) |
5 (3.3) |
|
Colitis ulcerative |
7 (6.8) |
0 |
7 (4.6) |
|
Arthralgia |
4 (3.9) |
0 |
4 (2.6) |
|
Vomiting |
0 |
4 (8.3) |
4 (2.6) |
|
Aphthous ulcer |
0 |
3 (6.3) |
3 (2) |
|
Asthenia |
3 (2.9) |
1 (2.1) |
4 (2.6) |
|
Patients with ≥1 AEs* by intensity# |
|||
|
Mild |
46 (45.1) |
26 (54.2) |
72 (48.0) |
|
Moderate |
7 (6.9) |
5 (10.4) |
12 (8.0) |
|
Severe |
3 (2.9) |
0 |
3 (2.0) |
|
Patients with ≥1 SAEs, n (%) * |
6 (5.9) |
2 (4.2) |
8 (5.3) |
|
Drug related |
2 (1.9) |
0 |
2 (1.3) |
|
Small intestine obstruction |
1 (0.9) |
1 (2.1) |
2 (1.3) |
|
Patients having ≥1 expected ADR |
5 (4.9) |
0 |
5 (3.3) |
|
Pulmonary tuberculosis |
1 (0.9) |
0 |
1 (0.6) |
|
Tuberculous pleurisy |
1 (0.9) |
0 |
1 (0.6) |
|
Patients having ≥1 AESI |
4 (3.9%) |
0 |
4 (2.7) |
|
Exposure -adjusted Incidence Rates, (95% CI) § |
|||
|
AEs |
3.94 (2.87–5.01) |
3.37 (2.12–4.62) |
3.72 (2.91–4.54) |
|
TEAEs |
3.94 (2.87–5.01) |
3.37 (2.12–4.62) |
3.72 (2.91–4.54) |
|
SAEs |
0.18 (0.04–0.32) |
0.04 (0.04–0.13) |
0.13 (0.03–0.22) |
|
ADRs |
0.15 (0.02–0.27) |
0 |
0.09 (0.01–0.17) |
|
Note 1: Percentages are based on number of subjects in safety analysis set. (N=150). Same patients can be counted multiple times under different categories but once within category. All AEs, TEAEs, ADRs, and AESIs are represented as patient count (% of patient). Note 2: *AEs were coded using the MedRA version 24.0. Data presented for patients with TEAE (≥2%) and SAE (≥1%). Note 3: #Mild (the event is transient and easily tolerated by the patient), Moderate (the event causes the patient discomfort and interrupts the patient’s usual activities), Severe (the event causes considerable interference with the patient’s usual activities). Note 4: §95% CI was calculated by using two-sided Poisson confidence interval: IR ±1.96*sqrt (IR/T). Abbreviations: ADR: Adverse Drug Reaction; AE: Adverse Event; CD: Crohn’s Disease; CI: Confidence Interval; IR: Incidence Rate; N/n: Number of Patients; SAE: Serious Adverse Event; TEAE: Treatment-Emergent Adverse Event; UC: Ulcerative Colitis. MedDRA: Medical Dictionary for Regulatory Activities; PT: Preferred Term |
|||
The EAIR for all AEs was 3.72, with a slightly higher rate in UC patients (3.94) than in CD patients (3.37). The EAIR for SAEs was 0.13 overall, with a higher rate in UC (0.18) compared to CD (0.04). No ADRs were reported in CD patients, and only 2 UC patients had unexpected ADRs, leading to an EAIR of 0.04 for such events.
Vedolizumab was discontinued due to AEs in 8 patients (5.3%), including 6 UC patients and 2 CD patients. In UC, reasons for discontinuation included infections (cytomegalovirus, pulmonary tuberculosis) and serious medical events (rectal adenocarcinoma, hypertension). In CD, the reasons were intestinal obstruction and fungal skin infection.
The most common outcome was hospitalization, with 4 patients discontinuing the study drug due to SAEs. No deaths occurred during the study.
Overall, four UC patients (2.7%) experienced at least one AESI, including pulmonary tuberculosis, tuberculous pleurisy, rectal adenocarcinoma, and hypertension. All these events were reported in 1 patient, each. Pulmonary tuberculosis, tuberculous pleurisy and hypertension were considered serious and linked to the study drug, while rectal adenocarcinoma was considered unrelated to the study drug.
Patients reported abnormal clinically significant (CS) laboratory values that were recorded as AEs. These included cases of anemia, iron deficiency anemia, and hypoproteinemia. However, the Investigator determined that none of these events were related to the study drug. No major clinically significant changes were observed in ECG results throughout the study. Regarding vital signs, hypertension was reported in 2 patients (1.3%), while pyrexia occurred in 14 patients (9.3%). All instances of pyrexia were resolved by the end of the study (EOS).
Discussion
This open-label, single-arm, Phase 4 study was designed to evaluate the safety and effectiveness of vedolizumab IV in Indian patients with UC or CD, both of whom had inadequate responses or intolerance to conventional therapies or TNF-α antagonist treatments. Vedolizumab, an integrin inhibitor, is widely utilized for the treatment of UC and CD in various regions globally [12–14]. This study was conducted to generate region-specific data to support its use in India.
The study included 150 patients with a median age of 36 years (range: 18–63). UC patients were older (median: 39 years) than CD patients (median: 31 years), consistent with findings from the GEMINI 1 study, where the median age for UC was 40 years, and the GEMINI 2 study, where it was 36 years for CD. The cohort comprised 55.3% males overall, with higher proportions in UC (65.7%) than CD (33.3%). In comparison, GEMINI 2 reported 46.6% male patients in CD, and GEMINI 1 reported 58% in UC. The duration of disease ranged from newly diagnosed cases to 34.3 years, with a mean of 9.6 years, aligning closely with the GEMINI 2 study (mean: 9.2 years for CD). However, the current study reported a longer disease duration for UC compared to GEMINI 1 (mean: 6.8 years). Most patients (93.3%) had disease localized to the large intestine, including all UC patients and 79.2% of CD patients. Disease severity scores reflected moderate to severe activity, with a mean Full Mayo Score of 8.64 for UC (similar to GEMINI 1’s 8.6) and a mean HBI score of 10.90 for CD [15,16].
The safety profile in this study was consistent with that observed in global clinical trials [14–16]. No deaths were reported, and no new safety concerns emerged. Safety analysis among patients revealed that 55.3% of patients experienced at least one AE, with a slightly higher incidence observed in CD patients (60.4%) compared to UC patients (52.9%). The overall AE incidence in this study was comparable to other clinical trials evaluating vedolizumab. Most AEs were mild, with severe AEs occurring in only 2.0% of patients, all of whom were in the UC group. The VARSITY trial, a Phase 3b, double-blind, active-controlled study in moderate-to-severe UC patients, reported an AE rate of 62.7%, while the pooled data from the GEMINI 1 and 2 trials showed an AE rate of 84% [14–16]. In the current study, the event rate for UC and CD was 3.94 and 3.37 per 100 patient-years, respectively, indicating a consistent safety profile across both diseases.
Among the most frequently reported AEs, GI disorders were the most common, accounting for 22.0% of all events, with anemia being the most prevalent extra-intestinal manifestation, reported in 10.7% of patients. Anemia is commonly associated with IBD and can be a marker of disease activity, as it tends to recur even after treatment is successful. The incidence of SAEs was 5.3% overall, with 5.9% in UC patients and 4.2% in CD patients. These figures are lower than those reported in the VARSITY trial (11%), the Phase 3 study by Amiot et al. (8.2%), and the pooled data from the GEMINI trials (19%) [14–17].
Serious AEs leading to study discontinuation were relatively uncommon, with only 5.9% of UC patients and 4.2% of CD patients discontinuing due to AEs. These rates are comparable to those reported by Amiot et al. (5.1%), and numerically lower compared to the pooled data from GEMINI 1 and 2 clinical trials (9%) [15–17].
In terms of effectiveness, the primary endpoint was clinical response, and secondary endpoints included clinical remission, mucosal healing, endoscopic response, and improvement in QoL. Clinical response, defined as a reduction in disease activity, was achieved in more than half of UC patients (54.9%) by Week 14, with the response rate improving to 73.5% by Week 46. In CD patients, the clinical response rate was high at 72.9% by Week 14 and was maintained at 68.8% by Week 46. Similarly, in the GEMINI 2 study conducted among an Asian cohort with CD reported that 63.6% of patients achieved a clinical response during the maintenance phase, while another study showed a clinical response rate of 56.6% in UC patients during the same phase [15,18]. The clinical remission rates were promising, with 39.2% of UC patients and 47.9% of CD patients in remission at Week 14. By Week 46, this proportion increased to 52.0% for UC patients and 56.3% for CD patients, indicating that vedolizumab was effective in inducing clinical remission over the treatment period. These results were comparable or slightly better than those observed in previous studies. For example, a 12-month study by Christensen et al. reported clinical remission rates of 51% at Week 14 for UC patients and 38% for CD patients [19]. Additionally, a systematic review of real-world data found remission rates of 32% for UC patients and 30% for CD patients at Week 14 [20]. These findings suggest that vedolizumab may offer better effectiveness in the Indian population than what has been observed globally.
Mucosal healing and endoscopic response were assessed in a subgroup of patients who underwent endoscopic procedures. However, only 42 patients (32 UC and 10 CD) underwent endoscopy, and the rates of mucosal healing and endoscopic response were modest. Mucosal healing was achieved in 22.5% of UC patients and 8.3% of CD patients, while endoscopic responses were observed in 21.6% of UC patients and 12.5% of CD patients. These results are lower than those reported in other studies, such as the Phase 3 VISIBLE 1 trial, which reported mucosal healing in 53.7% of patients at Week 52, and the GEMINI trials, which showed mucosal healing in 45.5% of UC patients [15,16,21]. A non-responder imputation method was applied for missing or dropout cases when calculating mucosal healing and endoscopic response rates. This approach, combined with the optional nature of endoscopy assessments, may have contributed to an apparent underestimation of mucosal healing rates.
The current study reported infections and infestations like COVID-19, cytomegalovirus infections, fungal skin infections, gastroenteritis, pulmonary tuberculosis, rectal abscess, tuberculous pleurisy, and urinary tract infections (UTI) in 6% (9) patients (5.9% (6) in UC; 6.3% (3) in CD). Post-marketing descriptive analysis published in 2020 reported 5,876 cases of infection in vedolizumab-treated patients, with 19% classified as serious infections [22]. The current study reported cases of COVID-19, gastroenteritis, UTI, and pulmonary tuberculosis, with tuberculosis being a particular concern due to India’s high prevalence of the disease. Despite active and latent tuberculosis being exclusion criteria, 2 cases were reported in the study, highlighting the importance of screening for tuberculosis before initiating vedolizumab treatment.
Fecal calprotectin and serum C-reactive protein (CRP) levels decreased significantly from baseline, indicating a reduction in inflammation over the treatment period. Similar to studies conducted by Feagan et al. and Sandborn et al. observed greater improvements in fecal calprotectin and CRP concentrations in patients who received vedolizumab [15,16]. These results were further supported by improvements in the quality of life, as measured by the patient-reported quality of life (SIBDQ) scores. The SIBDQ scores of this study showed improvement over the period with both median absolute and percentage changes indicating positive effects, particularly by Week 46, which is similar to reports from the Greek VEDO-IBD representing the UC cohort with improved scores by week 54 [23]. Subgroup analysis revealed that BNP patients showed a higher clinical response and remission rate compared to BEP patients, though the small size of the BEP group limits statistical interpretation.
Overall, the findings indicate positive treatment effects across various effectiveness endpoints, with improvements in clinical response, remission, and quality of life for both UC and CD patients. However, the small sample size in certain subgroups and the variability in dropout rates should be considered when interpreting these results.
Conclusion
In conclusion, this Phase 4 study provides important evidence supporting the safety and effectiveness of vedolizumab in the Indian population with UC or CD. The drug demonstrated a favorable safety profile, with most adverse events being mild and manageable, and the effectiveness results were promising, with significant improvements in clinical response, remission, and quality of life. While mucosal healing rates were lower than those observed in other studies, the overall findings suggest that vedolizumab is a viable treatment option for patients with moderate-to-severe UC or CD in India.
Acknowledgement
The authors would like to thank Dr. Vandana Govindan, and Vaishali Kumar SM from IQVIA, India, for their medical writing, editing, and technical support.
Funding
The study was funded by Takeda Biopharmaceuticals India Private Limited.
Data Sharing Statement
The datasets will be provided after its de-identification, in compliance with applicable privacy laws, data protection and requirements for anonymization.
Author Disclosures
Gaurav Bahuguna and Avi Sharma are employees of Takeda Biopharmaceuticals India Private Limited, India. Sandeep Arora was the former Head of Medical Affairs at Takeda Biopharmaceuticals India Private Limited, India and may have stocks in Takeda. All other authors have no conflict of interest.
Author Contributions
All authors participated in the conception and design, acquisition, analysis and interpretation of data, critical manuscript revision for significant intellectual content, final approval, and consensus.
Declaration of Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
2. Zhou JL, Bao JC, Liao XY, Chen YJ, Wang LW, Fan YY, et al. Trends and projections of inflammatory bowel disease at the global, regional and national levels, 1990-2050: a bayesian age-period-cohort modeling study. BMC Public Health. 2023 Dec 14;23(1):2507.
3. Borowitz SM. The epidemiology of inflammatory bowel disease: Clues to pathogenesis? Front Pediatr. 2023 Jan 17;10:1103713.
4. Dutta AK. Indian Journal of Gastroenterology—January–February 2024 issue highlights. Indian J. Gastroenterol. 2024 Feb;43(1):1–6.
5. Khosla SN, Girdhar NK, Lal S, Mishra DS. Epidemiology of ulcerative colitis in hospital and select general population of northern India. J Assoc Physicians India. 1986 Jun;34(6):405–7.
6. Sood A, Midha V, Sood N, Bhatia AS, Avasthi G. Incidence and prevalence of ulcerative colitis in Punjab, North India. Gut. 2003 Nov;52(11):1587–90.
7. Jain M, Venkataraman J. Inflammatory bowel disease: An Indian perspective. Indian J Med Res. 2021 Apr;153(4):421–30.
8. Hanauer SB, Feagan BG, Lichtenstein GR, Mayer LF, Schreiber S, Colombel JF, et al ACCENT I Study Group. Maintenance infliximab for Crohn's disease: the ACCENT I randomised trial. Lancet. 2002 May 4;359(9317):1541–9.
9. Rutgeerts P, Sandborn WJ, Feagan BG, Reinisch W, Olson A, Johanns J, et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2005 Dec 8;353(23):2462-76.
10. Selinger CP, Rosiou K, Lenti MV. Biological therapy for inflammatory bowel disease: cyclical rather than lifelong treatment? BMJ Open Gastroenterol. 2024 Feb 10;11(1):e001225.
11. Soler D, Chapman T, Yang LL, Wyant T, Egan R, Fedyk ER. The binding specificity and selective antagonism of vedolizumab, an anti-alpha4beta7 integrin therapeutic antibody in development for inflammatory bowel diseases. J Pharmacol Exp Ther. 2009 Sep;330(3):864–75.
12. Patel D, Martin S, Luo M, Ursos L, Lirio RA, Kamble P, et al. Real-World Effectiveness of Vedolizumab Dose Escalation in Patients With Inflammatory Bowel Disease: A Systematic Literature Review. Crohns Colitis 360. 2022 Jul 8;4(3):otac020.
13. Attauabi M, Madsen GR, Bendtsen F, Seidelin JB, Burisch J. Vedolizumab as the first line of biologic therapy for ulcerative colitis and Crohn's disease - a systematic review with meta-analysis. Dig Liver Dis. 2022 Sep;54(9):1168–78.
14. Sands BE, Peyrin-Biroulet L, Loftus EV Jr, Danese S, Colombel JF, Törüner M, et al. VARSITY Study Group. Vedolizumab versus Adalimumab for Moderate-to-Severe Ulcerative Colitis. N Engl J Med. 2019 Sep 26;381(13):1215–26.
15. Feagan BG, Rutgeerts P, Sands BE, Hanauer S, Colombel JF, Sandborn WJ, et al. GEMINI 1 Study Group. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013 Aug 22;369(8):699–710.
16. Sandborn WJ, Feagan BG, Rutgeerts P, Hanauer S, Colombel JF, Sands BE, et al. GEMINI 2 Study Group. Vedolizumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. 2013 Aug 22;369(8):711–21.
17. Amiot A, Grimaud JC, Peyrin-Biroulet L, Filippi J, Pariente B, Roblin X, et al. Observatory on Efficacy and of Vedolizumab in Patients With Inflammatory Bowel Disease Study Group; Groupe d'Etude Therapeutique des Affections Inflammatoires du tube Digestif. Effectiveness and Safety of Vedolizumab Induction Therapy for Patients With Inflammatory Bowel Disease. Clin Gastroenterol Hepatol. 2016 Nov;14(11):1593–601.e2.
18. Banerjee R, Chuah SW, Hilmi IN, Wu DC, Yang SK, Demuth D, et al. Efficacy and safety of vedolizumab in Crohn's disease in patients from Asian countries in the GEMINI 2 study. Intest Res. 2021 Jan;19(1):83–94.
19. Christensen B, Colman RJ, Micic D, Gibson PR, Goeppinger SR, Yarur A, et al. Vedolizumab as Induction and Maintenance for Inflammatory Bowel Disease: 12-month Effectiveness and Safety. Inflamm Bowel Dis. 2018 Mar 19;24(4):849–60.
20. Schreiber S, Dignass A, Peyrin-Biroulet L, Hather G, Demuth D, Mosli M, et al. Systematic review with meta-analysis: real-world effectiveness and safety of vedolizumab in patients with inflammatory bowel disease. J Gastroenterol. 2018 Sep;53(9):1048–64.
21. Sandborn WJ, Baert F, Danese S, Krznarić Ž, Kobayashi T, Yao X, et al. Efficacy and Safety of Vedolizumab Subcutaneous Formulation in a Randomized Trial of Patients With Ulcerative Colitis. Gastroenterology. 2020 Feb;158(3):562–72.e12.
22. Cohen RD, Bhayat F, Blake A, Travis S. The Safety Profile of Vedolizumab in Ulcerative Colitis and Crohn's Disease: 4 Years of Global Post-marketing Data. J Crohns Colitis. 2020 Feb 10;14(2):192–204.
23. Bamias G, Kokkotis G, Gizis M, Kapizioni C, Karmiris K, Koureta E, et al. Predictors of Response to Vedolizumab in Patients with Ulcerative Colitis: Results from the Greek VEDO-IBD Cohort. Dig Dis Sci. 2022 Mar;67(3):1007–17.
